GlobeNewswire04.25.19
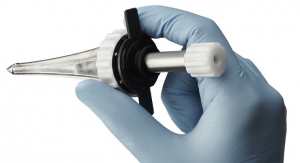
Orchestra BioMed Inc., a biomedical innovation company developing advanced therapeutic solutions to address major unmet healthcare needs, has secured Breakthrough Device designation by the U.S. Food and Drug Administration (FDA) for its Virtue Sirolimus-Eluting Balloon for the treatment of coronary in-stent restenosis (ISR).
Breakthrough Designation is granted to certain medical devices and device-led combination products that provide for a more effective treatment of life-threatening or irreversibly debilitating diseases. It enables manufacturers to provide patients and health care providers with timely access to medical devices by expediting their development, assessment, and review. Benefits of this designation include frequent interactions and feedback from FDA during the premarket review phase. Through this program, Orchestra BioMed can expect prioritized review of its submission for Virtue SEB.
“In Virtue SEB, we finally have a therapeutic innovation that truly warrants breakthrough designation for the treatment of coronary ISR, a particularly challenging condition for which available treatment options are limited,” commented Dean Kereiakes, M.D., FACC, FSCAI, medical director of The Christ Hospital Heart and Vascular Center and professor of Clinical Medicine at The Ohio State University. “The unique design of Virtue SEB provides a reliable way to apply the proven anti-proliferative, anti-restenotic benefits of sirolimus during balloon angioplasty without the potential hazards of a permanent metal implant or a balloon coating that may produce downstream particulates and micro-emboli. Clinical results at three-year follow-up with Virtue SEB in ISR are very promising and I am very much looking forward to utilizing this exciting new technology in clinical trials in the U.S.”
“We believe Virtue SEB addresses an important unmet clinical need and provides an improved treatment alternative for a patient population with limited options. It is the first and only non-coated angioplasty balloon that provides arterial delivery of sirolimus, the proven gold standard drug used on drug-eluting stents for preventing restenosis of treated arteries.” commented Darren R. Sherman, president, chief operating officer and founder of Orchestra BioMed. “We plan to fully leverage the benefits of FDA Breakthrough Device designation as we seek to accelerate the U.S. clinical and regulatory development of Virtue SEB with the goal of providing physicians and patients with the benefits of our novel therapeutic device.”
Breakthrough Designation is granted to certain medical devices and device-led combination products that provide for a more effective treatment of life-threatening or irreversibly debilitating diseases. It enables manufacturers to provide patients and health care providers with timely access to medical devices by expediting their development, assessment, and review. Benefits of this designation include frequent interactions and feedback from FDA during the premarket review phase. Through this program, Orchestra BioMed can expect prioritized review of its submission for Virtue SEB.
“In Virtue SEB, we finally have a therapeutic innovation that truly warrants breakthrough designation for the treatment of coronary ISR, a particularly challenging condition for which available treatment options are limited,” commented Dean Kereiakes, M.D., FACC, FSCAI, medical director of The Christ Hospital Heart and Vascular Center and professor of Clinical Medicine at The Ohio State University. “The unique design of Virtue SEB provides a reliable way to apply the proven anti-proliferative, anti-restenotic benefits of sirolimus during balloon angioplasty without the potential hazards of a permanent metal implant or a balloon coating that may produce downstream particulates and micro-emboli. Clinical results at three-year follow-up with Virtue SEB in ISR are very promising and I am very much looking forward to utilizing this exciting new technology in clinical trials in the U.S.”
“We believe Virtue SEB addresses an important unmet clinical need and provides an improved treatment alternative for a patient population with limited options. It is the first and only non-coated angioplasty balloon that provides arterial delivery of sirolimus, the proven gold standard drug used on drug-eluting stents for preventing restenosis of treated arteries.” commented Darren R. Sherman, president, chief operating officer and founder of Orchestra BioMed. “We plan to fully leverage the benefits of FDA Breakthrough Device designation as we seek to accelerate the U.S. clinical and regulatory development of Virtue SEB with the goal of providing physicians and patients with the benefits of our novel therapeutic device.”