Globe Newswire12.19.18
Nemaura Medical Inc., a medical technology company developing sugarBEAT as a non-invasive affordable and flexible Continuous Glucose Monitor (CGM) for use by people with diabetes and pre-diabetics, has developed predictive algorithms that will allow the inclusion of alarms in its sugarBEAT CGM.
This development is expected to make sugarBEAT more appealing to insulin users, a total addressable market valued at $50 billion. CGM adoption amongst people with diabetes is minimal and growing rapidly, with the United States the largest single market, with an estimated 2.6 percent (630,000 users) of all diagnosed diabetics using CGM in 2018, representing annualized growth of 117 percent.1
Over the course of the last 14 months Nemaura Medical has gathered and analysed data stemming from over 25,000 paired data points between sugarBEAT and blood serum samples taken using a venous catheter at 15 minute intervals for 12 consecutive hours. This analysis has led to substantial algorithm improvements, including predictive functionality, allowing alarms to be incorporated into sugarBEAT.
Results indicate the alarm functionality can predict glucose level five minutes in advance with 98 percent accuracy, 10 minutes in advance with 95 percent accuracy and 15 minutes in advance with 90 percent accuracy.
SugarBEAT is anticipated to receive CE Mark approval soon and launch in the United Kingdom shortly thereafter.
SugarBEAT has the potential to expand the use of CGM amongst people with diabetes and pre-diabetics given it is non-invasive and flexible, as compared to competitor CGMs, which are invasive and designed to be continuously worn for periods of up to seven days or longer.
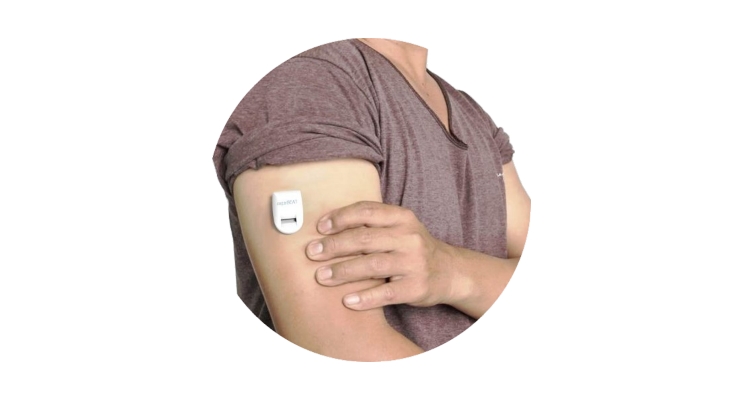
Nemaura Medical is a medical technology company developing sugarBEAT as a non-invasive, affordable and flexible Continuous Glucose Monitor (CGM) designed to help people with diabetes and pre-diabetics to better manage their glucose levels. Insulin users can adjunctively use sugarBEAT when calibrated with a finger-stick glucose reading. SugarBEAT consists of a daily disposable adhesive skin-patch connected to a rechargeable transmitter, with an app displaying glucose readings at five minute intervals for periods up to 24 hours.
Reference
1. PiperJaffray Company Note DXCM Sep 5, 2018
This development is expected to make sugarBEAT more appealing to insulin users, a total addressable market valued at $50 billion. CGM adoption amongst people with diabetes is minimal and growing rapidly, with the United States the largest single market, with an estimated 2.6 percent (630,000 users) of all diagnosed diabetics using CGM in 2018, representing annualized growth of 117 percent.1
Over the course of the last 14 months Nemaura Medical has gathered and analysed data stemming from over 25,000 paired data points between sugarBEAT and blood serum samples taken using a venous catheter at 15 minute intervals for 12 consecutive hours. This analysis has led to substantial algorithm improvements, including predictive functionality, allowing alarms to be incorporated into sugarBEAT.
Results indicate the alarm functionality can predict glucose level five minutes in advance with 98 percent accuracy, 10 minutes in advance with 95 percent accuracy and 15 minutes in advance with 90 percent accuracy.
SugarBEAT is anticipated to receive CE Mark approval soon and launch in the United Kingdom shortly thereafter.
SugarBEAT has the potential to expand the use of CGM amongst people with diabetes and pre-diabetics given it is non-invasive and flexible, as compared to competitor CGMs, which are invasive and designed to be continuously worn for periods of up to seven days or longer.
Nemaura Medical is a medical technology company developing sugarBEAT as a non-invasive, affordable and flexible Continuous Glucose Monitor (CGM) designed to help people with diabetes and pre-diabetics to better manage their glucose levels. Insulin users can adjunctively use sugarBEAT when calibrated with a finger-stick glucose reading. SugarBEAT consists of a daily disposable adhesive skin-patch connected to a rechargeable transmitter, with an app displaying glucose readings at five minute intervals for periods up to 24 hours.
Reference
1. PiperJaffray Company Note DXCM Sep 5, 2018