Business Wire11.19.18
Tandem Diabetes Care Inc., a medical device company and manufacturer of the only touchscreen insulin pumps with continuous glucose monitoring (CGM) integration, has received a Heath Canada medical device license for the t:slim X2 Insulin Pump. The t:slim X2 Pump features Dexcom G5 Mobile CGM integration,1 making it the only CGM-integrated insulin pump approved in Canada for making daily diabetes treatment decisions without fingersticks.2 The company plans to launch the t:slim X2 Insulin Pump on a province-by-province basis throughout the fourth quarter of 2018 pending the completion of pre-launch activities, including product registration with provincial health programs.
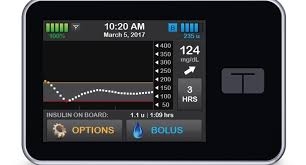
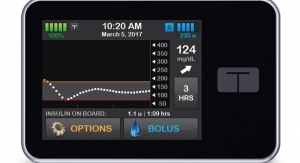
The simple-to-use t:slim X2 Insulin Pump includes advanced features like a large color touchscreen, rechargeable battery, USB connectivity and watertight construction (IPX7).3 As the only insulin pump integrated with Dexcom G5 Mobile CGM, dynamic glucose data can be easily accessed and shared using a compatible mobile device.4 The t:slim X2 Pump is up to 38 percent smaller than other insulin pumps and holds up to 300 units of insulin.5
“People with diabetes in Canada have been asking for our simple-to-use pump technology since the inception of Tandem and I am thrilled that we are now able to offer a fresh, new choice in insulin pump therapy,” said Kim Blickenstaff, president and CEO of Tandem Diabetes Care. “This launch is especially important since insulin pump options in Canada have become increasingly limited in recent years, and this approval will allow us to bring the benefits of our technology to more people with diabetes.”
According to Diabetes Canada, there are more than 3.4 million Canadians living with diabetes, of which approximately 10 percent have type 1 diabetes.6 Insulin pumps and diabetes supplies are a covered benefit for qualifying recipients of disability tax credit, assistive devices programs, and qualifying expenses for registered disability savings plans offered in various forms across Canadian provinces.7
Tandem Diabetes Care Inc. is a medical device company dedicated to improving the lives of people with diabetes through innovation and customer experience. Tandem takes a user-centric approach to the design, development, and commercialization of products for people with diabetes who use insulin. Tandem manufactures and sells the t:slim X2 Insulin Pump, which features integrated continuous glucose monitoring. Tandem is based in San Diego, Calif.
Tandem Diabetes Care, t:slim X2, and t:simulator are trademarks, registered trademarks, and/or the subject of a pending trademark application in the United States and other territories worldwide. Dexcom and Dexcom G5 are registered trademarks of Dexcom Inc. All other trademarks are the property of their respective owners.
References
1. Dexcom G5 Mobile CGM System sold separately. The Dexcom G5 Mobile CGM transmitter can only be paired with one medical device (either a Dexcom receiver or t:slim X2 Pump) and one consumer device (phone or tablet) at the same time.
2. If glucose alerts and CGM readings do not match symptoms or expectations, use a blood glucose meter to make diabetes treatment decisions. A fingerstick is needed every 12 hours to calibrate.
3. Tested to a depth of three feet for 30 minutes.
4. Following your shared data requires the Dexcom Follow app.
5. Thirty-eight percent smaller than MiniMed 630G and 670G and at least 28 percent smaller than MiniMed 530G, Animas Vibe and Omnipod System. Data on file, Tandem Diabetes Care.
6. Estimated diabetes statistics in Canada are generated by the Canadian Diabetes Cost Model; 2015.
7. http://www.diabetes.ca/about-cda/public-policy-position-statements/access-to-diabetes-medication-supplies-medical-devices.
The simple-to-use t:slim X2 Insulin Pump includes advanced features like a large color touchscreen, rechargeable battery, USB connectivity and watertight construction (IPX7).3 As the only insulin pump integrated with Dexcom G5 Mobile CGM, dynamic glucose data can be easily accessed and shared using a compatible mobile device.4 The t:slim X2 Pump is up to 38 percent smaller than other insulin pumps and holds up to 300 units of insulin.5
“People with diabetes in Canada have been asking for our simple-to-use pump technology since the inception of Tandem and I am thrilled that we are now able to offer a fresh, new choice in insulin pump therapy,” said Kim Blickenstaff, president and CEO of Tandem Diabetes Care. “This launch is especially important since insulin pump options in Canada have become increasingly limited in recent years, and this approval will allow us to bring the benefits of our technology to more people with diabetes.”
According to Diabetes Canada, there are more than 3.4 million Canadians living with diabetes, of which approximately 10 percent have type 1 diabetes.6 Insulin pumps and diabetes supplies are a covered benefit for qualifying recipients of disability tax credit, assistive devices programs, and qualifying expenses for registered disability savings plans offered in various forms across Canadian provinces.7
Tandem Diabetes Care Inc. is a medical device company dedicated to improving the lives of people with diabetes through innovation and customer experience. Tandem takes a user-centric approach to the design, development, and commercialization of products for people with diabetes who use insulin. Tandem manufactures and sells the t:slim X2 Insulin Pump, which features integrated continuous glucose monitoring. Tandem is based in San Diego, Calif.
Tandem Diabetes Care, t:slim X2, and t:simulator are trademarks, registered trademarks, and/or the subject of a pending trademark application in the United States and other territories worldwide. Dexcom and Dexcom G5 are registered trademarks of Dexcom Inc. All other trademarks are the property of their respective owners.
References
1. Dexcom G5 Mobile CGM System sold separately. The Dexcom G5 Mobile CGM transmitter can only be paired with one medical device (either a Dexcom receiver or t:slim X2 Pump) and one consumer device (phone or tablet) at the same time.
2. If glucose alerts and CGM readings do not match symptoms or expectations, use a blood glucose meter to make diabetes treatment decisions. A fingerstick is needed every 12 hours to calibrate.
3. Tested to a depth of three feet for 30 minutes.
4. Following your shared data requires the Dexcom Follow app.
5. Thirty-eight percent smaller than MiniMed 630G and 670G and at least 28 percent smaller than MiniMed 530G, Animas Vibe and Omnipod System. Data on file, Tandem Diabetes Care.
6. Estimated diabetes statistics in Canada are generated by the Canadian Diabetes Cost Model; 2015.
7. http://www.diabetes.ca/about-cda/public-policy-position-statements/access-to-diabetes-medication-supplies-medical-devices.