Sam Brusco, Associate Editor11.26.18
Flu season has officially begun. Inevitably, any doctor or pharmacy visit taken over the past few weeks has included “have you gotten your flu shot yet?” somewhere in the dialogue.
Misconceptions about vaccines aside, certain scientific and timing factors can make developing an effective influenza vaccine challenging. Current U.S. Food and Drug Administration (FDA) regulatory requirements allow influenza vaccines to be grown in chicken eggs or cells. According to the CDC, today, the majority of influenza vaccines are still grown in chicken eggs. Some influenza viruses—like H3N2—grow poorly in eggs, making it difficult to obtain candidate vaccine viruses. Further, in some years, certain flu viruses may not appear and spread until later in the season, making it difficult to prepare a candidate vaccine virus in time for vaccine production.
Research undertaken at Georgia State University last year is attempting to boost flu shots’ effectiveness. Georgia State researchers developed a biodegradable microneedle patch and a protein constructed from sequences of influenza virus subtypes, which could improve conventional influenza vaccines’ effectiveness. Because circulating flu viruses can rapidly undergo antigenic changes (small genetic alterations that happen over time as the virus replicates), a fast, cost-efficient vaccination method is necessary for broader cross-protection against seasonal and pandemic virus infection.
To achieve this goal, the patch is designed to deliver vaccines into the skin’s dermis and epidermis, and may improve the vaccine’s ability to provoke immune response. The skin is a potent site for vaccination due to an abundance of blood and lymphatic vessels, and many different types of immune cells are present. The patch was given as a “booster” vaccine to mice four weeks after an inactive flu vaccine, then introduced to H1N1 and H3N2 influenza viruses. Mice receiving the microneedle patch displayed significantly enhanced survival and cellular immune responses, as well as long-term antibody-mediated immunity.
Further, microneedle vaccines are much friendlier to patients. Microneedles penetrate the upper layer of the skin, just enough to avoid touching the nerves. A successful microneedle product could have major implications for healthcare delivery—the chronically ill would not have to take daily injections. They also offer a hygiene benefit. Since the patch doesn’t enter the bloodstream, there is less risk of spreading infections.
Implantable devices also offer a number of advantages for improved drug delivery. An implantable pump or drug-eluting implant allows site-specific administration wherever the pharmaceutical is most needed. It can also allow significantly lower doses of the medicine, which can minimize potential side effects. Brachytherapy, for example, involves radioactive implants placed into or near a brain tumor during surgery. Since the radiation only travels a short distance within the body, damage to nearby tissue—a concern common to radiation therapy—is reduced.
The brain is particularly challenging for drug delivery because of the blood-brain barrier (BBB), a highly selective semipermeable border separating circulating blood from the brain and extracellular fluid in the central nervous system. Though it allows water, some gases, glucose, and amino acids to pass through, large molecules (i.e., pharmaceuticals) are restricted access.
In addition, the pharmaceuticals themselves can make the method of delivery challenging. Chemotherapeutic agents can be particularly damaging, and oncologists are working hard to find ways to reduce the chemo dose while effectively treating a tumor. Targeted drug delivery via implantable pharmaceutical vehicles are one way to tackle this issue.
“We have started seeing requirements for drug delivery systems into more challenging areas,” said Ed White, Manager of OEM & International Sales Operations for B. Braun Medical Inc., a Bethlehem, Pa.-based firm that specializes in infusion therapy and pain management and develops, manufactures, and markets medical products and services for the healthcare industry. “We are developing long-term implantable devices to serve this need as a drug delivery method.”
Medical devices have been smartening up for years, and drug delivery technology has certainly followed suit. An intelligent drug delivery system may include functions like alerts to remind users to dose, wireless connectivity to an app to provide historical dosing data and communicate with clinicians, or analytical software to optimize dosage based on external factors. The drive to beef up drug delivery technology with these functions has fostered collaboration between companies who develop the delivery platform and those that specialize in digital services.
For example, in late October, Ypsomed—a developer and manufacturer of injection and infusion systems for self-medication—began a partnership with global health technology firm Philips. The two companies will jointly develop digital self-medication adherence monitoring services based on Ypsomed’s connected devices and Philips’ cloud-based HealthSuite digital platform. Ypsomed’s SmartServices will provide a new turnkey digital solution to monitor medication adherence and enable smart device management for its pharmaceutical customers.
“Our partnership with Philips aims at embedding smart devices in a digital health ecosystem that simplifies access to therapy-relevant medication adherence data,” commented Ulrike Bauer, senior vice president of Ypsomed. “This will allow our pharmaceutical customers to focus on building therapy-specific digital solutions for patients, caregivers, and physicians.”
Philips’ HealthSuite digital platform is purpose-built to streamline complex healthcare challenges, tools and services for ease-of-use integration, digital lifecycle management, and end-to-end security. HealthSuite’s tools help support medication delivery with a framework to build contextual predictive analytics, decision support algorithms, as well as task automation and communications coordination. It also provides management, update, monitoring, and collection of data from smart devices. The new digital services arising from the partnership aim to address device-oriented challenges and support self-medication services for patients with chronic illnesses like diabetes, multiple sclerosis, and rheumatoid arthritis.
“The use of electronics in devices and apps as part of the patient interface have the potential to improve the patient experience, reduce use errors, and ensure adherence,” noted Bill Welch, chief technology officer of Phillips-Medisize, a Molex company. Phillips-Medisize is a Hudson, Wis.-based global outsource provider of design and manufacturing services, with significant business in the drug delivery, medical device, and primary pharmaceutical packaging markets. “These are all aimed at greater patient engagement to improve outcomes, which ultimately go back to drug effectiveness.”
Medtronic’s SynchroMed II pump and catheter, for example, are part of an implantable, programmable drug infusion system. SynchroMed II was initially indicated to administer Infumorph (a morphine sulfate sterile solution) to treat chronic intractable pain and Prialt (a ziconotide sterile solution) to manage severe chronic pain. It worked by injecting these pharmaceuticals into the intrathecal (or subarachnoid) space so it reaches the cerebrospinal fluid. In addition to the pump and catheter, the system comes equipped with a tablet with touchscreen interface to be used as a clinician programmer. The programmer offers workflow guides and steps for refill and implantation, auto-calculations for volume and rates, and visuals to accompany prime bolus and flex dosing. It also connects wirelessly to SynchroMed II’s communicator.
In late January, SynchroMed II was also approved by the FDA to treat pulmonary arterial hypertension by delivering Remodulin into the superior caval-atrial junction (the joint between the superior vena cava and the heart). In this case, the pump is implanted under the skin of the abdomen. It can be refilled via injection through the skin whenever the reservoir runs low.
In late October, the FDA also approved the myPTM Personal Therapy Manager for SynchroMed II for chronic pain patients. The application runs on a touchscreen Samsung J3 smart device that can be customized to assist patients in managing their pain. Clinicians can set daily therapeutic doses, enabling on-demand bolusing within pre-established limits. myPTM makes bolus delivery clear, offers access to therapy details, and provides lockout alerts if patient demand exceeds prescribed limits. Physicians can also use myPTM to view reports providing the necessary insights to track progress and collaborate on therapy goals with their patients.
Insulin pumps are also receiving high-tech upgrades to further assist patients in successfully managing their diabetes. In June, the FDA gave a nod to Tandem Diabetes Care’s t:slim X2 Insulin Pump with Basal-IQ technology, an automated delivery feature with predictive low glucose suspend (PLGS) technology to help reduce the frequency and duration of hypoglycemic events. To determine optimal insulin dosage, Basal-IQ looks 30 minutes into the future to predict where glucose levels are trending. When low glucose is detected (either predicted to drop below 80 mg/dL or currently below 70 mg/dL), it suspends insulin delivery. It automatically resumes delivery once glucose levels begin to rise.
Further simplifying delivery for diabetics, t:slim X2 with the Basal-IQ feature comes integrated with the Dexcom G6 continuous glucose monitoring (CGM) system. The Dexcom G6 CGM notably requires no fingersticks for mealtime dosing or calibration, and can share real-time CGM data with up to five followers. Patients already using the t:slim X2 pump prior to the launch of Basal-IQ can also receive remote software updates to add the predictive algorithm to their pumps using a personal computer.
“Digital health is everywhere,” said Alexa Konstantinos, vice president of strategic marketing, medical devices & health analytics for Battelle, a Columbus, Ohio-based firm that conducts research and development, manages laboratories, designs and manufactures products, and delivers critical services to the national security, health and life sciences, and energy and environmental industries. “Devices are getting connected and all the major players are looking to artificial intelligence (AI) as the next frontier to make sense of the data. Right now, connectedness primarily involves tracking ‘historical’ compliance/dosing data. But in the not-too-distant future, much more sophisticated AI algorithms will also combine wellness data, environmental and nutritional data, sleep patterns, phone usage, and other cues to predict dosing needs and health status.”
Though not yet incorporated directly into a drug delivery device, the Sugar.IQ smart diabetes assistant leverages AI and analytical technologies to assist diabetic patients who undergo multiple daily insulin injections, and may form a framework for the type of software that could become standard in drug delivery technology.
Jointly developed by Medtronic and IBM Watson Health and available for Medtronic Guardian Connect CGM users, the Sugar.IQ diabetes assistant continually analyzes how a patient’s glucose level responds to food intake, insulin dosages, daily routines, and other information keyed in by the user. That way, people with diabetes can uncover patterns affecting glucose levels to help make adjustments to their regimen. The app’s advanced analytics and AI offers a full picture of current glucose levels, providing insights and individualized guidance for those on multiple daily insulin injections. According to data presented at this year’s American Diabetes Association Scientific Sessions, those using Sugar.IQ spent 36 minutes more per day in a healthy glucose range than before using the app, including a half hour less time in hyperglycemia, and six minutes less time in hypoglycemia—translating to over nine additional days a year spent in a healthy glucose range.
As it stands, Sugar.IQ is used only indirectly in conjunction with insulin delivery to determine how the medication and lifestyle factors affect glucose levels. However, as the medical device landscape continues to crave holistic solutions to chronic disease management, it is that not far-fetched to say analytical software like Sugar.IQ could be used either in tandem with or be integrated into a therapeutic device for other conditions. Chronic pain management, for instance, also relies on tweaking medication, lifestyle choices, and environmental factors. In the future, an accompanying app could come included with a pain management device, or perhaps even be part of its functionality.
When Industries Collide
Because pharmaceuticals are involved, manufacturing a drug delivery device or combination product cannot be approached with the same protocols as a device that consists purely of technology. For successful drug delivery or combination product commercialization, the device company and pharmaceutical firm must work closely throughout the development cycle. The need for improved drug delivery systems has fostered stronger collaboration between the two industries on an unprecedented scale.
According to research done by PMMI, The Association for Packaging and Processing Technologies, collaboration between medical device and pharmaceutical companies is one of the top five trends influencing growth in the medtech and pharmaceutical industries (notably, just behind the increased use of outside contracting services). Rapid expansion in the biologic drug market is a partial driver of this, with protein-based therapeutics like monoclonal antibodies for cancer treatment necessitating novel drug administration methods. These drugs are usually delivered via infusion or injection, which is less desirable from the patient’s point of view. As a result, pharmaceutical and medical device companies must collaborate to develop more convenient and less painful technological solutions.
“Pharma companies now work with medtech companies much earlier in the development phase,” stated Meredith Canty, director of drug delivery systems for SMC Ltd., a Somerset, Wis.-based global contract manufacturer of pharmaceutical and finished medical devices. “Years ago, the formulators would develop a drug product, then the drug delivery device teams would need to figure out a way to deliver it. Now the formulation and device teams work together much sooner in the program to deliver a more user-friendly experience. This requires multiple rounds of human factors testing with design iterations to enhance the user experience as much as possible.”
“As an integral part of therapy, devices are being considered much earlier in the drug delivery process, as opposed to years past when devices were inserted into the development process toward the end, or were as simplistic as a syringe and vial,” described Konstantinos. “There also has been a drive for several years to ensure devices are usable. Mere usability in the regulatory sense is no longer enough; to gain an advantage over competition, devices must now be preferred by end-users over alternatives, which can help drive better compliance to dosing regimens. This resonates throughout the development process from the very earliest conceptual stages.”
As the adoption and acceptance of self-administered therapies rises, patients are becoming more involved in the delivery and management of their own treatments. The demand for extended release and high concentration drug formulations are breeding more delivery system choices.
For example, patients may opt to use an auto-injector, a drug delivery system that delivers a single dose of a drug, usually with a ≤1mL fill volume. Most of them act like spring-loaded syringes, and are intended for administration by either the patient or an untrained caregiver. Auto-injectors were developed to overcome hesitation patients had associated with self-administration of a pre-filled syringe, but even these presented problems. With a spring-loaded injector, the patient doesn’t have control over the injection rate, which can occasionally cause increased injection site pain during dosing. This issue, among others, is driving the need for closer partnerships between pharmaceutical and medical device companies as well.
“Cost reductions and contamination control are also major goals concerning single-use devices and systems,” explained Thomas Taylor, president and CEO of Foxx Life Sciences, a Salem, N.H.-based firm that offers custom single-use bottle, flask, and carboy bioprocess assemblies and filtration and fluid management products for the research, biotech, and pharmaceutical industries. “A new, user-loaded injector that can safely deliver high volumes of viscous drugs is critical in the next generation of delivery devices. These products will need a major collaboration between pharma and medical device companies. In the bioprocess market, we already are working together with major pharmaceutical companies to manufacture single-use systems.”
When a pharmaceutical is added to the equation, the approach to product design is vastly different as well. The FDA advocates the adoption of “quality by design” (QbD) practices in pharmaceutical development, and the approach is crucial to drug delivery device development as well. QbD in this context involves in-depth understanding of both the design space and manufacturing processes to make the product.
In terms of the design space, this means laying out key functional requirements, defining moving parts, understanding forces or pressures in the system and interactions between parts, developing sound mechanisms, and analyzing the design. For the manufacturing process, it means identifying critical processes and material attributes, carrying out experiments, and constructing a control strategy. It’s not really a revolutionary philosophy, but since regulatory bodies are in such high support of QbD, drug delivery device engineers must become familiar with the approach.
“In a drug delivery product, the efficacy of the product (Primary Mode of Action) is the drug, not the device, so the device performance and assessment of performance is indirect,” explained Welch. “In a medical device, the efficacy of the product is direct. You push the mechanism hard, it pushes that hard on the tissue. If that’s not hard enough, you change the design of the device. In drug delivery, this process is indirect. Inhalers are a great example of this. You need airflow behind the drug to create a certain plume geometry, but the plume geometry is also dependent on the formulation of the drug. In a drug delivery device, you must understand all the characteristics of what makes the drug effective in the body, and how the device will interact with the body, to design and test the device. In a medical device, you must only worry about the device and its interaction with the body.”
Collaboration is an integral part of doing business, but the languages spoken by pharmaceutical and medical device companies are different, particularly concerning development timelines and regulatory approval pathways. These differences may bring about communication failures between pharmaceutical companies and their medical device partners. Getting ready for a partnership early on helps to mediate these concerns.
Early communication lets both parties make more informed development choices. If all the decisions regarding the product have been made by the pharmaceutical company, with little room for adjustments on the medtech side, speed and reliability of delivering new products can be negatively impacted. Quality control, regulatory guidelines, and reimbursement work very differently across the pharmaceutical and medtech industries. Pharmaceutical companies must decide whether to run production in-house or to partner with a medical device manufacturer. Where and how to perform quality control must also be fully understood prior to production.
“Drug/device combination products must comply with both drug and device requirements as well as combination product requirements,” said White. Testing needs to be done to ensure the device and drug are safe and effective for the patient. We rely on the drug company for drug-specific testing. We collaborate with the pharmaceutical company on plans to ensure all required tests are identified and completed.”
Differences in Current Good Manufacturing Practices (CGMP) requirements between the pharmaceutical and medical device industries can also present a challenge when the two industries partner to develop a drug delivery device or combination product. Since both are a part of the healthcare industry, it would make sense that both sets of companies would have a more thorough understanding of each other’s CGMP practices. But despite the seemingly obvious overlap between the sectors, drug and device firms seem to have issues understanding any CGMP mandates but their own.
For example, prior to a partnership, would a pharmaceutical expert be able to evaluate a medical device company’s CGMP compliance using Part 820 as a checklist, or vice versa? Is one set of requirements “better” to use than the other—that is, more stringent or technical? Only close collaboration and constant communication between the two can result in effective drug delivery dive or combination product development and manufacturing. (For more information about this challenge, read James Dunning of QPC Services LLC’s series of articles entitled “Current Good Manufacturing Practices: Pharma vs. Device,” which begins here: http://bit.ly/mpo181040.)
Misconceptions about vaccines aside, certain scientific and timing factors can make developing an effective influenza vaccine challenging. Current U.S. Food and Drug Administration (FDA) regulatory requirements allow influenza vaccines to be grown in chicken eggs or cells. According to the CDC, today, the majority of influenza vaccines are still grown in chicken eggs. Some influenza viruses—like H3N2—grow poorly in eggs, making it difficult to obtain candidate vaccine viruses. Further, in some years, certain flu viruses may not appear and spread until later in the season, making it difficult to prepare a candidate vaccine virus in time for vaccine production.
Research undertaken at Georgia State University last year is attempting to boost flu shots’ effectiveness. Georgia State researchers developed a biodegradable microneedle patch and a protein constructed from sequences of influenza virus subtypes, which could improve conventional influenza vaccines’ effectiveness. Because circulating flu viruses can rapidly undergo antigenic changes (small genetic alterations that happen over time as the virus replicates), a fast, cost-efficient vaccination method is necessary for broader cross-protection against seasonal and pandemic virus infection.
To achieve this goal, the patch is designed to deliver vaccines into the skin’s dermis and epidermis, and may improve the vaccine’s ability to provoke immune response. The skin is a potent site for vaccination due to an abundance of blood and lymphatic vessels, and many different types of immune cells are present. The patch was given as a “booster” vaccine to mice four weeks after an inactive flu vaccine, then introduced to H1N1 and H3N2 influenza viruses. Mice receiving the microneedle patch displayed significantly enhanced survival and cellular immune responses, as well as long-term antibody-mediated immunity.
Further, microneedle vaccines are much friendlier to patients. Microneedles penetrate the upper layer of the skin, just enough to avoid touching the nerves. A successful microneedle product could have major implications for healthcare delivery—the chronically ill would not have to take daily injections. They also offer a hygiene benefit. Since the patch doesn’t enter the bloodstream, there is less risk of spreading infections.
Implantable devices also offer a number of advantages for improved drug delivery. An implantable pump or drug-eluting implant allows site-specific administration wherever the pharmaceutical is most needed. It can also allow significantly lower doses of the medicine, which can minimize potential side effects. Brachytherapy, for example, involves radioactive implants placed into or near a brain tumor during surgery. Since the radiation only travels a short distance within the body, damage to nearby tissue—a concern common to radiation therapy—is reduced.
The brain is particularly challenging for drug delivery because of the blood-brain barrier (BBB), a highly selective semipermeable border separating circulating blood from the brain and extracellular fluid in the central nervous system. Though it allows water, some gases, glucose, and amino acids to pass through, large molecules (i.e., pharmaceuticals) are restricted access.
In addition, the pharmaceuticals themselves can make the method of delivery challenging. Chemotherapeutic agents can be particularly damaging, and oncologists are working hard to find ways to reduce the chemo dose while effectively treating a tumor. Targeted drug delivery via implantable pharmaceutical vehicles are one way to tackle this issue.
“We have started seeing requirements for drug delivery systems into more challenging areas,” said Ed White, Manager of OEM & International Sales Operations for B. Braun Medical Inc., a Bethlehem, Pa.-based firm that specializes in infusion therapy and pain management and develops, manufactures, and markets medical products and services for the healthcare industry. “We are developing long-term implantable devices to serve this need as a drug delivery method.”
Medical devices have been smartening up for years, and drug delivery technology has certainly followed suit. An intelligent drug delivery system may include functions like alerts to remind users to dose, wireless connectivity to an app to provide historical dosing data and communicate with clinicians, or analytical software to optimize dosage based on external factors. The drive to beef up drug delivery technology with these functions has fostered collaboration between companies who develop the delivery platform and those that specialize in digital services.
For example, in late October, Ypsomed—a developer and manufacturer of injection and infusion systems for self-medication—began a partnership with global health technology firm Philips. The two companies will jointly develop digital self-medication adherence monitoring services based on Ypsomed’s connected devices and Philips’ cloud-based HealthSuite digital platform. Ypsomed’s SmartServices will provide a new turnkey digital solution to monitor medication adherence and enable smart device management for its pharmaceutical customers.
“Our partnership with Philips aims at embedding smart devices in a digital health ecosystem that simplifies access to therapy-relevant medication adherence data,” commented Ulrike Bauer, senior vice president of Ypsomed. “This will allow our pharmaceutical customers to focus on building therapy-specific digital solutions for patients, caregivers, and physicians.”
Philips’ HealthSuite digital platform is purpose-built to streamline complex healthcare challenges, tools and services for ease-of-use integration, digital lifecycle management, and end-to-end security. HealthSuite’s tools help support medication delivery with a framework to build contextual predictive analytics, decision support algorithms, as well as task automation and communications coordination. It also provides management, update, monitoring, and collection of data from smart devices. The new digital services arising from the partnership aim to address device-oriented challenges and support self-medication services for patients with chronic illnesses like diabetes, multiple sclerosis, and rheumatoid arthritis.
“The use of electronics in devices and apps as part of the patient interface have the potential to improve the patient experience, reduce use errors, and ensure adherence,” noted Bill Welch, chief technology officer of Phillips-Medisize, a Molex company. Phillips-Medisize is a Hudson, Wis.-based global outsource provider of design and manufacturing services, with significant business in the drug delivery, medical device, and primary pharmaceutical packaging markets. “These are all aimed at greater patient engagement to improve outcomes, which ultimately go back to drug effectiveness.”
Medtronic’s SynchroMed II pump and catheter, for example, are part of an implantable, programmable drug infusion system. SynchroMed II was initially indicated to administer Infumorph (a morphine sulfate sterile solution) to treat chronic intractable pain and Prialt (a ziconotide sterile solution) to manage severe chronic pain. It worked by injecting these pharmaceuticals into the intrathecal (or subarachnoid) space so it reaches the cerebrospinal fluid. In addition to the pump and catheter, the system comes equipped with a tablet with touchscreen interface to be used as a clinician programmer. The programmer offers workflow guides and steps for refill and implantation, auto-calculations for volume and rates, and visuals to accompany prime bolus and flex dosing. It also connects wirelessly to SynchroMed II’s communicator.
In late January, SynchroMed II was also approved by the FDA to treat pulmonary arterial hypertension by delivering Remodulin into the superior caval-atrial junction (the joint between the superior vena cava and the heart). In this case, the pump is implanted under the skin of the abdomen. It can be refilled via injection through the skin whenever the reservoir runs low.
In late October, the FDA also approved the myPTM Personal Therapy Manager for SynchroMed II for chronic pain patients. The application runs on a touchscreen Samsung J3 smart device that can be customized to assist patients in managing their pain. Clinicians can set daily therapeutic doses, enabling on-demand bolusing within pre-established limits. myPTM makes bolus delivery clear, offers access to therapy details, and provides lockout alerts if patient demand exceeds prescribed limits. Physicians can also use myPTM to view reports providing the necessary insights to track progress and collaborate on therapy goals with their patients.
Insulin pumps are also receiving high-tech upgrades to further assist patients in successfully managing their diabetes. In June, the FDA gave a nod to Tandem Diabetes Care’s t:slim X2 Insulin Pump with Basal-IQ technology, an automated delivery feature with predictive low glucose suspend (PLGS) technology to help reduce the frequency and duration of hypoglycemic events. To determine optimal insulin dosage, Basal-IQ looks 30 minutes into the future to predict where glucose levels are trending. When low glucose is detected (either predicted to drop below 80 mg/dL or currently below 70 mg/dL), it suspends insulin delivery. It automatically resumes delivery once glucose levels begin to rise.
Further simplifying delivery for diabetics, t:slim X2 with the Basal-IQ feature comes integrated with the Dexcom G6 continuous glucose monitoring (CGM) system. The Dexcom G6 CGM notably requires no fingersticks for mealtime dosing or calibration, and can share real-time CGM data with up to five followers. Patients already using the t:slim X2 pump prior to the launch of Basal-IQ can also receive remote software updates to add the predictive algorithm to their pumps using a personal computer.
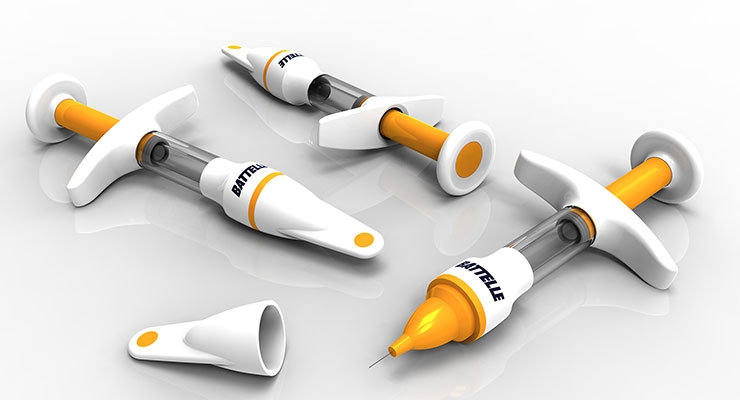
“Digital health is everywhere,” said Alexa Konstantinos, vice president of strategic marketing, medical devices & health analytics for Battelle, a Columbus, Ohio-based firm that conducts research and development, manages laboratories, designs and manufactures products, and delivers critical services to the national security, health and life sciences, and energy and environmental industries. “Devices are getting connected and all the major players are looking to artificial intelligence (AI) as the next frontier to make sense of the data. Right now, connectedness primarily involves tracking ‘historical’ compliance/dosing data. But in the not-too-distant future, much more sophisticated AI algorithms will also combine wellness data, environmental and nutritional data, sleep patterns, phone usage, and other cues to predict dosing needs and health status.”
Though not yet incorporated directly into a drug delivery device, the Sugar.IQ smart diabetes assistant leverages AI and analytical technologies to assist diabetic patients who undergo multiple daily insulin injections, and may form a framework for the type of software that could become standard in drug delivery technology.
Jointly developed by Medtronic and IBM Watson Health and available for Medtronic Guardian Connect CGM users, the Sugar.IQ diabetes assistant continually analyzes how a patient’s glucose level responds to food intake, insulin dosages, daily routines, and other information keyed in by the user. That way, people with diabetes can uncover patterns affecting glucose levels to help make adjustments to their regimen. The app’s advanced analytics and AI offers a full picture of current glucose levels, providing insights and individualized guidance for those on multiple daily insulin injections. According to data presented at this year’s American Diabetes Association Scientific Sessions, those using Sugar.IQ spent 36 minutes more per day in a healthy glucose range than before using the app, including a half hour less time in hyperglycemia, and six minutes less time in hypoglycemia—translating to over nine additional days a year spent in a healthy glucose range.
As it stands, Sugar.IQ is used only indirectly in conjunction with insulin delivery to determine how the medication and lifestyle factors affect glucose levels. However, as the medical device landscape continues to crave holistic solutions to chronic disease management, it is that not far-fetched to say analytical software like Sugar.IQ could be used either in tandem with or be integrated into a therapeutic device for other conditions. Chronic pain management, for instance, also relies on tweaking medication, lifestyle choices, and environmental factors. In the future, an accompanying app could come included with a pain management device, or perhaps even be part of its functionality.
When Industries Collide
Because pharmaceuticals are involved, manufacturing a drug delivery device or combination product cannot be approached with the same protocols as a device that consists purely of technology. For successful drug delivery or combination product commercialization, the device company and pharmaceutical firm must work closely throughout the development cycle. The need for improved drug delivery systems has fostered stronger collaboration between the two industries on an unprecedented scale.
According to research done by PMMI, The Association for Packaging and Processing Technologies, collaboration between medical device and pharmaceutical companies is one of the top five trends influencing growth in the medtech and pharmaceutical industries (notably, just behind the increased use of outside contracting services). Rapid expansion in the biologic drug market is a partial driver of this, with protein-based therapeutics like monoclonal antibodies for cancer treatment necessitating novel drug administration methods. These drugs are usually delivered via infusion or injection, which is less desirable from the patient’s point of view. As a result, pharmaceutical and medical device companies must collaborate to develop more convenient and less painful technological solutions.
“Pharma companies now work with medtech companies much earlier in the development phase,” stated Meredith Canty, director of drug delivery systems for SMC Ltd., a Somerset, Wis.-based global contract manufacturer of pharmaceutical and finished medical devices. “Years ago, the formulators would develop a drug product, then the drug delivery device teams would need to figure out a way to deliver it. Now the formulation and device teams work together much sooner in the program to deliver a more user-friendly experience. This requires multiple rounds of human factors testing with design iterations to enhance the user experience as much as possible.”
“As an integral part of therapy, devices are being considered much earlier in the drug delivery process, as opposed to years past when devices were inserted into the development process toward the end, or were as simplistic as a syringe and vial,” described Konstantinos. “There also has been a drive for several years to ensure devices are usable. Mere usability in the regulatory sense is no longer enough; to gain an advantage over competition, devices must now be preferred by end-users over alternatives, which can help drive better compliance to dosing regimens. This resonates throughout the development process from the very earliest conceptual stages.”
As the adoption and acceptance of self-administered therapies rises, patients are becoming more involved in the delivery and management of their own treatments. The demand for extended release and high concentration drug formulations are breeding more delivery system choices.
For example, patients may opt to use an auto-injector, a drug delivery system that delivers a single dose of a drug, usually with a ≤1mL fill volume. Most of them act like spring-loaded syringes, and are intended for administration by either the patient or an untrained caregiver. Auto-injectors were developed to overcome hesitation patients had associated with self-administration of a pre-filled syringe, but even these presented problems. With a spring-loaded injector, the patient doesn’t have control over the injection rate, which can occasionally cause increased injection site pain during dosing. This issue, among others, is driving the need for closer partnerships between pharmaceutical and medical device companies as well.
“Cost reductions and contamination control are also major goals concerning single-use devices and systems,” explained Thomas Taylor, president and CEO of Foxx Life Sciences, a Salem, N.H.-based firm that offers custom single-use bottle, flask, and carboy bioprocess assemblies and filtration and fluid management products for the research, biotech, and pharmaceutical industries. “A new, user-loaded injector that can safely deliver high volumes of viscous drugs is critical in the next generation of delivery devices. These products will need a major collaboration between pharma and medical device companies. In the bioprocess market, we already are working together with major pharmaceutical companies to manufacture single-use systems.”
When a pharmaceutical is added to the equation, the approach to product design is vastly different as well. The FDA advocates the adoption of “quality by design” (QbD) practices in pharmaceutical development, and the approach is crucial to drug delivery device development as well. QbD in this context involves in-depth understanding of both the design space and manufacturing processes to make the product.
In terms of the design space, this means laying out key functional requirements, defining moving parts, understanding forces or pressures in the system and interactions between parts, developing sound mechanisms, and analyzing the design. For the manufacturing process, it means identifying critical processes and material attributes, carrying out experiments, and constructing a control strategy. It’s not really a revolutionary philosophy, but since regulatory bodies are in such high support of QbD, drug delivery device engineers must become familiar with the approach.
“In a drug delivery product, the efficacy of the product (Primary Mode of Action) is the drug, not the device, so the device performance and assessment of performance is indirect,” explained Welch. “In a medical device, the efficacy of the product is direct. You push the mechanism hard, it pushes that hard on the tissue. If that’s not hard enough, you change the design of the device. In drug delivery, this process is indirect. Inhalers are a great example of this. You need airflow behind the drug to create a certain plume geometry, but the plume geometry is also dependent on the formulation of the drug. In a drug delivery device, you must understand all the characteristics of what makes the drug effective in the body, and how the device will interact with the body, to design and test the device. In a medical device, you must only worry about the device and its interaction with the body.”
Collaboration is an integral part of doing business, but the languages spoken by pharmaceutical and medical device companies are different, particularly concerning development timelines and regulatory approval pathways. These differences may bring about communication failures between pharmaceutical companies and their medical device partners. Getting ready for a partnership early on helps to mediate these concerns.
Early communication lets both parties make more informed development choices. If all the decisions regarding the product have been made by the pharmaceutical company, with little room for adjustments on the medtech side, speed and reliability of delivering new products can be negatively impacted. Quality control, regulatory guidelines, and reimbursement work very differently across the pharmaceutical and medtech industries. Pharmaceutical companies must decide whether to run production in-house or to partner with a medical device manufacturer. Where and how to perform quality control must also be fully understood prior to production.
“Drug/device combination products must comply with both drug and device requirements as well as combination product requirements,” said White. Testing needs to be done to ensure the device and drug are safe and effective for the patient. We rely on the drug company for drug-specific testing. We collaborate with the pharmaceutical company on plans to ensure all required tests are identified and completed.”
Differences in Current Good Manufacturing Practices (CGMP) requirements between the pharmaceutical and medical device industries can also present a challenge when the two industries partner to develop a drug delivery device or combination product. Since both are a part of the healthcare industry, it would make sense that both sets of companies would have a more thorough understanding of each other’s CGMP practices. But despite the seemingly obvious overlap between the sectors, drug and device firms seem to have issues understanding any CGMP mandates but their own.
For example, prior to a partnership, would a pharmaceutical expert be able to evaluate a medical device company’s CGMP compliance using Part 820 as a checklist, or vice versa? Is one set of requirements “better” to use than the other—that is, more stringent or technical? Only close collaboration and constant communication between the two can result in effective drug delivery dive or combination product development and manufacturing. (For more information about this challenge, read James Dunning of QPC Services LLC’s series of articles entitled “Current Good Manufacturing Practices: Pharma vs. Device,” which begins here: http://bit.ly/mpo181040.)