Business Wire08.29.18
AVITA Medical has expanded its management team in the commercial, legal, and medical affairs functions to support the planned U.S. launch of the RECELL Autologous Cell Harvesting Device in burn treatments. Joining AVITA Medical are Donna Shiroma, general counsel; Terry Bromley, vice president, Commercial Operations; and Debbie Garner, vice president, Global Marketing. In addition, Katie Bush, Ph.D., has been promoted to vice president, Medical Affairs.
As general counsel, Shiroma is responsible for all legal operations of AVITA. With over 20 years of legal and compliance experience in the pharmaceutical and medical device industries, she has played an instrumental role in transitioning companies from clinical to commercial entities. Prior to joining AVITA Medical, Shiroma served in roles of increasing responsibility as corporate counsel, general counsel, vice president of legal, chief privacy and compliance officer, and chief commercial officer for Ascend Therapeutics, PDL BioPharma, and several Johnson & Johnson companies.
Bromley has broad commercial responsibility for the planned U.S. launch of the RECELL device including management of the commercial field team, sales training, sales operations, and customer service. Bromley led sales and marketing operations for a number of medical products, including wound healing and skin care, with Crawford Healthcare, Emergent BioSolutions, Convatech and Bristol-Myers Squibb.
Garner is responsible for leading the long-term commercial plan for AVITA Medical, focusing initially on the RECELL Device. Garner joins AVITA Medical from Allergan, where she served as executive marketing director for the Medical Dermatology Franchise. While at Allergan she also led global pipeline and business development for aesthetic and therapeutic dermatology products including BOTOX Cosmetic and Juvederm. Previously, Garner held various marketing roles at Takeda Abbott Pharmaceuticals.
As a result of her promotion, Dr. Bush has responsibility for the development and implementation of educational programs, medical-scientific communications, and supporting company-sponsored scientific research and clinical studies. Dr. Bush will also oversee AVITA’s Medical Science Liaison and Medical Education teams. Through her career Dr. Bush has contributed to the development and commercialization of products for soft tissue regenerative applications, including multiple product lines focused on skin regeneration. She has a doctorate in biomedical engineering and medical physics from The University of Massachusetts & Worcester Polytechnic Institute (Worcester, Mass.), completing a thesis on the development of a novel collagen-based skin substitute to enhance re-epithelialization.
“These leaders and their respective teams join AVITA Medical at an exciting time as we take the steps to prepare for a successful launch of the RECELL Device in the U.S.,” said Dr. Michael Perry, CEO. “We are pleased to have such a strong group of professionals join us and look forward to benefiting from their experience in the commercialization of products in the regenerative medicine and skin care space.”
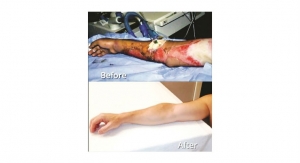
The RECELL Device is an investigational medical device in the United States that is designed to enable medical professionals to produce, at the point-of-care, a REGENERATIVE EPIDERMAL SUSPENSION (RES) using a small sample of the patient’s own skin. The autologous suspension contains cells necessary to regenerate epidermis and provides a new way to achieve permanent closure in burns and other wounds while reducing the amount of skin harvested at the time of surgery. Reduction in donor-site skin requirements has important benefits from both clinical and health economic perspectives.
A U.S. Premarket Approval (PMA) application for the treatment of burn injuries is currently under review by the U.S. Food and Drug Administration (FDA). The company expects completion of the FDA review of the PMA during the third quarter of calendar 2018, followed by U.S. approval and market launch.
AVITA Medical is a regenerative medicine company with a technology platform positioned to address unmet medical needs in burns, chronic wounds, and aesthetics indications. AVITA Medical’s patented and proprietary collection and application technology provides innovative treatment solutions derived from the regenerative properties of a patient’s own skin. The medical devices work by preparing a regenerative epithelial suspension, an autologous suspension comprised of the patient’s skin cells and wound healing factors necessary to regenerate natural healthy epidermis. This autologous suspension is then sprayed onto the areas of the patient requiring treatment.
In the United States, the RECELL Device is an investigational device limited by federal law to investigational use. In September 2017, AVITA Medical submitted a PMA application to the FDA for the RECELL Device for the treatment of burn injuries.
In all countries outside of Europe, our portfolio is marketed under the RECELL Device brand to promote skin healing in a wide range of applications including burns, chronic wounds and aesthetics. The RECELL Device is TGA-registered in Australia, and CFDA-cleared in China.
In Europe, AVITA Medical's portfolio of products received CE-mark approval as three tailored product presentations, with three individual brand names. The RECELL Autologous Cell Harvesting Device is designed for the treatment of burns and plastic reconstructive procedures; REGENERCELL Autologous Cell Harvesting Device has been formulated for chronic wounds including leg and foot ulcers; and RENOVACELL Autologous Cell Harvesting Device is tailored for aesthetic applications including the restoration of pigmentation.
As general counsel, Shiroma is responsible for all legal operations of AVITA. With over 20 years of legal and compliance experience in the pharmaceutical and medical device industries, she has played an instrumental role in transitioning companies from clinical to commercial entities. Prior to joining AVITA Medical, Shiroma served in roles of increasing responsibility as corporate counsel, general counsel, vice president of legal, chief privacy and compliance officer, and chief commercial officer for Ascend Therapeutics, PDL BioPharma, and several Johnson & Johnson companies.
Bromley has broad commercial responsibility for the planned U.S. launch of the RECELL device including management of the commercial field team, sales training, sales operations, and customer service. Bromley led sales and marketing operations for a number of medical products, including wound healing and skin care, with Crawford Healthcare, Emergent BioSolutions, Convatech and Bristol-Myers Squibb.
Garner is responsible for leading the long-term commercial plan for AVITA Medical, focusing initially on the RECELL Device. Garner joins AVITA Medical from Allergan, where she served as executive marketing director for the Medical Dermatology Franchise. While at Allergan she also led global pipeline and business development for aesthetic and therapeutic dermatology products including BOTOX Cosmetic and Juvederm. Previously, Garner held various marketing roles at Takeda Abbott Pharmaceuticals.
As a result of her promotion, Dr. Bush has responsibility for the development and implementation of educational programs, medical-scientific communications, and supporting company-sponsored scientific research and clinical studies. Dr. Bush will also oversee AVITA’s Medical Science Liaison and Medical Education teams. Through her career Dr. Bush has contributed to the development and commercialization of products for soft tissue regenerative applications, including multiple product lines focused on skin regeneration. She has a doctorate in biomedical engineering and medical physics from The University of Massachusetts & Worcester Polytechnic Institute (Worcester, Mass.), completing a thesis on the development of a novel collagen-based skin substitute to enhance re-epithelialization.
“These leaders and their respective teams join AVITA Medical at an exciting time as we take the steps to prepare for a successful launch of the RECELL Device in the U.S.,” said Dr. Michael Perry, CEO. “We are pleased to have such a strong group of professionals join us and look forward to benefiting from their experience in the commercialization of products in the regenerative medicine and skin care space.”
The RECELL Device is an investigational medical device in the United States that is designed to enable medical professionals to produce, at the point-of-care, a REGENERATIVE EPIDERMAL SUSPENSION (RES) using a small sample of the patient’s own skin. The autologous suspension contains cells necessary to regenerate epidermis and provides a new way to achieve permanent closure in burns and other wounds while reducing the amount of skin harvested at the time of surgery. Reduction in donor-site skin requirements has important benefits from both clinical and health economic perspectives.
A U.S. Premarket Approval (PMA) application for the treatment of burn injuries is currently under review by the U.S. Food and Drug Administration (FDA). The company expects completion of the FDA review of the PMA during the third quarter of calendar 2018, followed by U.S. approval and market launch.
AVITA Medical is a regenerative medicine company with a technology platform positioned to address unmet medical needs in burns, chronic wounds, and aesthetics indications. AVITA Medical’s patented and proprietary collection and application technology provides innovative treatment solutions derived from the regenerative properties of a patient’s own skin. The medical devices work by preparing a regenerative epithelial suspension, an autologous suspension comprised of the patient’s skin cells and wound healing factors necessary to regenerate natural healthy epidermis. This autologous suspension is then sprayed onto the areas of the patient requiring treatment.
In the United States, the RECELL Device is an investigational device limited by federal law to investigational use. In September 2017, AVITA Medical submitted a PMA application to the FDA for the RECELL Device for the treatment of burn injuries.
In all countries outside of Europe, our portfolio is marketed under the RECELL Device brand to promote skin healing in a wide range of applications including burns, chronic wounds and aesthetics. The RECELL Device is TGA-registered in Australia, and CFDA-cleared in China.
In Europe, AVITA Medical's portfolio of products received CE-mark approval as three tailored product presentations, with three individual brand names. The RECELL Autologous Cell Harvesting Device is designed for the treatment of burns and plastic reconstructive procedures; REGENERCELL Autologous Cell Harvesting Device has been formulated for chronic wounds including leg and foot ulcers; and RENOVACELL Autologous Cell Harvesting Device is tailored for aesthetic applications including the restoration of pigmentation.