Business Wire05.24.18
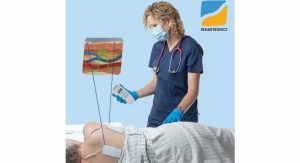
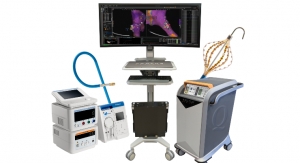
XableCath Inc., a developer of products designed to aid in the treatment of peripheral artery disease (PAD), announced that its second catheter, XableCath abrasion tip support catheter, was cleared by the FDA. The XableCath blunt tip catheter received FDA clearance at the end of 2017. This clearance gives peripheral interventionalists another innovative choice in managing all types of lesions and procedures in the peripheral arterials.
“This FDA clearance allows physicians to select between both of XableCath’s unique designs to easily slide past an occluding atheroma through the true lumen of the vessel. It further validates our efforts to deliver safe and effective products for peripheral arterial interventions,” said XableCath president and CEO, Lisa Dunlea.
The company’s Medical Director, Steve Lauterbach, M.D., commented, “We are delighted with this new FDA clearance. Interventionalists can now choose the XableCath that is best suited for their patients’ anatomy. Both our blunt and abrasion tips have had remarkable initial clinical success in very challenging cases. We look forward to increasing our clinical experience throughout the year.”
Oded Ben-Joseph, Ph.D., Board Member at XableCath commented, “With the addition of this second FDA clearance, the company is rapidly preparing to commercialize its product portfolio. Combining its clinical case success rate, patent portfolio, and now FDA cleared product lines, XableCath will have an impact on the peripheral arterial disease marketplace. We are confident that XableCath’s innovative products will seamlessly integrate into the cath lab workflow and provide the interventionalist with a much-needed tool to enable treatment of challenging occlusions.”
XableCath performed its first cases in the United States during the first quarter of this year. The Company is anticipating a full commercial launch in the second half of 2018.
“This FDA clearance allows physicians to select between both of XableCath’s unique designs to easily slide past an occluding atheroma through the true lumen of the vessel. It further validates our efforts to deliver safe and effective products for peripheral arterial interventions,” said XableCath president and CEO, Lisa Dunlea.
The company’s Medical Director, Steve Lauterbach, M.D., commented, “We are delighted with this new FDA clearance. Interventionalists can now choose the XableCath that is best suited for their patients’ anatomy. Both our blunt and abrasion tips have had remarkable initial clinical success in very challenging cases. We look forward to increasing our clinical experience throughout the year.”
Oded Ben-Joseph, Ph.D., Board Member at XableCath commented, “With the addition of this second FDA clearance, the company is rapidly preparing to commercialize its product portfolio. Combining its clinical case success rate, patent portfolio, and now FDA cleared product lines, XableCath will have an impact on the peripheral arterial disease marketplace. We are confident that XableCath’s innovative products will seamlessly integrate into the cath lab workflow and provide the interventionalist with a much-needed tool to enable treatment of challenging occlusions.”
XableCath performed its first cases in the United States during the first quarter of this year. The Company is anticipating a full commercial launch in the second half of 2018.