Business Wire05.22.18
Aziyo Biologics Inc., a fully integrated regenerative medicine company, has entered into a U.S. co-distribution agreement for its CanGaroo Bio Envelope with Biotronik Inc., a global leader in cardio- and endovascular medical technology.
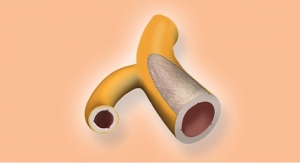
CanGaroo Bio Envelopes are a natural extracellular matrix (ECM) scaffold that securely hold cardiovascular implantable electronic devices (CIEDs) to create a stable environment following implantation. In-vivo studies have demonstrated that use of CanGaroo when implanting CIEDs reduces the risk of device migration and erosion and may facilitate device removal when future exchange or revision is required.
“BIOTRONIK is trusted and respected by hospitals, physicians and patients, and our distribution partnership expands access to a meaningful cardiovascular solution,” said Andrew Green, Aziyo’s executive vice president and general manager of Cardiovascular Products. “Our ECM envelopes help deliver the best possible patient care and can reduce cost, providing advantages that complement BIOTRONIK’s comprehensive portfolio of cardiac rhythm management devices. This partnership changes the landscape of cardiac care.”
CanGaroo is available in five sizes for use with the full spectrum of transvenous and subcutaneous CIEDs. Per the strategic distribution agreement, Biotronik is selling all five sizes in the United States branded as Bio Envelope.
Aziyo Biologics is a fully integrated, commercially oriented regenerative medicine company. Since its founding in 2015, the company has expanded through acquisitions and strategic partnerships, creating a high-growth commercial entity. Its proprietary products are used in orthopedic, cardiovascular and other medical specialties.
The CanGaroo Bio Envelope is constructed from multi-laminate sheets of decellularized, non-crosslinked, lyophilized extracellular matrix (ECM) derived from porcine small intestinal submucosa (SIS). Once implanted, CanGaroo secures the CIED in place, and evidence shows that over time it remodels into a vascularized pocket that may facilitate CIED removal or revision. CanGaroo Bio Envelope is a 510(k) U.S. Food and Drug Administration-cleared medical device.
Porcine SIS-ECM is a decellularized matrix that serves as a bioscaffold to allow vascular ingrowth from adjacent tissues to deliver progenitor cells and nutrients to the matrix, which then differentiate into tissue-specific cells and structures. The ECM is gradually replaced as the patient’s own cells restore the diseased or damaged site. During repair, the matrix is naturally degraded and resorbed, leaving remodeled functional tissue where damaged or injured tissue would normally be expected. Since 1999, an estimated 2 million patients worldwide have received SIS-ECM implants.
CanGaroo Bio Envelopes are a natural extracellular matrix (ECM) scaffold that securely hold cardiovascular implantable electronic devices (CIEDs) to create a stable environment following implantation. In-vivo studies have demonstrated that use of CanGaroo when implanting CIEDs reduces the risk of device migration and erosion and may facilitate device removal when future exchange or revision is required.
“BIOTRONIK is trusted and respected by hospitals, physicians and patients, and our distribution partnership expands access to a meaningful cardiovascular solution,” said Andrew Green, Aziyo’s executive vice president and general manager of Cardiovascular Products. “Our ECM envelopes help deliver the best possible patient care and can reduce cost, providing advantages that complement BIOTRONIK’s comprehensive portfolio of cardiac rhythm management devices. This partnership changes the landscape of cardiac care.”
CanGaroo is available in five sizes for use with the full spectrum of transvenous and subcutaneous CIEDs. Per the strategic distribution agreement, Biotronik is selling all five sizes in the United States branded as Bio Envelope.
Aziyo Biologics is a fully integrated, commercially oriented regenerative medicine company. Since its founding in 2015, the company has expanded through acquisitions and strategic partnerships, creating a high-growth commercial entity. Its proprietary products are used in orthopedic, cardiovascular and other medical specialties.
The CanGaroo Bio Envelope is constructed from multi-laminate sheets of decellularized, non-crosslinked, lyophilized extracellular matrix (ECM) derived from porcine small intestinal submucosa (SIS). Once implanted, CanGaroo secures the CIED in place, and evidence shows that over time it remodels into a vascularized pocket that may facilitate CIED removal or revision. CanGaroo Bio Envelope is a 510(k) U.S. Food and Drug Administration-cleared medical device.
Porcine SIS-ECM is a decellularized matrix that serves as a bioscaffold to allow vascular ingrowth from adjacent tissues to deliver progenitor cells and nutrients to the matrix, which then differentiate into tissue-specific cells and structures. The ECM is gradually replaced as the patient’s own cells restore the diseased or damaged site. During repair, the matrix is naturally degraded and resorbed, leaving remodeled functional tissue where damaged or injured tissue would normally be expected. Since 1999, an estimated 2 million patients worldwide have received SIS-ECM implants.