Business Wire01.04.18
Avita Medical, a regenerative medicine company with a technology platform positioned to address opportunities and unmet medical needs in burns, chronic wounds, and aesthetics indications, has appointed Dale A. Sander as chief financial officer.
Sander will be responsible for overseeing the global finance and investor relations functions, and aligning them with the company’s commercialization strategy. Current CFO Tim Rooney has been appointed Avita’s chief administrative officer (CAO), responsible for global operations and supply chain management, human resources, and information technology. Both executives will be based in Valencia, Calif.
“These executive changes come at a pivotal time in the transformation of our company,” said Dr. Mike Perry, CEO of Avita Medical. “Dale has more than 20 years of experience, including serving as CFO of four medical device and pharmaceutical companies as well as 11 years of leading public companies in the CFO role. Dale brings extensive experience in leading both public and private equity offerings, including multiple initial public offerings in the U.S. and Europe. He also has a strong track record of managing investor relations in these markets as well as in Asia. Dale’s expertise will be critical in supporting our commercialization strategy in advance of our anticipated approval of ReCell in the United States.”
Rooney, who has been with Avita Medical since 2012 in various key executive roles, will now lead various critical operational and strategic functions. “I’m thrilled to continue my work with Avita as part of our senior executive team,” Rooney said. “In my new role, I look forward to continuing my contributions to critical elements in Avita’s growth, especially as we prepare for the anticipated approval of ReCell.”
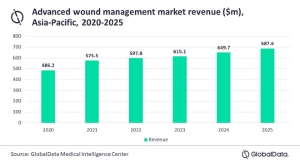
A premarket approval (PMA) application for the ReCell device was filed on Sept. 28, 2017, and is currently under review by the U.S. Food and Drug Administration (FDA). ReCell is a regenerative medicine platform with the potential to offer patients benefits across a broad range of applications, including burns, chronic wounds, and aesthetics.
Avita’s patented and proprietary collection and application technology provides treatment solutions derived from the regenerative properties of a patient’s own skin. The firm's medical devices work by preparing a regenerative epithelial suspension (RES), an autologous suspension comprised of the patients’ own skin cells and wound healing factors that are necessary to regenerate natural healthy skin. This is then applied to the area to be treated.
In all countries outside of Europe, Avita's portfolio is marketed under the ReCell brand to promote skin healing in a wide range of applications, including burns, chronic wounds, and aesthetics.
ReCell is TGA-registered in Australia, and cleared by the China Food and Drug Administration. In the United States, ReCell is an investigational device limited by federal law to investigational use. In Europe, the company's portfolio of medical device products received CE-mark approval as three tailored product presentations, with three individual brand names. ReCell is designed for the treatment of burns and plastic reconstructive procedures; ReGenerCell has been formulated for chronic wounds, including leg and foot ulcers; and ReNovaCell is tailored for aesthetic applications, including the restoration of pigmentation.
Sander will be responsible for overseeing the global finance and investor relations functions, and aligning them with the company’s commercialization strategy. Current CFO Tim Rooney has been appointed Avita’s chief administrative officer (CAO), responsible for global operations and supply chain management, human resources, and information technology. Both executives will be based in Valencia, Calif.
“These executive changes come at a pivotal time in the transformation of our company,” said Dr. Mike Perry, CEO of Avita Medical. “Dale has more than 20 years of experience, including serving as CFO of four medical device and pharmaceutical companies as well as 11 years of leading public companies in the CFO role. Dale brings extensive experience in leading both public and private equity offerings, including multiple initial public offerings in the U.S. and Europe. He also has a strong track record of managing investor relations in these markets as well as in Asia. Dale’s expertise will be critical in supporting our commercialization strategy in advance of our anticipated approval of ReCell in the United States.”
Rooney, who has been with Avita Medical since 2012 in various key executive roles, will now lead various critical operational and strategic functions. “I’m thrilled to continue my work with Avita as part of our senior executive team,” Rooney said. “In my new role, I look forward to continuing my contributions to critical elements in Avita’s growth, especially as we prepare for the anticipated approval of ReCell.”
A premarket approval (PMA) application for the ReCell device was filed on Sept. 28, 2017, and is currently under review by the U.S. Food and Drug Administration (FDA). ReCell is a regenerative medicine platform with the potential to offer patients benefits across a broad range of applications, including burns, chronic wounds, and aesthetics.
Avita’s patented and proprietary collection and application technology provides treatment solutions derived from the regenerative properties of a patient’s own skin. The firm's medical devices work by preparing a regenerative epithelial suspension (RES), an autologous suspension comprised of the patients’ own skin cells and wound healing factors that are necessary to regenerate natural healthy skin. This is then applied to the area to be treated.
In all countries outside of Europe, Avita's portfolio is marketed under the ReCell brand to promote skin healing in a wide range of applications, including burns, chronic wounds, and aesthetics.
ReCell is TGA-registered in Australia, and cleared by the China Food and Drug Administration. In the United States, ReCell is an investigational device limited by federal law to investigational use. In Europe, the company's portfolio of medical device products received CE-mark approval as three tailored product presentations, with three individual brand names. ReCell is designed for the treatment of burns and plastic reconstructive procedures; ReGenerCell has been formulated for chronic wounds, including leg and foot ulcers; and ReNovaCell is tailored for aesthetic applications, including the restoration of pigmentation.