Business Wire05.10.18
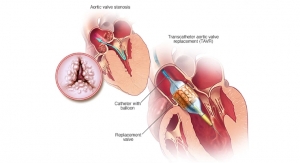
JenaValve Technology Inc., a developer, manufacturer, and marketer of differentiated transcatheter aortic valve replacement (TAVR) systems for the treatment of aortic valve disease, has announced successful patient enrollment in the CE Mark study of the next-generation JenaValve Pericardial TAVR System utilizing the Coronatix Transfemoral Delivery Catheter for the percutaneous treatment of severe aortic stenosis. The JenaValve Pericardial TAVR System is an investigational device in the United States and internationally.
The JenaValve system is proprietary and differentiated from prior technologies, comprising the Everdur Pericardial Aortic Valve with locator-based technology designed for more predictable implantation, combined with the new 18-Fr compatible Coronatix Transfemoral Delivery Catheter. The optimized TAVR system has now been used to treat 10 patients across three sites in Germany and New Zealand. Technical success for those procedures was 100 percent, with no reported adverse events and no moderate or higher paravalvular leakage reported.
The CE Mark study is an international, prospective, non-randomized, single-arm trial of the JenaValve Pericardial TAVR System for the treatment of severe aortic stenosis in patients who are at increased risk for conventional surgical valve replacement.
The German national Principal Investigator, Prof. Dr. med. Hendrik Treede (Halle University, Halle, Germany), said, “The novel JenaValve transcatheter valve may address the ongoing clinical needs for these patients. Specifically, early experience with this innovative transcatheter heart valve demonstrates low permanent pacemaker rates, negligible mean pressure gradients across the valve and no significant paravalvular leaks. Additionally, the new transfemoral system enables rapid, safe, and reliable delivery of this new valve.”
“We were already extremely pleased with the initial clinical results of our next generation TAVR valve, Everdur. Now, the most recent implants using the optimized delivery system have shown significantly improved ease of use and performance,” said JenaValve CEO Victoria Carr-Brendel, Ph.D. “We have invested a substantial amount of intellectual know-how, capital and time to develop the Everdur valve and Coronatix delivery catheter, and they are exceeding our expectations. We look forward to expanding our clinical development programs and building on this initial body of clinical evidence to eventually improve results, quality of life and survival among patients suffering these very serious conditions.”
The company expects to complete patient enrollment by mid-2018 at clinical sites in Germany, The Netherlands, New Zealand, and the United States. The company anticipates CE Mark approval by the end of 2018.
The JenaValve Pericardial TAVR System consists of the Everdur Pericardial Aortic Valve and the Transfemoral Delivery System. The bioprosthesis comprises a self-expanding nitinol stent with a porcine pericardial valve manufactured using state-of-the-art tissue processing techniques. The Coronatix transfemoral delivery catheter is designed to deliver the bioprosthesis using a simple stepped approach with anatomic positioning over the native valve. The System is available in three sizes intended for aortic annulus diameters from 21mm to 27mm. A larger bioprosthesis size is in development.
JenaValve Technology Inc., with locations in Irvine, Calif.; Leeds, England; and Munich, Germany; develops, manufactures, and markets transcatheter aortic valve replacement (TAVR) systems to treat patients suffering from aortic valve disease. The company is in clinical development of its next-generation transfemoral TAVR system, consisting of the Everdur valve and Coronatix transfemoral delivery catheter, in both the United States and CE-Mark countries for treating patients with aortic stenosis and/or aortic regurgitation. The company’s first-generation transapical system, consisting of the JenaValve valve with Cathlete PLUS delivery system, was commercialized under CE Mark approval for aortic valve stenosis and for aortic valve regurgitation. JenaValve is backed by U.S., European and Asian investors, including Andera Partners (formerly Edmond de Rothschild Investment Partners), Gimv (a Euronext-listed investment company), Legend Capital, NeoMed Management, Omega Funds, RMM, Valiance Life Sciences, and VI Partners.
The JenaValve system is proprietary and differentiated from prior technologies, comprising the Everdur Pericardial Aortic Valve with locator-based technology designed for more predictable implantation, combined with the new 18-Fr compatible Coronatix Transfemoral Delivery Catheter. The optimized TAVR system has now been used to treat 10 patients across three sites in Germany and New Zealand. Technical success for those procedures was 100 percent, with no reported adverse events and no moderate or higher paravalvular leakage reported.
The CE Mark study is an international, prospective, non-randomized, single-arm trial of the JenaValve Pericardial TAVR System for the treatment of severe aortic stenosis in patients who are at increased risk for conventional surgical valve replacement.
The German national Principal Investigator, Prof. Dr. med. Hendrik Treede (Halle University, Halle, Germany), said, “The novel JenaValve transcatheter valve may address the ongoing clinical needs for these patients. Specifically, early experience with this innovative transcatheter heart valve demonstrates low permanent pacemaker rates, negligible mean pressure gradients across the valve and no significant paravalvular leaks. Additionally, the new transfemoral system enables rapid, safe, and reliable delivery of this new valve.”
“We were already extremely pleased with the initial clinical results of our next generation TAVR valve, Everdur. Now, the most recent implants using the optimized delivery system have shown significantly improved ease of use and performance,” said JenaValve CEO Victoria Carr-Brendel, Ph.D. “We have invested a substantial amount of intellectual know-how, capital and time to develop the Everdur valve and Coronatix delivery catheter, and they are exceeding our expectations. We look forward to expanding our clinical development programs and building on this initial body of clinical evidence to eventually improve results, quality of life and survival among patients suffering these very serious conditions.”
The company expects to complete patient enrollment by mid-2018 at clinical sites in Germany, The Netherlands, New Zealand, and the United States. The company anticipates CE Mark approval by the end of 2018.
The JenaValve Pericardial TAVR System consists of the Everdur Pericardial Aortic Valve and the Transfemoral Delivery System. The bioprosthesis comprises a self-expanding nitinol stent with a porcine pericardial valve manufactured using state-of-the-art tissue processing techniques. The Coronatix transfemoral delivery catheter is designed to deliver the bioprosthesis using a simple stepped approach with anatomic positioning over the native valve. The System is available in three sizes intended for aortic annulus diameters from 21mm to 27mm. A larger bioprosthesis size is in development.
JenaValve Technology Inc., with locations in Irvine, Calif.; Leeds, England; and Munich, Germany; develops, manufactures, and markets transcatheter aortic valve replacement (TAVR) systems to treat patients suffering from aortic valve disease. The company is in clinical development of its next-generation transfemoral TAVR system, consisting of the Everdur valve and Coronatix transfemoral delivery catheter, in both the United States and CE-Mark countries for treating patients with aortic stenosis and/or aortic regurgitation. The company’s first-generation transapical system, consisting of the JenaValve valve with Cathlete PLUS delivery system, was commercialized under CE Mark approval for aortic valve stenosis and for aortic valve regurgitation. JenaValve is backed by U.S., European and Asian investors, including Andera Partners (formerly Edmond de Rothschild Investment Partners), Gimv (a Euronext-listed investment company), Legend Capital, NeoMed Management, Omega Funds, RMM, Valiance Life Sciences, and VI Partners.