Stryker 03.08.17
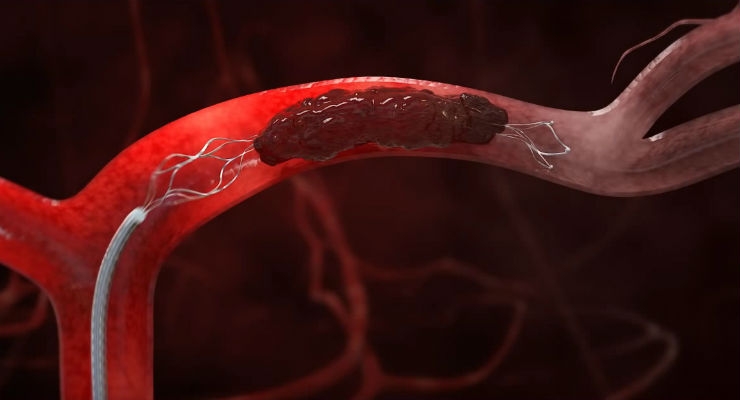
Stryker announced an early end to patient enrollment in the DAWN Trial, a clinical study designed to compare mechanical thrombectomy with the Trevo Retriever plus medical therapy against medical therapy alone when initiated within six to 24 hours after time last known well. The independent Data Safety Monitoring Board (DSMB) recommended stopping study enrollment based on a pre-planned interim review of data from the first 200 patients, which concluded that multiple pre-specified stopping criteria were met. A final analysis of the data will be conducted upon completion of the remaining patient follow-up.
The study was designed to enroll up to a maximum of 500 patients with a pre-specified interim analysis to assess for efficacy initiated upon enrollment of the first 200 patients.
Stroke survivors commonly experience devastating disabilities and loss of independence due to impaired movement, paralysis, loss of speech and memory. Randomized clinical data has proven the benefit of mechanical thrombectomy with stent retrievers in helping patients with large vessel occlusion strokes, but these devices have only been indicated to reduce disability if used within six hours of stroke onset. For patients presenting with stroke symptoms beyond six hours, the benefit of clot retrieval using a stent retriever is unknown.
“Treating acute stroke patients with large vessel occlusion who present later than six hours from last seen well has the potential to help thousands of stroke patients around the world,” said co-principal investigator Tudor Jovin, MD, from the University of Pittsburgh Medical Center. “These patients, many of whom present to the hospital outside of the six hour time window could have a better chance for an independent life with improved clinical outcomes.”
Dr. Raul Nogueira, co-principal investigator from Grady Memorial Hospital/ Emory University commented, “If the final results of the DAWN Trial are positive, it will provide physicians who treat stroke with evidence of the benefits of thrombectomy even when administered out as far as 24 hours, and should help to make decisions clearer as to which patients to treat.”
Stryker and the DAWN investigators are now focused on gathering and securing all of the remaining patient data for final statistical analysis. If confirmed positive, the outcomes of the DAWN Trial may represent a major change in patient selection for endovascular therapy for stroke.
The following video demonstrates the device's function.
The study was designed to enroll up to a maximum of 500 patients with a pre-specified interim analysis to assess for efficacy initiated upon enrollment of the first 200 patients.
Stroke survivors commonly experience devastating disabilities and loss of independence due to impaired movement, paralysis, loss of speech and memory. Randomized clinical data has proven the benefit of mechanical thrombectomy with stent retrievers in helping patients with large vessel occlusion strokes, but these devices have only been indicated to reduce disability if used within six hours of stroke onset. For patients presenting with stroke symptoms beyond six hours, the benefit of clot retrieval using a stent retriever is unknown.
“Treating acute stroke patients with large vessel occlusion who present later than six hours from last seen well has the potential to help thousands of stroke patients around the world,” said co-principal investigator Tudor Jovin, MD, from the University of Pittsburgh Medical Center. “These patients, many of whom present to the hospital outside of the six hour time window could have a better chance for an independent life with improved clinical outcomes.”
Dr. Raul Nogueira, co-principal investigator from Grady Memorial Hospital/ Emory University commented, “If the final results of the DAWN Trial are positive, it will provide physicians who treat stroke with evidence of the benefits of thrombectomy even when administered out as far as 24 hours, and should help to make decisions clearer as to which patients to treat.”
Stryker and the DAWN investigators are now focused on gathering and securing all of the remaining patient data for final statistical analysis. If confirmed positive, the outcomes of the DAWN Trial may represent a major change in patient selection for endovascular therapy for stroke.
The following video demonstrates the device's function.