DNA Genotek Inc.06.14.16
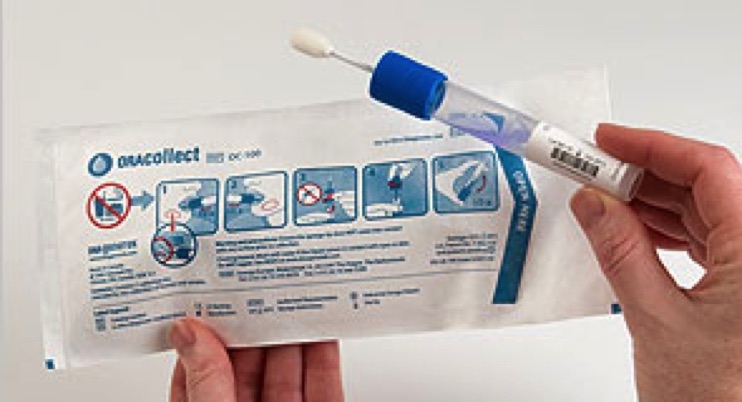
DNA Genotek Inc., a leading provider of sample collection kits and end-to-end services and a wholly-owned subsidiary of OraSure Technologies Inc., announced that the company’s ORAcollect•Dx collection device has been granted 510(k) clearance by the U.S. Food and Drug Administration (FDA). ORAcollect•Dx joins the company’s Oragene•Dx family of products as the only saliva based DNA collection and stabilization devices to receive 510(k) clearance.
ORAcollect•Dx is an attractive alternative to buccal swabs and offers several advantages including a liquid based sample for improved lab efficiency, and the elimination of manual steps like cutting tips which reduces the chance of errors and cross-contamination. In addition, the product has been demonstrated to provide higher DNA yields than buccal swabs, reduces bacterial contamination and is stable for months at ambient temperatures.
ORAcollect•Dx is a complementary offering to Oragene•Dx, which received 510(k) clearance in 2011. DNA Genotek’s FDA clearances provide access to cleared, saliva-based collection devices for diagnostic companies and healthcare facilities of all sizes who want to deliver an improved patient experience as well as facilitate growth of their testing services through easier access to patients.
“Our portfolio of FDA cleared products offers our diagnostic customers in the U.S. a broad range of options for DNA collection for tests requiring both low and high amounts of high quality DNA. ORAcollect•Dx fills a critical need for a cleared device that improves on the stability and yield of buccal swabs while offering a liquid sample,” said Brian Smith, general manager, Genomics and Microbiome, at DNA Genotek Inc. “Molecular diagnostic and laboratory developed test manufacturers can reduce the time and regulatory burden of their own FDA submissions by leveraging the 510(k) clearance of ORAcollect•Dx.”
ORAcollect•Dx is FDA-cleared for in-vitro diagnostic use with the eSensor Warfarin Sensitivity Saliva test.
DNA Genotek Inc., a subsidiary of OraSure Technologies Inc. provides biological sample collection products and end-to-end services for human genomics, microbiome and infectious disease applications. The Canadian company also offers research use only products to collect and preserve large amounts of DNA or RNA from multiple sample types. DNA Genotek markets its products worldwide and has a global customer base with thousands of customers in more than 100 countries.
ORAcollect•Dx is an attractive alternative to buccal swabs and offers several advantages including a liquid based sample for improved lab efficiency, and the elimination of manual steps like cutting tips which reduces the chance of errors and cross-contamination. In addition, the product has been demonstrated to provide higher DNA yields than buccal swabs, reduces bacterial contamination and is stable for months at ambient temperatures.
ORAcollect•Dx is a complementary offering to Oragene•Dx, which received 510(k) clearance in 2011. DNA Genotek’s FDA clearances provide access to cleared, saliva-based collection devices for diagnostic companies and healthcare facilities of all sizes who want to deliver an improved patient experience as well as facilitate growth of their testing services through easier access to patients.
“Our portfolio of FDA cleared products offers our diagnostic customers in the U.S. a broad range of options for DNA collection for tests requiring both low and high amounts of high quality DNA. ORAcollect•Dx fills a critical need for a cleared device that improves on the stability and yield of buccal swabs while offering a liquid sample,” said Brian Smith, general manager, Genomics and Microbiome, at DNA Genotek Inc. “Molecular diagnostic and laboratory developed test manufacturers can reduce the time and regulatory burden of their own FDA submissions by leveraging the 510(k) clearance of ORAcollect•Dx.”
ORAcollect•Dx is FDA-cleared for in-vitro diagnostic use with the eSensor Warfarin Sensitivity Saliva test.
DNA Genotek Inc., a subsidiary of OraSure Technologies Inc. provides biological sample collection products and end-to-end services for human genomics, microbiome and infectious disease applications. The Canadian company also offers research use only products to collect and preserve large amounts of DNA or RNA from multiple sample types. DNA Genotek markets its products worldwide and has a global customer base with thousands of customers in more than 100 countries.