Business Wire05.02.16
Direct Flow Medical Inc., a company developing and commercializing transcatheter heart valve therapy products to treat structural heart disease, has modified its U.S. SALUS pivotal trial to include best practices and clinical comparison with all currently available U.S. transcatheter aortic valve replacement (TAVR) devices.
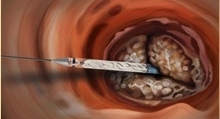
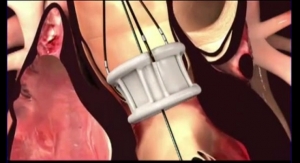
The Direct Flow Medical transcatheter aortic valve system, with more than 2,500 implants and excellent published clinical results including low rates of paravalvular leak, pacemaker implant, and mortality, is the only non-metallic, conformable transcatheter aortic valve that can be functionally assessed, repositioned, and retrieved prior to final implant.
“The strengthened SALUS trial design reflects our agility in a dynamic global TAVR market to provide the scientific community with a truly meaningful investigation and enhances our ability to achieve our year-end enrollment target,” said Dan Lemaitre, president and CEO.
The SALUS pivotal trial was initiated June 2015 with target enrollment of 1,262 subjects at up to 45 U.S. investigational sites to evaluate the Direct Flow Medical TAVR System with one TAVR device. Based on recent U.S. Food and Drug Administration approvals, the trial now features improved screening, enhanced options for anesthesia, optional intraprocedural dilatation, use of the new DirecTrack delivery system, comparison with all current commercially-available U.S. TAVR devices and a new target enrollment of 648 subjects.
“Utilizing best practices, comparing with all TAVR devices and being able to evaluate a new delivery system places the SALUS Trial at the leading edge of TAVR science,” stated Scott Lim, M.D., University of Virginia, and SALUS trial co-principal investigator. “These changes elevate SALUS trial value and meaningfully evaluate product performance relative to available TAVR technology,” echoed Isaac George, M.D. Columbia University, and SALUS trial co-principal investigator.
Direct Flow Medical is a privately held medical device company developing and commercializing novel transcatheter heart valve therapy products to treat structural heart disease. The Direct Flow Medical Transcatheter Aortic Valve System for treating high and extreme risk severe symptomatic aortic stenosis is commercially available in Europe for treating extreme surgical risk aortic stenosis.
The Direct Flow Medical transcatheter aortic valve system, with more than 2,500 implants and excellent published clinical results including low rates of paravalvular leak, pacemaker implant, and mortality, is the only non-metallic, conformable transcatheter aortic valve that can be functionally assessed, repositioned, and retrieved prior to final implant.
“The strengthened SALUS trial design reflects our agility in a dynamic global TAVR market to provide the scientific community with a truly meaningful investigation and enhances our ability to achieve our year-end enrollment target,” said Dan Lemaitre, president and CEO.
The SALUS pivotal trial was initiated June 2015 with target enrollment of 1,262 subjects at up to 45 U.S. investigational sites to evaluate the Direct Flow Medical TAVR System with one TAVR device. Based on recent U.S. Food and Drug Administration approvals, the trial now features improved screening, enhanced options for anesthesia, optional intraprocedural dilatation, use of the new DirecTrack delivery system, comparison with all current commercially-available U.S. TAVR devices and a new target enrollment of 648 subjects.
“Utilizing best practices, comparing with all TAVR devices and being able to evaluate a new delivery system places the SALUS Trial at the leading edge of TAVR science,” stated Scott Lim, M.D., University of Virginia, and SALUS trial co-principal investigator. “These changes elevate SALUS trial value and meaningfully evaluate product performance relative to available TAVR technology,” echoed Isaac George, M.D. Columbia University, and SALUS trial co-principal investigator.
Direct Flow Medical is a privately held medical device company developing and commercializing novel transcatheter heart valve therapy products to treat structural heart disease. The Direct Flow Medical Transcatheter Aortic Valve System for treating high and extreme risk severe symptomatic aortic stenosis is commercially available in Europe for treating extreme surgical risk aortic stenosis.