09.28.15
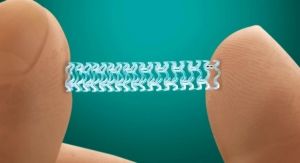
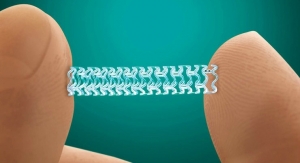
Stentys Inc.’s drug-eluting stent has earned the CE mark for treatment of below-the-knee (BTK) arteries. This approval makes the product the first self-expanding and drug-eluting stent with regulatory approval in Europe for this indication, according to Stentys.
The CE mark was obtained following the strong results achieved by the stent in the PES BTK-70 study. Stentys reported that the product prevented foot amputation in 99 percent of the 70 patients treated for critical limb ischemia (CLI).
The most severe cases of CLI, in which tissue of the foot or ankle does not receive a sufficient supply of oxygen and begins to die (necrosis), often require amputation. To prevent this outcome, surgeons open up the artery with balloons and frequently implant stents. Approximately 25 percent of diabetics globally suffer from CLI and, according to the International Diabetes Foundation, the world-wide diabetic population is projected to increase from 387 million in 2014 to nearly 600 million individuals by 2035.
“Entering the peripheral arterial disease market, which, outside of the US and Japan, is estimated at $1.3 billion, represents a significant long term growth opportunity for Stentys,” said Gonzague Issenmann, CEO and co-founder of Stentys. “Now that the product is CE Marked, becoming the first stent that combines the benefits of self-expansion and drug-elution in this indication in Europe, we intend to formalize new distribution partnerships and expect to begin commercializing the product in 2016.”
The PES BTK-70 trial treated 70 patients suffering from CLI of class 4 and 5 in the Rutherford scale with a Stentys Paclitaxel-eluting stent from January 2012 to May 2013 in five hospitals. The primary endpoint was the 12 month primary patency rate defined as absence of restenosis (≥50%) or occlusion within the originally treated lesion based on angiography verified by Core Lab. At 12 months, the primary patency rate was 73 percent, freedom from target-lesion revascularization was 79 percent and freedom from amputation was 99 percent.
Stentys is headquartered in Paris, France. It’s U.S. head offices are in Princeton, N.J.
The CE mark was obtained following the strong results achieved by the stent in the PES BTK-70 study. Stentys reported that the product prevented foot amputation in 99 percent of the 70 patients treated for critical limb ischemia (CLI).
The most severe cases of CLI, in which tissue of the foot or ankle does not receive a sufficient supply of oxygen and begins to die (necrosis), often require amputation. To prevent this outcome, surgeons open up the artery with balloons and frequently implant stents. Approximately 25 percent of diabetics globally suffer from CLI and, according to the International Diabetes Foundation, the world-wide diabetic population is projected to increase from 387 million in 2014 to nearly 600 million individuals by 2035.
“Entering the peripheral arterial disease market, which, outside of the US and Japan, is estimated at $1.3 billion, represents a significant long term growth opportunity for Stentys,” said Gonzague Issenmann, CEO and co-founder of Stentys. “Now that the product is CE Marked, becoming the first stent that combines the benefits of self-expansion and drug-elution in this indication in Europe, we intend to formalize new distribution partnerships and expect to begin commercializing the product in 2016.”
The PES BTK-70 trial treated 70 patients suffering from CLI of class 4 and 5 in the Rutherford scale with a Stentys Paclitaxel-eluting stent from January 2012 to May 2013 in five hospitals. The primary endpoint was the 12 month primary patency rate defined as absence of restenosis (≥50%) or occlusion within the originally treated lesion based on angiography verified by Core Lab. At 12 months, the primary patency rate was 73 percent, freedom from target-lesion revascularization was 79 percent and freedom from amputation was 99 percent.
Stentys is headquartered in Paris, France. It’s U.S. head offices are in Princeton, N.J.