Codman Neuro08.04.16
Codman Neuro, part of DePuy Synthes Companies of Johnson & Johnson, has entered into an agreement with InNeuroCo, Inc. for exclusive U.S. and European distribution rights for intermediate catheters that enable minimally invasive treatment of hemorrhagic stroke. The announcement was made here at the Society of NeuroInterventional Surgery (SNIS) 13th Annual Meeting.
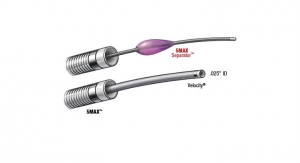
The agreement includes the InNeuroCo Intermediate Catheter (IC) and the InNeuroCo Super Distal Access (SDA) device. The InNeuroCo IC, which has already received regulatory approval in the U.S. and Europe, has shown optimized tracking during insertion.1 The InNeuroCo SDA is pending U.S. FDA 510 (k) clearance. The catheters will be offered alongside the full line of Codman Neuro microcatheters and guiding catheters, which support the delivery of neurovascular products including the CODMAN ENTERPRISE 2 Vascular Reconstruction Device.
“Intermediate catheters are increasingly used by neurovascular specialists to facilitate the delivery of other devices, including stents and thrombectomy devices for the treatment of stroke,” said P. Laxmin Laxminarain, worldwide president of Codman Neuro. “This agreement with InNeuroCo demonstrates our commitment to neurovascular care and will help us strengthen our access portfolio and our ability to provide a total solution in this fast growing market.”
InNeuroCo, Inc. is a privately held, Florida-based company that develops and manufactures neurovascular products for hemorrhagic stroke (aneurysms).
According to the Centers for Disease Control and Prevention (CDC), stroke is the fifth leading cause of death in the United States and is a major cause of adult disability. About 800,000 people have a stroke each year and one American dies from a stroke every four minutes.2 The American Stroke Association estimates 3 to 5 million people in the United States have some form of brain aneurysm, though most do not produce any symptoms. However, between 0.5 and 3 percent of people with a brain aneurysm may suffer from bleeding and rupture and require treatment.3
The CODMAN ENTERPRISE Vascular Reconstruction Device and the CODMAN ENTERPRISE 2 Vascular Reconstruction Device are Humanitarian Use Devices approved by the FDA under a Humanitarian Device Exemption (HDE) in the United States Only, where it is authorized by Federal Law for use with embolic coils for the treatment of wide-neck, intracranial, saccular or fusiform aneurysms arising from a parent vessel with a diameter of ≥2.5 mm and ≤4 mm. Wide-neck is defined as having a neck width ≥4 mm or a dome-to-neck ratio <2.
1 Codman Neuro, Data on File: Market Preference Case Evaluation.
2 http://www.cdc.gov/stroke/ accessed May 2016
3 http://www.strokeassociation.org/STROKEORG/AboutStroke/TypesofStroke/HemorrhagicBleeds/What-You-Should-Know-About-Cerebral-Aneurysms_UCM_310103_Article.jsp#.Vz9MO2NllBy; accessed May 2016.
The agreement includes the InNeuroCo Intermediate Catheter (IC) and the InNeuroCo Super Distal Access (SDA) device. The InNeuroCo IC, which has already received regulatory approval in the U.S. and Europe, has shown optimized tracking during insertion.1 The InNeuroCo SDA is pending U.S. FDA 510 (k) clearance. The catheters will be offered alongside the full line of Codman Neuro microcatheters and guiding catheters, which support the delivery of neurovascular products including the CODMAN ENTERPRISE 2 Vascular Reconstruction Device.
“Intermediate catheters are increasingly used by neurovascular specialists to facilitate the delivery of other devices, including stents and thrombectomy devices for the treatment of stroke,” said P. Laxmin Laxminarain, worldwide president of Codman Neuro. “This agreement with InNeuroCo demonstrates our commitment to neurovascular care and will help us strengthen our access portfolio and our ability to provide a total solution in this fast growing market.”
InNeuroCo, Inc. is a privately held, Florida-based company that develops and manufactures neurovascular products for hemorrhagic stroke (aneurysms).
According to the Centers for Disease Control and Prevention (CDC), stroke is the fifth leading cause of death in the United States and is a major cause of adult disability. About 800,000 people have a stroke each year and one American dies from a stroke every four minutes.2 The American Stroke Association estimates 3 to 5 million people in the United States have some form of brain aneurysm, though most do not produce any symptoms. However, between 0.5 and 3 percent of people with a brain aneurysm may suffer from bleeding and rupture and require treatment.3
The CODMAN ENTERPRISE Vascular Reconstruction Device and the CODMAN ENTERPRISE 2 Vascular Reconstruction Device are Humanitarian Use Devices approved by the FDA under a Humanitarian Device Exemption (HDE) in the United States Only, where it is authorized by Federal Law for use with embolic coils for the treatment of wide-neck, intracranial, saccular or fusiform aneurysms arising from a parent vessel with a diameter of ≥2.5 mm and ≤4 mm. Wide-neck is defined as having a neck width ≥4 mm or a dome-to-neck ratio <2.
1 Codman Neuro, Data on File: Market Preference Case Evaluation.
2 http://www.cdc.gov/stroke/ accessed May 2016
3 http://www.strokeassociation.org/STROKEORG/AboutStroke/TypesofStroke/HemorrhagicBleeds/What-You-Should-Know-About-Cerebral-Aneurysms_UCM_310103_Article.jsp#.Vz9MO2NllBy; accessed May 2016.