08.07.15
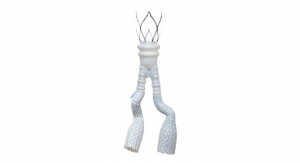
TriVascular Technologies Inc. has received U.S. Food and Drug Administration (FDA) approval for its Ovation iX Abdominal Stent Graft System.
Delivery system enhancements of Ovation iX --- which stands for integrated exchange --- are designed to provide physicians with additional flexibility for even more predictable deployment and greater ease of use, according to the company. A new, built-in procedural option is available to replace retrograde cannulation of the main body device, facilitating placement of the contralateral iliac stent graft. A low profile, 12-13F integrated sheath is designed to minimize vessel trauma and reduce procedural steps. At 14F OD, the Ovation iX abdominal stent graft system remains the lowest profile FDA-approved EVAR (endovascular aneurysm) device, TriVascular bigwigs claim.
"The improvements reflected in the new Ovation iX aortic body set the Ovation(R) system even further ahead in terms of ease of use and predictability of deployment. This allows me to treat both straightforward and more challenging AAA anatomies with confidence," said Steve Henao, M.D., a vascular surgeon at New Mexico Heart Institute. "By providing an alternative to retrograde cannulation --- there when I need it, imperceptible when I don't --- the Ovation iX system offers not only excellent accuracy, but also procedural predictability. As a result, I am confident in my ability to offer my patients consistent, high quality clinical outcomes with the Ovation system."
The FDA approval of the Ovation iX Abdominal Stent Graft System marks the sixth FDA approval related to the Ovation platform since September 2011. The ease-of-use enhancements incorporated into the iX aortic body delivery system, as well as the recently approved iX iliac limb technology, are a direct result of physician input and indicative of TriVascular's responsiveness to clinical feedback.
"The launch of the complete Ovation iX system supports our mission to improve and safely expand EVAR for all and serves as evidence of our commitment to deliver a strong pipeline of products," said Christopher G. Chavez, chairman/CEO/president of TriVascular. "We believe the Ovation iX delivery system enhancements will complement the broad IFU of the Ovation system and further enable our physician partners to provide improved, on-label EVAR treatment to more patients."
The Ovation platform has been used in the successful treatment of more than 7,000 patients worldwide, TriVascular executives said. Positive clinical results have been reported from both the Ovation global pivotal trial and a 501-patient European Post-Market Registry. The Ovation platform is available for sale in more than 35 countries.
TriVascular is a medical device company developing and commercializing abdominal aortic aneurysm treatment technologies. The company is based in Santa Rosa, Calif.
Delivery system enhancements of Ovation iX --- which stands for integrated exchange --- are designed to provide physicians with additional flexibility for even more predictable deployment and greater ease of use, according to the company. A new, built-in procedural option is available to replace retrograde cannulation of the main body device, facilitating placement of the contralateral iliac stent graft. A low profile, 12-13F integrated sheath is designed to minimize vessel trauma and reduce procedural steps. At 14F OD, the Ovation iX abdominal stent graft system remains the lowest profile FDA-approved EVAR (endovascular aneurysm) device, TriVascular bigwigs claim.
"The improvements reflected in the new Ovation iX aortic body set the Ovation(R) system even further ahead in terms of ease of use and predictability of deployment. This allows me to treat both straightforward and more challenging AAA anatomies with confidence," said Steve Henao, M.D., a vascular surgeon at New Mexico Heart Institute. "By providing an alternative to retrograde cannulation --- there when I need it, imperceptible when I don't --- the Ovation iX system offers not only excellent accuracy, but also procedural predictability. As a result, I am confident in my ability to offer my patients consistent, high quality clinical outcomes with the Ovation system."
The FDA approval of the Ovation iX Abdominal Stent Graft System marks the sixth FDA approval related to the Ovation platform since September 2011. The ease-of-use enhancements incorporated into the iX aortic body delivery system, as well as the recently approved iX iliac limb technology, are a direct result of physician input and indicative of TriVascular's responsiveness to clinical feedback.
"The launch of the complete Ovation iX system supports our mission to improve and safely expand EVAR for all and serves as evidence of our commitment to deliver a strong pipeline of products," said Christopher G. Chavez, chairman/CEO/president of TriVascular. "We believe the Ovation iX delivery system enhancements will complement the broad IFU of the Ovation system and further enable our physician partners to provide improved, on-label EVAR treatment to more patients."
The Ovation platform has been used in the successful treatment of more than 7,000 patients worldwide, TriVascular executives said. Positive clinical results have been reported from both the Ovation global pivotal trial and a 501-patient European Post-Market Registry. The Ovation platform is available for sale in more than 35 countries.
TriVascular is a medical device company developing and commercializing abdominal aortic aneurysm treatment technologies. The company is based in Santa Rosa, Calif.