06.08.15
Bigfoot Biomedical Inc. is picking up its product development pace with a recent acquisition and partnership.
The startup has acquired all assets of Asante Solutions, manufacturer of the Asante Snap insulin pump and entered a product development deal with Dexcom Inc.
Asante announced on May 15 that it was closing its business. Though no official reason was given for the company’s closure, industry watchers speculate that despite the popularity of Asante’s Snap pump technology with users, tough industry competition and reimbursement challenges led to the decision to close.
Terms of the Bigfoot/Asante deal were not disclosed.
Bigfoot is developing a Type 1 diabetes (T1D) management system that the company claims will employ a simple design, cloud connectivity, and human-centered automation. According to Bigfoot’s leadership, the Asante acquisition marks a “strategically meaningful step” toward the firm’s goal of providing all people with T1D an “accessible, affordable, automated, connected solution.”
“The 10 years of work and technology behind Asante’s Snap pump was impressive. This technology will live on, and provides Bigfoot a cornerstone for our next generation T1D management system,” said Jeffrey Brewer, president and CEO of Bigfoot. “The acquisition will dramatically accelerate our time to market for our automated insulin delivery system, which we believe will be a transformative therapy for people affected by T1D.”
The acquired assets will afford Bigfoot a significant portfolio of intellectual property and operational infrastructure upon which to build, and will specifically enable the development of the insulin pump component of the Bigfoot automated insulin delivery system. The company has no current plans to market the existing Asante Snap, but will focus on the development and testing of its integrated and automated T1D management system.
“We also expect this investment to de-risk the regulatory approval pathway, as well as substantially expedite our clinical trial plans for our automated insulin delivery system,” said Bryan Mazlish, Bigfoot’s chief technology officer and inventor of the Bigfoot prototype system. “The Asante Snap has real-world use history and a 510(k) clearance from the FDA (U.S. Food and Drug Administration), which provides an excellent precedent upon which to build with the agency.”
“We remain committed to delivering a holistic outcome-based T1D management system. This acquisition accelerates our timelines and will provide a key enabler for our broader service strategy,” added Lane Desborough, Bigfoot’s chief engineer. “With the acquisition of Asante’s assets, we’ll be positioned to deliver a fully comprehensive T1D management solution much faster—to a community that needs it now.”
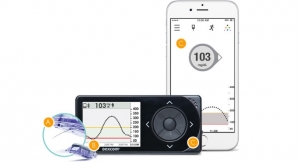
Bigfoot also recently formed development agreement with Dexcom to integrate data from the Dexcom Continuous Glucose Monitoring (CGM) System into the Bigfoot insulin delivery system.
The newly announced partnership will enable glucose data to travel seamlessly from the Dexcom transmitter into the Bigfoot system to enable automated delivery of insulin for people with T1D.
“Dexcom’s advanced technologies are a great fit for Bigfoot’s pioneering efforts towards an artificial pancreas,” said Steve Pacelli, executive vice president, strategy and corporate development, at Dexcom. “Bigfoot has made a commitment to automated insulin control based on CGM data, and Dexcom is excited to participate in their development e orts with our sensor technology.”
“We are thrilled to be working with such an innovative, impactful and highly-respected partner,” said Brewer. “Bigfoot Biomedical exists to serve the needs of the T1D community, and this partnership is our most important collaboration towards delivering an automated insulin delivery solution for people with T1D.”
Bigfoot has built a prototype automated insulin delivery system, otherwise known as an artificial pancreas, that’s been used in the real world for more than 30,000 hours. The device is being used by one of the founders’ children. Originally based in New York, N.Y., the company will move its headquarters to Milpitas, Calif., following the Asante purchase.
Earlier this year, the company secured a $3.5 million bridge convertible note. Officials want to raise between $10 million and $15 million in a Series A this summer. No word yet on clinical trial plans or timeline for FDA submission.
The startup has acquired all assets of Asante Solutions, manufacturer of the Asante Snap insulin pump and entered a product development deal with Dexcom Inc.
Asante announced on May 15 that it was closing its business. Though no official reason was given for the company’s closure, industry watchers speculate that despite the popularity of Asante’s Snap pump technology with users, tough industry competition and reimbursement challenges led to the decision to close.
Terms of the Bigfoot/Asante deal were not disclosed.
Bigfoot is developing a Type 1 diabetes (T1D) management system that the company claims will employ a simple design, cloud connectivity, and human-centered automation. According to Bigfoot’s leadership, the Asante acquisition marks a “strategically meaningful step” toward the firm’s goal of providing all people with T1D an “accessible, affordable, automated, connected solution.”
“The 10 years of work and technology behind Asante’s Snap pump was impressive. This technology will live on, and provides Bigfoot a cornerstone for our next generation T1D management system,” said Jeffrey Brewer, president and CEO of Bigfoot. “The acquisition will dramatically accelerate our time to market for our automated insulin delivery system, which we believe will be a transformative therapy for people affected by T1D.”
The acquired assets will afford Bigfoot a significant portfolio of intellectual property and operational infrastructure upon which to build, and will specifically enable the development of the insulin pump component of the Bigfoot automated insulin delivery system. The company has no current plans to market the existing Asante Snap, but will focus on the development and testing of its integrated and automated T1D management system.
“We also expect this investment to de-risk the regulatory approval pathway, as well as substantially expedite our clinical trial plans for our automated insulin delivery system,” said Bryan Mazlish, Bigfoot’s chief technology officer and inventor of the Bigfoot prototype system. “The Asante Snap has real-world use history and a 510(k) clearance from the FDA (U.S. Food and Drug Administration), which provides an excellent precedent upon which to build with the agency.”
“We remain committed to delivering a holistic outcome-based T1D management system. This acquisition accelerates our timelines and will provide a key enabler for our broader service strategy,” added Lane Desborough, Bigfoot’s chief engineer. “With the acquisition of Asante’s assets, we’ll be positioned to deliver a fully comprehensive T1D management solution much faster—to a community that needs it now.”
Bigfoot also recently formed development agreement with Dexcom to integrate data from the Dexcom Continuous Glucose Monitoring (CGM) System into the Bigfoot insulin delivery system.
The newly announced partnership will enable glucose data to travel seamlessly from the Dexcom transmitter into the Bigfoot system to enable automated delivery of insulin for people with T1D.
“Dexcom’s advanced technologies are a great fit for Bigfoot’s pioneering efforts towards an artificial pancreas,” said Steve Pacelli, executive vice president, strategy and corporate development, at Dexcom. “Bigfoot has made a commitment to automated insulin control based on CGM data, and Dexcom is excited to participate in their development e orts with our sensor technology.”
“We are thrilled to be working with such an innovative, impactful and highly-respected partner,” said Brewer. “Bigfoot Biomedical exists to serve the needs of the T1D community, and this partnership is our most important collaboration towards delivering an automated insulin delivery solution for people with T1D.”
Bigfoot has built a prototype automated insulin delivery system, otherwise known as an artificial pancreas, that’s been used in the real world for more than 30,000 hours. The device is being used by one of the founders’ children. Originally based in New York, N.Y., the company will move its headquarters to Milpitas, Calif., following the Asante purchase.
Earlier this year, the company secured a $3.5 million bridge convertible note. Officials want to raise between $10 million and $15 million in a Series A this summer. No word yet on clinical trial plans or timeline for FDA submission.