02.10.15
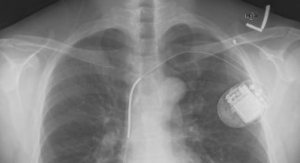
St. Jude Medical Inc. has launched its Halo U.S. IDE (investigational device exemption) trial. The trial will evaluate the safety and efficacy of the St. Jude Medical Masters HP series 15 mm mechanical pediatric heart valve. According to St. Jude, this is the smallest pediatric heart valve yet, and the Halo trial will enroll pediatric patients in need of mitral valve replacement who cannot be implanted with the current range of approved valves without additional risks and who have no alternative approved treatment options.
In the United States alone, more than 35,000 babies are born each year with congenital heart defects, some of which will impact valve function to the point they’ll require surgical valve replacement. For many of these patients, mechanical heart valves can effectively mimic natural heart valves, opening and closing with each heart beat and permitting blood flow into the heart. While there are several replacement valves of varying size available for older patients, there are no prosthetic heart valves under 16 mm in tissue diameter approved for standard use in newborns, infants and toddlers. With limited availability of valves that can suitably match the anatomy of children under the age of five, some surgeons are forced to implant larger valves in their smallest patients, which may lead to suboptimal placement that can disrupt hemodynamics, damage to surrounding tissue and an elevated risk of side effects such as poor blood pressure and potential heart blockage.
“There’s a profound need within the medical community for a mechanical heart valve dedicated to our tiniest patients who can’t safely be implanted with the current range of valve sizes,” said Kirk R. Kanter, M.D., professor of surgery at the Emory University School of Medicine and cardiothoracic surgeon and surgical director of the Heart Transplant Program at Children’s Healthcare of Atlanta. “This small valve represents a big step forward in helping cardiovascular surgeons provide our patients the optimal level of care.”
The launch of the Halo IDE stems from a new approach to the design, evaluation and regulatory approval of pediatric heart valves that emerged from a U.S. Food and Drug Administration-led workshop in 2010 organized in response to the pressing need to develop products for the pediatric patient population.
The Halo trial will enroll patients at up to 40 sites nationwide. The data collected across all trial sites will be used to support FDA approval of the Masters Series 15 mm mechanical heart valve, which is currently available only under “emergency or compassionate use” protocols.
“The Masters Series 15 mm mechanical heart valve was developed based on feedback we received from physicians and the FDA’s Office of Pediatric Therapeutics, who have expressed the need for a more suitable mechanical heart valve to offer pediatric patients for which there are simply no other options,” said Mark Carlson, M.D., vice president of global clinical affairs and chief medical officer at St. Jude Medical. “As a market leader in pediatric cardiology solutions, St. Jude Medical has the capability to develop devices that fill critical needs, and we will continue to look for more opportunities to create solutions for some of the biggest challenges facing pediatric surgeons and cardiologists today.”
St. Jude Medical is a medical device company based in St. Paul, Minn.
In the United States alone, more than 35,000 babies are born each year with congenital heart defects, some of which will impact valve function to the point they’ll require surgical valve replacement. For many of these patients, mechanical heart valves can effectively mimic natural heart valves, opening and closing with each heart beat and permitting blood flow into the heart. While there are several replacement valves of varying size available for older patients, there are no prosthetic heart valves under 16 mm in tissue diameter approved for standard use in newborns, infants and toddlers. With limited availability of valves that can suitably match the anatomy of children under the age of five, some surgeons are forced to implant larger valves in their smallest patients, which may lead to suboptimal placement that can disrupt hemodynamics, damage to surrounding tissue and an elevated risk of side effects such as poor blood pressure and potential heart blockage.
“There’s a profound need within the medical community for a mechanical heart valve dedicated to our tiniest patients who can’t safely be implanted with the current range of valve sizes,” said Kirk R. Kanter, M.D., professor of surgery at the Emory University School of Medicine and cardiothoracic surgeon and surgical director of the Heart Transplant Program at Children’s Healthcare of Atlanta. “This small valve represents a big step forward in helping cardiovascular surgeons provide our patients the optimal level of care.”
The launch of the Halo IDE stems from a new approach to the design, evaluation and regulatory approval of pediatric heart valves that emerged from a U.S. Food and Drug Administration-led workshop in 2010 organized in response to the pressing need to develop products for the pediatric patient population.
The Halo trial will enroll patients at up to 40 sites nationwide. The data collected across all trial sites will be used to support FDA approval of the Masters Series 15 mm mechanical heart valve, which is currently available only under “emergency or compassionate use” protocols.
“The Masters Series 15 mm mechanical heart valve was developed based on feedback we received from physicians and the FDA’s Office of Pediatric Therapeutics, who have expressed the need for a more suitable mechanical heart valve to offer pediatric patients for which there are simply no other options,” said Mark Carlson, M.D., vice president of global clinical affairs and chief medical officer at St. Jude Medical. “As a market leader in pediatric cardiology solutions, St. Jude Medical has the capability to develop devices that fill critical needs, and we will continue to look for more opportunities to create solutions for some of the biggest challenges facing pediatric surgeons and cardiologists today.”
St. Jude Medical is a medical device company based in St. Paul, Minn.