Data comparing the Linx device and laparoscopic fundoplication surgery in 249 patients concluded that Torax's product provides protection from reflux without compromising the patient’s stomach while minimizing the side effects commonly associated with current surgical approaches. Patients suffering from gastro-esophageal reflux disease (GERD) often must undergo surgery to have their stomach tissue wrapped around the esophagus to create a barrier to reflux.
“When patients fail acid suppression therapy for their reflux disease it presents a significant clinical problem. In this study, the majority of patients reported having reflux related symptoms that were severe enough to interfere with daily activities despite taking acid suppression medications,” said Martin Riegler, M.D., of the Department of Surgery at Medical University, Vienna (Austria), and an author of the prospective, multicenter study, "Magnetic Sphincter Augmentation and Fundoplication for GERD in Clinical Practice," published in Surgical Endoscopy.
Following minimally invasive laparoscopic anti-reflux surgery with either the LINX device or fundoplication, both groups demonstrated a marked improvement in their quality of life, according to a news release from the company. Patients receiving the Linx device were less likely to experience bloating and were more likely to be able to belch and vomit following the procedure as compared to the fundoplication group. Discontinuation of acid suppression medications was significantly greater in the Linx group (81.8 percent) compared to the fundoplication group (63 percent).
“This study introduces an important breakthrough in the treatment options for reflux disease. Linx allows patients to have the reflux control benefits of fundoplication surgery without the significant side effects,” said Riegler.
The Linx device was shown to be both safe and effective following U.S. Food and Drug Administration (FDA)-approved clinical investigations and was later approved for U.S. distribution by the FDA in early 2012. The Linx device is available in both the United States and Europe.
Gastro-esophageal Reflux Disease is a chronic, often progressive disease resulting from a weak lower esophageal sphincter that allows harmful gastric fluid to reflux into the esophagus, resulting in both pain and injury to the esophageal lining. Symptoms of GERD include heartburn and regurgitation, often associated with chronic sleep disruption, and may also include persistent cough, excessive throat clearing, hoarseness and a feeling of a “lump” in the throat. Acid reflux medications, such as Prevacid, Nexium, and Prilosec affect gastric acid production, but do not repair the sphincter defect, allowing continued reflux. Anti-reflux surgery called Nissen Fundoplication reconstructs a new reflux barrier using a portion of the patient’s stomach which is wrapped around the lower portion of the esophagus. GERD is associated with a pre-cancerous condition known as Barrett’s esophagus, which increases the risk of esophageal cancer.
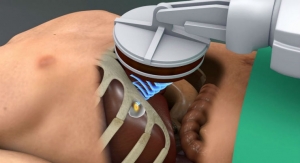
Linx is a small implant comprised of interlinked titanium beads with magnetic cores. The magnetic attraction between the beads augments the existing esophageal sphincter’s barrier function to prevent reflux. The device is implanted using a standard laparoscopic procedure and is an alternative to fundoplication, commonly used in surgical anti-reflux procedures. The Linx Reflux Management System is indicated for those patients diagnosed with Gastro-esophageal Reflux Disease as defined by abnormal pH testing, and who continue to have chronic GERD symptoms despite maximum medical therapy for the treatment of reflux.
Torax Medical is a privately-held company headquartered in St. Paul, Minn., that develops and markets products designed to treat sphincter disorders utilizing its technology platform, Magnetic Sphincter Augmentation (MSA). Torax Medical currently is marketing the Linx Reflux Management System for the treatment of GERD in the United States and Europe and the Fenix Continence Restoration System for the treatment of fecal incontinence in Europe.