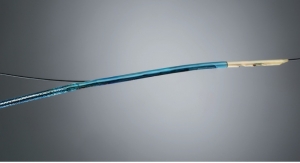
Veniti used funds from its $9.7 million Series B funding round in June 2013 to help develop the device, which already is commercially available in Europe. Veniti officials will start enrolling 170 patients in its U.S. clinical trials and begin selecting sites before the end of the year. After that, investigators will monitor those patients for another year.
Co-Principal investigators for the VIRTUS trial are William Marston, M.D., chief, Division of Vascular Surgery and professor, Department of Surgery at UNC Department of Surgery in Chapel Hill, N.C., and Mahmood Razavi, M.D., director for Clinical Trials & Research at the Heart and Vascular Center at St. Joseph Hospital in Orange, Calif.
The FDA milestone means the stent system likely will hit the U.S. market in late 2017.
"Obstruction of the iliac veins and vena cava have been identified as a frequent cause of severe venous disease resulting in major disabling symptoms including leg swelling, pain, and ulceration," said Marston. "The development of stents specifically designed for the unique characteristics of the venous system provide the potential to reduce these debilitating symptoms for patients, while improving the cost effectiveness of interventional treatment for venous obstruction."
Aside from the stent system, Veniti is building a portfolio of devices that address vein-related ailments, from treating varicose veins to catching blood clots and treating venous reflux disease. Its two other products are still in development.
“We are very pleased with the approval of our IDE and excited to be conducting this important research with physicians in both Europe and the U.S.” Veniti President/CEO Scott Solano said in a statement. “The unique advantages of a stent designed specifically for the venous system, including end-to-end crush resistance, flexibility, and continuous vein coverage, may offer significant patient benefit.”
The trial has begun in Europe, with first patients enrolled and treated by Marta Ramirez Ortega, M.D., at Hospital Madrid Monteprincipe in Madrid, Spain. "It is important for my patients to be able to be treated with a stent specifically designed for the venous anatomy and for them to have their data collected so that future patients can benefit from knowing how well venous stenting works," she said.
Hundreds of thousands of people worldwide suffer from lower-extremity venous disease, and until recently, the only treatment options available involved the use of re-purposed vascular stents originally designed for use in the arterial system.
"This is an important landmark for Veniti and patients with deep venous disease. Due to the absence of vein-specific stents, physicians have thus far been using stents that were not designed for veins. We look forward to the completion of this study and availability of optimal stents to treat our patients with deep vein obstructive lesions," Razavi said.
Veniti is located in St. Louis, Mo., but its manufacturing and research and development divisions are in Fremont, California. The company has 19 employees.