06.09.14
Mainstay Medical has secured approval from the Ethics Committees in Australia to start a clinical trial of ReActiv8, its implantable neurostimulation device for the treatment of people with Chronic Low Back Pain (CLBP). Patient recruitment for the trial already has begun at three clinical sites Down Under.
The purpose of the clinical trial is to investigate ReActiv8 as a treatment for adults with debilitating CLBP for whom surgery is not indicated.
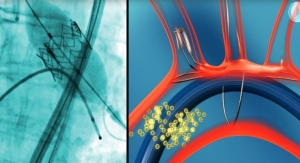
ReActiv8 helps patients with CLBP by providing electrical stimulation to impaired nerves near the spine. The device contracts the muscles, restoring muscle control and improving spine stability. Unlike traditional neuromodulation products, which rely on subcutaneous nerve stimulation, Mainstay's device can be implanted, delivering treatment in two, 30-minute daily sessions. The company has already started enrollment and plans on recruiting 96 subjects for tests at three test clinical sites in Australia, executives said.
Mainstay presented results from its European Feasibility Study of the device at the International Neuromodulation Society meeting in Berlin in June 2013. The study showed that its ReActiv8 device significantly reduced pain and disability from CLBP and improved patients' quality of life, the company said in a statement.
With promising clinical results for its ReActiv8 device, Mainstay hopes to cash in on a rapidly growing market. While industry leaders like Medtronic Inc. and Boston Scientific Corp. remain a step ahead of smaller operations, Mainstay could provide some healthy competition with its implantable neuromodulation technology. The company plans on securing CE mark approval for its device by the fourth quarter of 2015, and will begin preparing a formal investigational device exemption submission to start clinical trials in the United States this year.
One of the root causes of CLBP is impaired control by the nervous system of the muscles that stabilize the spine in the lower back, and an unstable spine can lead to back pain. ReActiv8 is designed to electrically stimulate the nerves responsible for contracting these muscles and thereby help to restore muscle control and improve spine stability, allowing the body to recover from chronic low back pain.
"Our novel approach of electrical stimulation to help restore the muscle control system is based on published scientific research, and the performance of the therapy was demonstrated in the recently completed European Feasibility Study," Mainstay CEO Peter Crosby said. "The energy and experience of the Mainstay Medical team has enabled us to complete the development of our innovative, therapy-specific device and obtain approval to start the ReActiv8 clinical trial within a year after the Feasibility Study results."
People with debilitating CLBP usually have a greatly reduced quality of life and score significantly higher on scales for disability, depression, anxiety and sleep disorders. Their pain and disability can persist despite the best available medical treatments, and most of these people have no indications for spine surgery. Their ability to work or be productive is seriously affected by the condition and the resulting days lost from work, disability benefits and health resource utilisation put a significant burden on economies.
"Back pain specialists from all over the world have told us that they need a new approach to help the large group of people with CLBP who are stranded without a viable alternative. We believe, based on published research, that there are millions of such people in Europe and the U.S.A. today. The results of the European Feasibility Study encouraged us to believe that ReActiv8 can play an important role in helping these people," Crosby said.
Mainstay Medical Limited is developing an implantable neurostimulation medical device for people suffering from debilitating CLBP. Lower back pain is the most common disability globally, with more research needed to better understand the disability, according to results of a recently published study.
The company is headquartered in Dublin, Ireland, and has subsidiaries operating in the United States and Australia. Mainstay Medical is backed by investors including Sofinnova Partners, Fountain Healthcare Partners, Medtronic, Capricorn Venture Partners, Seventure Partners and Twin Cities Angels.
The purpose of the clinical trial is to investigate ReActiv8 as a treatment for adults with debilitating CLBP for whom surgery is not indicated.
ReActiv8 helps patients with CLBP by providing electrical stimulation to impaired nerves near the spine. The device contracts the muscles, restoring muscle control and improving spine stability. Unlike traditional neuromodulation products, which rely on subcutaneous nerve stimulation, Mainstay's device can be implanted, delivering treatment in two, 30-minute daily sessions. The company has already started enrollment and plans on recruiting 96 subjects for tests at three test clinical sites in Australia, executives said.
Mainstay presented results from its European Feasibility Study of the device at the International Neuromodulation Society meeting in Berlin in June 2013. The study showed that its ReActiv8 device significantly reduced pain and disability from CLBP and improved patients' quality of life, the company said in a statement.
With promising clinical results for its ReActiv8 device, Mainstay hopes to cash in on a rapidly growing market. While industry leaders like Medtronic Inc. and Boston Scientific Corp. remain a step ahead of smaller operations, Mainstay could provide some healthy competition with its implantable neuromodulation technology. The company plans on securing CE mark approval for its device by the fourth quarter of 2015, and will begin preparing a formal investigational device exemption submission to start clinical trials in the United States this year.
One of the root causes of CLBP is impaired control by the nervous system of the muscles that stabilize the spine in the lower back, and an unstable spine can lead to back pain. ReActiv8 is designed to electrically stimulate the nerves responsible for contracting these muscles and thereby help to restore muscle control and improve spine stability, allowing the body to recover from chronic low back pain.
"Our novel approach of electrical stimulation to help restore the muscle control system is based on published scientific research, and the performance of the therapy was demonstrated in the recently completed European Feasibility Study," Mainstay CEO Peter Crosby said. "The energy and experience of the Mainstay Medical team has enabled us to complete the development of our innovative, therapy-specific device and obtain approval to start the ReActiv8 clinical trial within a year after the Feasibility Study results."
People with debilitating CLBP usually have a greatly reduced quality of life and score significantly higher on scales for disability, depression, anxiety and sleep disorders. Their pain and disability can persist despite the best available medical treatments, and most of these people have no indications for spine surgery. Their ability to work or be productive is seriously affected by the condition and the resulting days lost from work, disability benefits and health resource utilisation put a significant burden on economies.
"Back pain specialists from all over the world have told us that they need a new approach to help the large group of people with CLBP who are stranded without a viable alternative. We believe, based on published research, that there are millions of such people in Europe and the U.S.A. today. The results of the European Feasibility Study encouraged us to believe that ReActiv8 can play an important role in helping these people," Crosby said.
Mainstay Medical Limited is developing an implantable neurostimulation medical device for people suffering from debilitating CLBP. Lower back pain is the most common disability globally, with more research needed to better understand the disability, according to results of a recently published study.
The company is headquartered in Dublin, Ireland, and has subsidiaries operating in the United States and Australia. Mainstay Medical is backed by investors including Sofinnova Partners, Fountain Healthcare Partners, Medtronic, Capricorn Venture Partners, Seventure Partners and Twin Cities Angels.