05.08.14
Core Manufacturing LLC will manufacture a line of implantable drug delivery pumps from Palyon Medical Corporation, a medical device startup based in Santa Clarita, Calif.
Palyon, a venture-backed, developmental-stage company, has a drug delivery pump portfolio that includes programmable devices for maximum clinical flexibility and constant-flow devices for lower cost steady drug delivery. Core Manufacturing, a division of Cirtec, is a contract manufacturer that specializes in minimally invasive devices, active and passive implants, and medical electronics systems
“Core brings a level of clinical manufacturing experience that will greatly streamline our process and allow our researchers and engineers to focus on developing the next generation of devices, such as an implantable insulin pump,” said Michael Sember, Palyon's CEO. “Having Core’s production facility in such close proximity to us will allow for enhanced efficiency, reduced manufacturing coordination costs and speed our path to market.”
Under the new partnership, Core Manufacturing will provide manufacturing services, procurement coordination and dual quality control oversight, with the goal of reducing Palyon’s time-to-market introduction.
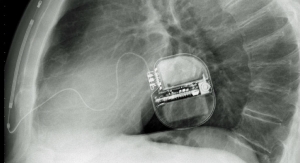
Palyon’s implantable infusion pump platform is designed to offer highly accurate drug delivery rates and incorporate unique safety features including the only sensor-based feedback flow control, refill error detection capability, a non-motorized pumping mechanism that is less susceptible to wear and tear, catheter occlusion detection and a highly favorable profile for use with magnetic resonance imaging, according to the company.
"Core Manufacturing is honored to partner with Palyon. We are uniquely qualified to develop and manufacture the lineup of Palyon’s active implantable devices. This partnership will allow both companies to leverage their core competencies," said Joel Hirsch, president of Core Manufacturing.
Core is located in Valencia, Calif., and is ISO 13485, ISO 9001, and U.S. Food and Drug Administration approved manufacturer of implantable, minimally invasive, and peripheral medical devices. The company's capabilities include rapid-turn prototyping, full turnkey device-level assembly, sterilization and testing.
Palyon, a venture-backed, developmental-stage company, has a drug delivery pump portfolio that includes programmable devices for maximum clinical flexibility and constant-flow devices for lower cost steady drug delivery. Core Manufacturing, a division of Cirtec, is a contract manufacturer that specializes in minimally invasive devices, active and passive implants, and medical electronics systems
“Core brings a level of clinical manufacturing experience that will greatly streamline our process and allow our researchers and engineers to focus on developing the next generation of devices, such as an implantable insulin pump,” said Michael Sember, Palyon's CEO. “Having Core’s production facility in such close proximity to us will allow for enhanced efficiency, reduced manufacturing coordination costs and speed our path to market.”
Under the new partnership, Core Manufacturing will provide manufacturing services, procurement coordination and dual quality control oversight, with the goal of reducing Palyon’s time-to-market introduction.
Palyon’s implantable infusion pump platform is designed to offer highly accurate drug delivery rates and incorporate unique safety features including the only sensor-based feedback flow control, refill error detection capability, a non-motorized pumping mechanism that is less susceptible to wear and tear, catheter occlusion detection and a highly favorable profile for use with magnetic resonance imaging, according to the company.
"Core Manufacturing is honored to partner with Palyon. We are uniquely qualified to develop and manufacture the lineup of Palyon’s active implantable devices. This partnership will allow both companies to leverage their core competencies," said Joel Hirsch, president of Core Manufacturing.
Core is located in Valencia, Calif., and is ISO 13485, ISO 9001, and U.S. Food and Drug Administration approved manufacturer of implantable, minimally invasive, and peripheral medical devices. The company's capabilities include rapid-turn prototyping, full turnkey device-level assembly, sterilization and testing.