04.07.14
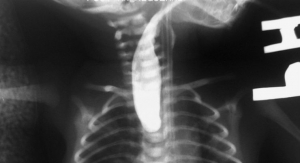
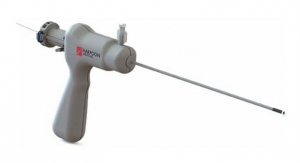
The U.S. Food and Drug Administration has approved the SafeSept Needle Free Transseptal Guidewire from Pressure Products Medical Supplies Inc. for use with any introducer system when crossing the interartrial septum. The device assists in transseptal procedures and is designed to create the primary puncture in the interatrial septum providing access from the right to left side of the heart without using a transseptal needle.
“The SafeSept NF adds another level of safety for TSP especially those done under fluoroscopy guidance alone and has overcome difficult cases of high fibrotic resistance,” said Anthony Chow, M.B.B.S., BSc, M.D., F.R.C.P., consultant cardiologist (electrophysiologist) at The Heart Hospital, UCLH, in London, United Kingdom.
The SafeSept Needle Free Transseptal Guidewire’s sharp tip requires little force to perforate and cross the fossa. Once penetrated and unsupported by the dilator and sheath, the tip assumes a ‘J’ shape, making it incapable of further tissue penetration, the company claims. A radiopaque coil allows for fluoroscopic visualization in the left atrium and subsequent pulmonary veins. “Over the wire” advancement of the transseptal dilator and sheath into the left atrium is accomplished without the use of a transseptal needle.
“The SafeSept Needle Free has become my preferred method for all transseptal punctures. Issues with thickened or floppy septums and concerns about scraping plastic from the inside of dilators have been eliminated,” said James McCready, M.B.B.S. M.R.C.P., consultant electrophysiologist at Royal Sussex County Hospital in Brighton, United Kingdom. “When the wire has not been available, I find going back to using a traditional needle awkward and uncomfortable.”
Launched by two cardiologists in 1988, Pressure Products offers hemostatic peel-away and sliceable introducers and accessories, as well as medical devices for femoral and pacemaker pocket hemostasis.
“The SafeSept NF adds another level of safety for TSP especially those done under fluoroscopy guidance alone and has overcome difficult cases of high fibrotic resistance,” said Anthony Chow, M.B.B.S., BSc, M.D., F.R.C.P., consultant cardiologist (electrophysiologist) at The Heart Hospital, UCLH, in London, United Kingdom.
The SafeSept Needle Free Transseptal Guidewire’s sharp tip requires little force to perforate and cross the fossa. Once penetrated and unsupported by the dilator and sheath, the tip assumes a ‘J’ shape, making it incapable of further tissue penetration, the company claims. A radiopaque coil allows for fluoroscopic visualization in the left atrium and subsequent pulmonary veins. “Over the wire” advancement of the transseptal dilator and sheath into the left atrium is accomplished without the use of a transseptal needle.
“The SafeSept Needle Free has become my preferred method for all transseptal punctures. Issues with thickened or floppy septums and concerns about scraping plastic from the inside of dilators have been eliminated,” said James McCready, M.B.B.S. M.R.C.P., consultant electrophysiologist at Royal Sussex County Hospital in Brighton, United Kingdom. “When the wire has not been available, I find going back to using a traditional needle awkward and uncomfortable.”
Launched by two cardiologists in 1988, Pressure Products offers hemostatic peel-away and sliceable introducers and accessories, as well as medical devices for femoral and pacemaker pocket hemostasis.