03.13.14
Interface Biologics Inc. (IBI) and Q3 Medical Devices Limited are teaming up to develop a next-generation drug-coated balloon for both coronary and peripheral applications.
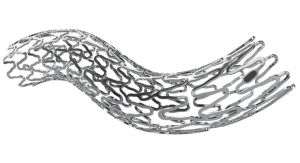
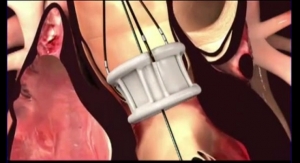
The product will combine IBI's Kinesyx bioactive oligomers with the balloon technology of Q3 Medical Devices' wholly owned subsidiary, Qualimed Innovative Medizinprodukte GmbH. "The combination of the Qualimed PTA/PTCA balloons with IBI's Kinesyx coating technology provides a compelling delivery platform for drugs that reduce restenosis in both coronary and peripheral applications," said Tom Reeves, president and CEO of Interface Biologics. "Coating uniformity and flexibility/pliability, low loss in transit and minimal particulate development as well as effective drug loading and tissue transfer combine to provide a strong competitive offering."
Reeves said the development and commercialization agreement is the first the company has signed involving its coating technology.
"We've been working with IBI over the last two years in developing the initial prototypes of a new drug coated balloon (DCB)," noted Eric Mangiardi, CEO of Q3 Medical. "Our customers have been asking for a DCB to complement our existing product portfolio and we believe that the Qualimed/IBI DCB will provide a compelling option. We look forward to working with IBI to get this product on the market and will leverage our experience in the CE Mark process to drive this forward."
IBI is a privately held commercial stage developer of biomedical-polymer medical device products. Q3 is an Irish based holding company with multiple operations in Germany focused on the development, manufacturing and distribution of minimally invasive devices for the treatment of patients with cardiology, peripheral vascular and non-vascular diseases.
Q3 Medical Devices Limited was formed by a global group of entrepreneurs, manufactures, distributors, industry doctors, and investors focused on the development and acquisition of medical device businesses with annual revenues between $1 million and $10 million. The acquisitions are targeted in areas that expand the groups manufacturing base and capabilities, growth of its distribution channels, and accelerating its products offering focused on the minimally invasive treatment of patients with cardiology, peripheral vascular and non-vascular diseases.
Qualimed, a wholly owned subsidiary of Q3 Medical, was founded in 1997 in Winsen, Germany near Hamburg, where it develops, manufactures, and sells implantable medical devices in the cardiology, peripheral vascular, and non-vascular areas. The innovations are focused on biodegradable products, drug-device combination technologies, catheter, and mechanical implant areas. Originally founded as an OEM, the company's products are now sold in more than 50 countries worldwide; the company and its development partners have obtained CE and FDA approvals for more than 70 different products including 3 different drug eluting stents.
The product will combine IBI's Kinesyx bioactive oligomers with the balloon technology of Q3 Medical Devices' wholly owned subsidiary, Qualimed Innovative Medizinprodukte GmbH. "The combination of the Qualimed PTA/PTCA balloons with IBI's Kinesyx coating technology provides a compelling delivery platform for drugs that reduce restenosis in both coronary and peripheral applications," said Tom Reeves, president and CEO of Interface Biologics. "Coating uniformity and flexibility/pliability, low loss in transit and minimal particulate development as well as effective drug loading and tissue transfer combine to provide a strong competitive offering."
Reeves said the development and commercialization agreement is the first the company has signed involving its coating technology.
"We've been working with IBI over the last two years in developing the initial prototypes of a new drug coated balloon (DCB)," noted Eric Mangiardi, CEO of Q3 Medical. "Our customers have been asking for a DCB to complement our existing product portfolio and we believe that the Qualimed/IBI DCB will provide a compelling option. We look forward to working with IBI to get this product on the market and will leverage our experience in the CE Mark process to drive this forward."
IBI is a privately held commercial stage developer of biomedical-polymer medical device products. Q3 is an Irish based holding company with multiple operations in Germany focused on the development, manufacturing and distribution of minimally invasive devices for the treatment of patients with cardiology, peripheral vascular and non-vascular diseases.
Q3 Medical Devices Limited was formed by a global group of entrepreneurs, manufactures, distributors, industry doctors, and investors focused on the development and acquisition of medical device businesses with annual revenues between $1 million and $10 million. The acquisitions are targeted in areas that expand the groups manufacturing base and capabilities, growth of its distribution channels, and accelerating its products offering focused on the minimally invasive treatment of patients with cardiology, peripheral vascular and non-vascular diseases.
Qualimed, a wholly owned subsidiary of Q3 Medical, was founded in 1997 in Winsen, Germany near Hamburg, where it develops, manufactures, and sells implantable medical devices in the cardiology, peripheral vascular, and non-vascular areas. The innovations are focused on biodegradable products, drug-device combination technologies, catheter, and mechanical implant areas. Originally founded as an OEM, the company's products are now sold in more than 50 countries worldwide; the company and its development partners have obtained CE and FDA approvals for more than 70 different products including 3 different drug eluting stents.