02.12.14
On Feb. 10, the U.S. Food and Drug Administration (FDA) announced that Tandem Diabetes Care Inc. is recalling certain lost of the insulin cartridges used in its flagship product, the t:slim insulin pump.
According to the company, the affected cartridges may be at risk for leaking. The cause of the recall was identified during Tandem’s internal product testing, and has not been associated with any complaints or adverse events reported by customers.
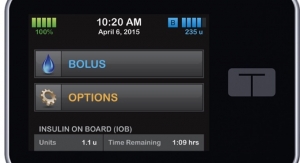
The t:slim insulin pump is a smart device that has the look an interface of a smartphone. Data from a multi center study presented by Tandem at the recent 7th Annual Advanced Technologies and Treatments for Diabetes International Conference in Vienna, Austria, showed that the t:slim required 27 percent shorter patient training times and resulted in 65 percent fewer errors than Medtronic’s Revel pump. The t:slim is a young device, only having earned FDA clearance in November 2011.
“Extensive human factors research went into the development of the t:slim Insulin Pump’s intuitive touch screen user interface,” said Kim Blickenstaff, president and CEO of Tandem Diabetes Care. “We believe the shorter training times and reduced use errors observed in this study are a testament to the importance of this kind of research in medical device development.”
Based in San Diego, Calif., Tandem Diabetes Care makes technology for the treatment of diabetes.
According to the company, the affected cartridges may be at risk for leaking. The cause of the recall was identified during Tandem’s internal product testing, and has not been associated with any complaints or adverse events reported by customers.
The t:slim insulin pump is a smart device that has the look an interface of a smartphone. Data from a multi center study presented by Tandem at the recent 7th Annual Advanced Technologies and Treatments for Diabetes International Conference in Vienna, Austria, showed that the t:slim required 27 percent shorter patient training times and resulted in 65 percent fewer errors than Medtronic’s Revel pump. The t:slim is a young device, only having earned FDA clearance in November 2011.
“Extensive human factors research went into the development of the t:slim Insulin Pump’s intuitive touch screen user interface,” said Kim Blickenstaff, president and CEO of Tandem Diabetes Care. “We believe the shorter training times and reduced use errors observed in this study are a testament to the importance of this kind of research in medical device development.”
Based in San Diego, Calif., Tandem Diabetes Care makes technology for the treatment of diabetes.