The study builds on positive results of the DIRECT I first-in-man study which evaluated the Svelte drug-eluting coronary stent IDS in 30 patients in New Zealand. The Svelte system met all study endpoints in DIRECT I while demonstrating in-stent neointimal volume obstruction of 2.7 percent at six months, a one-third to one-half reduction in volumetric obstruction observed in similar studies with other drug-eluting stents. Clinical outcomes reflected the angiographic findings, with no clinically driven major adverse cardiac events reported through 18 months.
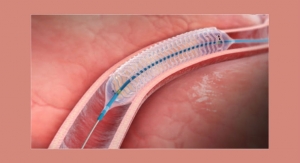
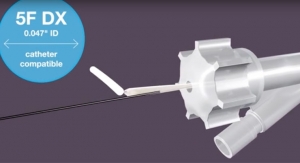
Providing the lowest crimped stent profile on the market, the fixed-wire Svelte drug-eluting stent IDS navigates the vasculature similar to a traditional guidewire and facilitates use of the trans-radial approach. The system combines a thin-strut cobalt chromium stent with the well-studied compound sirolimus (rapamycin) and a new class of bioabsorbable drug carrier composed of naturally-occurring amino acids which elicit a reduced inflammatory response compared with competitive technologies. Proprietary balloon control bands envelop the balloon shoulders to provide a smooth leading edge during delivery and uniform, controlled balloon growth during deployment to safely perform direct stenting as well as high-pressure post-dilatation.
“We are pleased to have completed this evaluation of the Svelte drug-eluting stent IDS following the outcomes of the DIRECT I study,” said Stefan Verheye, M.D., Ph.D., and co-director of the Antwerp Cardiovascular Institute at the Middelheim Hospital in Antwerp, Belgium. “As continued emphasis is placed on downsizing access sites and improving procedural efficiencies as well as clinical outcomes, this system presents an important option to interventional cardiologists.”
Primary endpoints of the DIRECT II study were target vessel failure and in-stent late loss. All patients are scheduled to receive six-month clinical and angiographic follow-up, with clinical follow-up for five years. A subset of patients will receive optical coherence tomography imaging at six months.
“We thank all investigators for their contributions to the DIRECT II study and look forward to bringing our technology to the wider interventional cardiology community,” said Jack Darby, President and CEO of Svelte Medical Systems. “Our IDS and RX systems are designed to facilitate use of the trans-radial and direct stenting approach to PCI, two segments of the market which are not currently served by dedicated technologies, yet have demonstrated significant clinical benefits and procedural efficiencies which are critical in today’s healthcare landscape.”
Headquartered in New Providence, N.J., Svelte Medical Systems is a privately-held developer of balloon expandable stents.