01.14.14
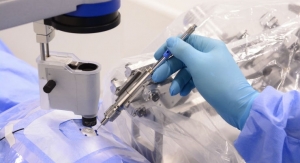
Lumenis Ltd., a developer of ophthalmic, surgical and aesthetic medical laser applications, has received U.S. Food and Drug Administration and Japanese Ministry of Health, Labour and Welfare clearance for its Array LaserLink pattern scanning device for multi-spot laser photocoagulation. The Array LaserLink expands the capabilities of the Lumenis laser system to deliver pattern scanning capabilities, aimed to provide greater predictability in retinal treatments and presumably less collateral damage to the retina. The Array LaserLink also is projected to save treatment time for clinicians and reduce patient discomfort with designed shorter pulse duration and sequential multi-spot delivery resulting in shorter laser sessions.
"The introduction of the Array LaserLink device in the United States and Japan demonstrates Lumenis' commitment to developing and introducing ophthalmic laser products and delivery devices that grow with our customers' needs," said Kfir Azoulay, vice president and general manager of the Ophthalmology Strategic Business Unit at Lumenis. "We believe Array will bring important benefits both to patients, clinicians and health care providers; and we look forward to working with the ophthalmology community to integrate our technology into clinical practice."
The Array LaserLink device adapts an examination slit lamp for use as a therapeutic laser delivery device. When attached to a compatible Lumenis photocoagulator the device offers a variety of features that expand the capabilities of the Lumenis laser system. Treatment parameters and micromanipulation are controllable from a remote touchpad designed to enhance the physician's workflow and efficiency. The touchpad allows the doctor to maintain visualization of the patient's retina through the slit lamp while changing treatment parameters such as spot size, spacing, number of spots in the pattern, pattern rotation and laser power. The Array's heads-up display shows confirmation of parameter changes, which are seen as text projected onto the retina.
The Array LaserLink device is indicated for various ophthalmic uses, including diabetic retinopathy, choroidal neovascularization, branch retinal vein occlusion, retinal tears and detachments. With the Array LaserLink device, compatible Lumenis lasers systems will be augmented with the following features:
Based in San Jose, Calif., Lumenis develops and commercializes energy-based technologies, including laser, intense pulsed light and radio-frequency.
"The introduction of the Array LaserLink device in the United States and Japan demonstrates Lumenis' commitment to developing and introducing ophthalmic laser products and delivery devices that grow with our customers' needs," said Kfir Azoulay, vice president and general manager of the Ophthalmology Strategic Business Unit at Lumenis. "We believe Array will bring important benefits both to patients, clinicians and health care providers; and we look forward to working with the ophthalmology community to integrate our technology into clinical practice."
The Array LaserLink device adapts an examination slit lamp for use as a therapeutic laser delivery device. When attached to a compatible Lumenis photocoagulator the device offers a variety of features that expand the capabilities of the Lumenis laser system. Treatment parameters and micromanipulation are controllable from a remote touchpad designed to enhance the physician's workflow and efficiency. The touchpad allows the doctor to maintain visualization of the patient's retina through the slit lamp while changing treatment parameters such as spot size, spacing, number of spots in the pattern, pattern rotation and laser power. The Array's heads-up display shows confirmation of parameter changes, which are seen as text projected onto the retina.
The Array LaserLink device is indicated for various ophthalmic uses, including diabetic retinopathy, choroidal neovascularization, branch retinal vein occlusion, retinal tears and detachments. With the Array LaserLink device, compatible Lumenis lasers systems will be augmented with the following features:
- --- Multi-spot patterns: Array provides a choice of laser patterns, permitting the doctor to select the application that is appropriate for the pathology and anatomy of the patient.
- --- Multi-wavelength compatibility: Array delivers the wavelengths of the laser system to which it is attached, allowing the doctor to select the preferred wavelength for the specific case and even change wavelength during the treatment if needed. Array can deliver 532nm and 577nm wavelengths in scanning mode and 532nm, 577nm and 659nm in single spot mode.
- --- Continuous Variable Spot Size: Array offers continuous variable spot size from 50 to 1000 micrometers, allowing the doctor to customize treatment for the disease state and tissue response.
- --- Variable distance between spots: The distance between spots in a pattern can be set up to three times the diameter of the spot, giving the doctor the flexibility to adjust the pattern to suit the retinal condition.
Based in San Jose, Calif., Lumenis develops and commercializes energy-based technologies, including laser, intense pulsed light and radio-frequency.