10.16.13
Smith & Nephew plc has inked a deal with Atlanta, Ga.-based Femasys Inc., a medical device developer of women's healthcare solutions, to become the sole provider of Femasys' FemVue Saline-Air Device in the United States.
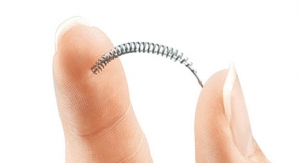
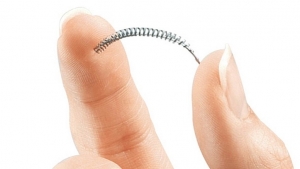
FemVue is a non-radiologic device that aids in the evaluation of a woman's fallopian tubes as part of a sono-hysterosalpingogram (sono HSG) procedure—a test that can help assess a woman's ability to become pregnant.
Femasys received 510(k) clearance from the U.S. Food and Drug Administration in April 2011.
The agreement was announced on Oct. 14 during a meeting of the International Federation of Fertility Societies and the American Society of Reproductive Medicine in Boston, Mass.
"We are pleased to offer gynecologists and their patients this innovative and minimally-invasive diagnostic test," said Mira Sahney, vice president and general manager of Smith & Nephew's Gynecology business. "FemVue complements our Truclear system and reinforces our commitment to offer industry-leading innovation that benefits healthcare providers and the patients they serve."
The FemVue device allows physicians to expand their office-based services with a procedure that is more convenient for patients and cost-effective for payers, according to Smith & Nephew. Rather than performing a more expensive hysterosalpingogram procedure in a hospital or outpatient radiology center setting, physicians are able to eliminate the use of X-rays by coupling saline and air with existing ultrasound technology when performing the sono-HSG procedure. The procedure can be performed in the outpatient setting of a gynecologist's office or fertility clinic. Results are generally available during the same office visit, officials said.
Truclear System is the primary technology of Smith & Nephew's uterine care business. As part of the diagnostic procedure during which FemVue is used, both the uterus and fallopian tubes are evaluated for abnormalities. If uterine polyps or fibroids are suspected, the patient could be a candidate for a follow-up Truclear procedure, during which the polyps or fibroids would be removed. Similar to tubal factors, polyps and fibroids are also a known cause of infertility.
"FemVue can be easily incorporated into any gynecological practice, allowing women to benefit from this much needed technology," said Kathy Lee-Sepsick, president and CEO of Femasys Inc. "We are excited to begin this partnership with Smith & Nephew and are confident FemVue's reach will be greatly expanded."
"There is anxiety and stress inherent in the fertility treatment process. FemVue helps ease some of that natural discomfort," said Chuck Miller, M.D., of the Advanced IVF Institute in Chicago, Ill. "My patients are more comfortable and the procedure is more efficient in the known environment of our office, rather than having to perform an HSG in the hospital."
Femasys is privately held.
With 2012 sales of more than $4 billion, London, United Kingdom-based Smith & Nephew’s primary markets are orthopedic reconstruction, advanced wound management, sports medicine and trauma.
FemVue is a non-radiologic device that aids in the evaluation of a woman's fallopian tubes as part of a sono-hysterosalpingogram (sono HSG) procedure—a test that can help assess a woman's ability to become pregnant.
Femasys received 510(k) clearance from the U.S. Food and Drug Administration in April 2011.
The agreement was announced on Oct. 14 during a meeting of the International Federation of Fertility Societies and the American Society of Reproductive Medicine in Boston, Mass.
"We are pleased to offer gynecologists and their patients this innovative and minimally-invasive diagnostic test," said Mira Sahney, vice president and general manager of Smith & Nephew's Gynecology business. "FemVue complements our Truclear system and reinforces our commitment to offer industry-leading innovation that benefits healthcare providers and the patients they serve."
The FemVue device allows physicians to expand their office-based services with a procedure that is more convenient for patients and cost-effective for payers, according to Smith & Nephew. Rather than performing a more expensive hysterosalpingogram procedure in a hospital or outpatient radiology center setting, physicians are able to eliminate the use of X-rays by coupling saline and air with existing ultrasound technology when performing the sono-HSG procedure. The procedure can be performed in the outpatient setting of a gynecologist's office or fertility clinic. Results are generally available during the same office visit, officials said.
Truclear System is the primary technology of Smith & Nephew's uterine care business. As part of the diagnostic procedure during which FemVue is used, both the uterus and fallopian tubes are evaluated for abnormalities. If uterine polyps or fibroids are suspected, the patient could be a candidate for a follow-up Truclear procedure, during which the polyps or fibroids would be removed. Similar to tubal factors, polyps and fibroids are also a known cause of infertility.
"FemVue can be easily incorporated into any gynecological practice, allowing women to benefit from this much needed technology," said Kathy Lee-Sepsick, president and CEO of Femasys Inc. "We are excited to begin this partnership with Smith & Nephew and are confident FemVue's reach will be greatly expanded."
"There is anxiety and stress inherent in the fertility treatment process. FemVue helps ease some of that natural discomfort," said Chuck Miller, M.D., of the Advanced IVF Institute in Chicago, Ill. "My patients are more comfortable and the procedure is more efficient in the known environment of our office, rather than having to perform an HSG in the hospital."
Femasys is privately held.
With 2012 sales of more than $4 billion, London, United Kingdom-based Smith & Nephew’s primary markets are orthopedic reconstruction, advanced wound management, sports medicine and trauma.