08.28.13
When it comes to fighting disease, one most often considers drug-based cures for mankind's worst ailments. But what about a device-based treatment for Alzheimer’s? Maybe it’s not so far-fetched. A Dallas, Texas-based company, Cerebain Biotech Corp., recently released photos of its first prototype of a medical device solution for the treatment of Alzheimer’s disease.
The company’s proposed technology centers around use of omentum, which is a pocket of fat and blood vessels lying over the intestines like a blanket. According to Cerebain’s website, omentum has biological agents that have regenerative properties that can have a “favorable effect” on the human brain.
In previous studies omental tissue was delivered from the abdomen to the surface of the brain, which is major neurosurgery on frail Alzheimer’s patients.
Cerebain’s technology will attempt to use omental tissue without major surgery.
“Our researchers observed that when gastrostomy tubes were placed in patients with dementia, there was an immediate improvement in baseline mental status generally in less than 24 hours. This discovery led us to believe that this procedure caused local stimulation of the omentum, which may have resulted in the systemic release of the neurotrophic factor or ‘brain booster,’” the company reports.
The firm’s patent describes various methods of harvesting this agent from the omentum and administering it to patients. According to the company, there is no risk of tissue rejection or formation of antibodies with this method.
The company plans to develop medical devices as well as an injectable form of this agent.
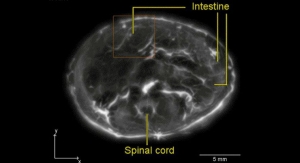
The photo highlights the electrodes that are intended to provide continuous omentum stimulation, which the company claims is a novel approach to the treatment for patients suffering from Alzheimer’s disease.
In October last year Cerebain announced that it had completed the first part of its initial phase of medical device development and that it had begun the next part of the process, which will involve research into the omentum as a basis for designing a fluid-extraction device. The company is working with Irvine, Calif.-based medical device product development company Sonos Models Inc.
"There have been recent public announcements about failed dementia and Alzheimer's disease drug trials by some of the larger companies,” said Gerald DeCiccio, Cerebain's president, last fall. “We firmly believe that if we are successful in implementing our approach it could be a game changer in the world of dementia and Alzheimer's disease."
Sonos' CEO, Carl Tenbrink, added: "We are very excited and vested after completing our initial investigation and exploration into Cerebain’s omentum project. This is really a leading-edge approach to treating dementia and Alzheimer’s disease using what seems to be an overlooked and poorly understood organ. We look forward to moving into the next phase of product development.”
Cerebain Biotech Corp. is a subsidiary of Discount Dental Materials Inc.
The company’s proposed technology centers around use of omentum, which is a pocket of fat and blood vessels lying over the intestines like a blanket. According to Cerebain’s website, omentum has biological agents that have regenerative properties that can have a “favorable effect” on the human brain.
In previous studies omental tissue was delivered from the abdomen to the surface of the brain, which is major neurosurgery on frail Alzheimer’s patients.
Cerebain’s technology will attempt to use omental tissue without major surgery.
“Our researchers observed that when gastrostomy tubes were placed in patients with dementia, there was an immediate improvement in baseline mental status generally in less than 24 hours. This discovery led us to believe that this procedure caused local stimulation of the omentum, which may have resulted in the systemic release of the neurotrophic factor or ‘brain booster,’” the company reports.
The firm’s patent describes various methods of harvesting this agent from the omentum and administering it to patients. According to the company, there is no risk of tissue rejection or formation of antibodies with this method.
The company plans to develop medical devices as well as an injectable form of this agent.
The photo highlights the electrodes that are intended to provide continuous omentum stimulation, which the company claims is a novel approach to the treatment for patients suffering from Alzheimer’s disease.
In October last year Cerebain announced that it had completed the first part of its initial phase of medical device development and that it had begun the next part of the process, which will involve research into the omentum as a basis for designing a fluid-extraction device. The company is working with Irvine, Calif.-based medical device product development company Sonos Models Inc.
"There have been recent public announcements about failed dementia and Alzheimer's disease drug trials by some of the larger companies,” said Gerald DeCiccio, Cerebain's president, last fall. “We firmly believe that if we are successful in implementing our approach it could be a game changer in the world of dementia and Alzheimer's disease."
Sonos' CEO, Carl Tenbrink, added: "We are very excited and vested after completing our initial investigation and exploration into Cerebain’s omentum project. This is really a leading-edge approach to treating dementia and Alzheimer’s disease using what seems to be an overlooked and poorly understood organ. We look forward to moving into the next phase of product development.”
Cerebain Biotech Corp. is a subsidiary of Discount Dental Materials Inc.