08.21.13
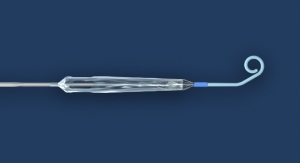
AngioDynamics Inc., an Albany, N.Y.-based maker of minimally invasive surgical devices for vascular access, surgery, peripheral vascular disease and oncology, has gained U.S. Food and Drug Administration (FDA) 510(k) clearance for its Bioflo port with Endexo technology (pictured left). The device is manufactured by Navilyst Medical Inc., a subsidiary of AngioDynamics based in Marlborough, Mass.
This marks the second U.S. clearance of a product line developed with Bioflo technology, and an initial launch is expected in the company’s third quarter of fiscal year 2014, which starts Jan. 1 2014.
Endexo technology is a permanent and non-eluting polymer designed to provide more resistance to the accumulation of platelets and thrombus. The port is designed to reduce the accumulation of catheter-related thrombus on and in the port catheter.
“We’re pleased to build on the success of our Bioflo PICC [peripherally inserted central catheter] by introducing the first port with a demonstrated reduction in thrombus accumulation,” said Joseph M. DeVivo, president and CEO of AngioDynamics. “The introduction of Bioflo ports is consistent with our plan to bring disruptive technologies into the vascular access space and reflects the strengthening of our Bioflo platform.”
Bioflo technology is hoped to decrease the accumulation of catheter-related thrombus without the incorporation of heparin, antibiotics, antimicrobials, or any other transient materials typically associated with coated or impregnated technologies. According to AngioDynamics, in-vitro blood loop model test results show that on average the Bioflo port catheter had 96 percent less thrombus accumulation on its surface compared to non-coated conventional port catheters (based on platelet count).
“We’re pleased to have received Bioflo port clearance ahead of our expected schedule and plan to launch Bioflo ports during the third fiscal quarter of 2014, bringing a financial impact to the second half of FY14.” said Chuck Greiner, vice president of AngioDynamics’ global vascular access franchise. “Given the strong performance we continue to see in Bioflo PICCs since our worldwide launch, we are excited to expand this premium technology into other segments. We will continue to grow our BioFlo vascular access portfolio by seeking FDA clearance for Bioflo dialysis catheters.”
This marks the second U.S. clearance of a product line developed with Bioflo technology, and an initial launch is expected in the company’s third quarter of fiscal year 2014, which starts Jan. 1 2014.
Endexo technology is a permanent and non-eluting polymer designed to provide more resistance to the accumulation of platelets and thrombus. The port is designed to reduce the accumulation of catheter-related thrombus on and in the port catheter.
“We’re pleased to build on the success of our Bioflo PICC [peripherally inserted central catheter] by introducing the first port with a demonstrated reduction in thrombus accumulation,” said Joseph M. DeVivo, president and CEO of AngioDynamics. “The introduction of Bioflo ports is consistent with our plan to bring disruptive technologies into the vascular access space and reflects the strengthening of our Bioflo platform.”
Bioflo technology is hoped to decrease the accumulation of catheter-related thrombus without the incorporation of heparin, antibiotics, antimicrobials, or any other transient materials typically associated with coated or impregnated technologies. According to AngioDynamics, in-vitro blood loop model test results show that on average the Bioflo port catheter had 96 percent less thrombus accumulation on its surface compared to non-coated conventional port catheters (based on platelet count).
“We’re pleased to have received Bioflo port clearance ahead of our expected schedule and plan to launch Bioflo ports during the third fiscal quarter of 2014, bringing a financial impact to the second half of FY14.” said Chuck Greiner, vice president of AngioDynamics’ global vascular access franchise. “Given the strong performance we continue to see in Bioflo PICCs since our worldwide launch, we are excited to expand this premium technology into other segments. We will continue to grow our BioFlo vascular access portfolio by seeking FDA clearance for Bioflo dialysis catheters.”