Niki Arrowsmith06.13.13
Bridgewater, N.J.-based Cordis Corporation (a division of Johnson & Johnson) has earned 510(k) clearance from the U.S. Food and Drug Administration for its new Adroit 6F guiding catheter.
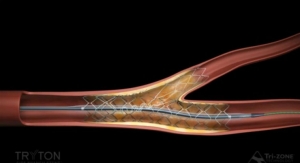
“The new Adroit guiding catheter allows physicians to have more options for treating a wide range of challenging cases,” said Celine Martin, worldwide president of Cordis Corporation. “It accommodates easier advancement of devices with its 0.072 inch diameter, the largest inner diameter of any guiding catheter in the United States, while providing better support and control at the same time. These features enhance treatment options for physicians, including the treatment of cases with complex lesions and those to be conducted using the radial approach.”
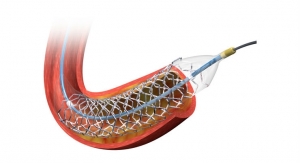
Adroit features a hybrid braid wire technology designed for better control and back up support. The catheter’s design incorporates a flexible distal segment and soft longer tip, allowing atraumatic placement and stability, claims Cordis. The Adroit guiding catheter comes in a range of shapes, including extra back-up shapes and radial specific shapes to support treatment needs of various coronary anatomies.
“Our vision at Cordis is to transform cardiovascular care, a field in which incredible progress has been made over the last decade, but one in which much more remains to be done,” added Martin. “We are excited for the launch of our new Adroit guiding catheter as it complements our guide catheter portfolio with its technological and design advances. We will continue to deliver innovative solutions that save lives and improve the quality of life for people around the world.”
Cordis focuses on interventional vascular technology.
“The new Adroit guiding catheter allows physicians to have more options for treating a wide range of challenging cases,” said Celine Martin, worldwide president of Cordis Corporation. “It accommodates easier advancement of devices with its 0.072 inch diameter, the largest inner diameter of any guiding catheter in the United States, while providing better support and control at the same time. These features enhance treatment options for physicians, including the treatment of cases with complex lesions and those to be conducted using the radial approach.”
Adroit features a hybrid braid wire technology designed for better control and back up support. The catheter’s design incorporates a flexible distal segment and soft longer tip, allowing atraumatic placement and stability, claims Cordis. The Adroit guiding catheter comes in a range of shapes, including extra back-up shapes and radial specific shapes to support treatment needs of various coronary anatomies.
“Our vision at Cordis is to transform cardiovascular care, a field in which incredible progress has been made over the last decade, but one in which much more remains to be done,” added Martin. “We are excited for the launch of our new Adroit guiding catheter as it complements our guide catheter portfolio with its technological and design advances. We will continue to deliver innovative solutions that save lives and improve the quality of life for people around the world.”
Cordis focuses on interventional vascular technology.