YOU SEARCHED BY...
SEARCH BY...
Outsourcing Directory
Country
- Austria(1)
- Belgium(1)
- Canada(13)
- China(12)
- Costa Rica(1)
- Denmark(1)
- France(2)
- Germany(13)
- Hong Kong(3)
- India(9)
- Ireland(8)
- Israel(4)
- Italia (4)
- Japan(2)
- Lithuania(1)
- Malaysia(1)
- Mexico(2)
- New Zealand(1)
- Russia(1)
- Singapore(5)
- Spain(1)
- Switzerland(6)
- Taiwan(15)
- Thailand(1)
- The Dominican Republic(1)
- The Netherlands(2)
- United Kingdom(18)
- United States(953)
Outsourcing Directory
Medical Device Manufacturers Association (MDMA)
Company Headquarters
1333 H Street Northwest, Suite 400
Washington, DC 20005
United States
Outsourcing Directory
In-Vitro Diagnostic Categories
Related Content
-
Tubing & Extrusion

Extrapolating Extrusion
A roundtable discussion of the myriad trends and challenges affecting extruders for the medical device industry.Sam Brusco, Associate Editor 10.16.18
-

Carbon Releases New MPU 100 Material for Medical Applications
MPU 100 offers a unique combination of mechanical strength, biocompatibility, and sterilizability.Business Wire 10.10.18
-

ivWatch Receives CE Mark for the ivWatch Model 400
Recent regulatory approvals and global certification pave the way for international distribution.Business Wire 10.04.18
-
D2 Consulting Appoints Senior Director of Federal Markets
New executive brings more than 15 years’ experience overseeing federal procurements.Business Wire 09.17.18
-
Taking Stock of MDR Compliance
Just over a year in since the new regulations were rolled out and companies are lagging well behind in meeting them.Peter Rose, Managing Director—Europe, Maetrics 08.23.18
-
Testing

Study: Medical Device Managers Rely on Physicians to Screen Out Defects Rather Than Issue Recalls
The FDA expressed interest in using this research to improve how they oversee medical device product quality.Indiana University 08.22.18
-

Industry Applauds House Passage of Device Tax Repeal Bill
Moves onto the Senate for consideration.Sean Fenske, Editor-in-Chief 07.25.18
-
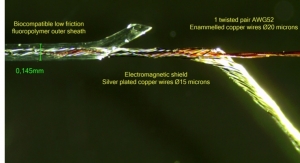
Nexans’ NEWSENSE Microcables Open Up New Dimensions in Invasive Surgery
Company has developed a micro-extrusion process that creates the thinnest possible insulating jacket for MI medical cables.Nexans 05.11.18
-
Diagnostics
IVDR Implementation—The Clock Is Ticking
Manufacturers should be preparing now to ensure compliance by the 2022 deadline.Brian Moan, Director of Solutions Delivery Europe, and Norm Rabin, Director of Solutions Delivery—Maetrics 04.27.18











