YOU SEARCHED BY...
SEARCH BY...
States
- California(1)
- Colorado(1)
- District of Columbia(1)
- Maryland(1)
- Minnesota(2)
- Ohio(1)
- Texas(1)
- Virginia(1)
Outsourcing Directory
F2 Labs
Company Headquarters
26501 Ridge Road
Damascus, MD 20872
United States
Outsourcing Directory
In-Vitro Diagnostic Categories
Related Content
-
Cardiovascular | Diagnostics

-
Materials
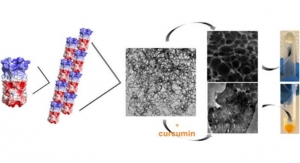
Researchers Develop Hydrogel Capable of Providing Sustained Medication Release
Delivers drugs at the cellular level and engineers tissue.NYU Tandon 09.18.19











