Sam Brusco, Associate Editor07.29.22
DyAnsys has earned U.S. Food and Drug Administration (FDA) clearance for Primary Relief, a percutaneous electrical neurostimulation device (PENS) to treat pain after a Caesarean section (C-section).
It can be used for up to three days to relieve post-operative pain after the C-section delivery.
"This device has been shown to make a difference for patients, effectively relieving pain without reliance on opioids or other analgesics. This is a significant advancement in providing options to women," DyAnsys chief executive Srini Nageshwar told the press. "We look forward to connecting with physicians and hospitals to providing this alternative to their patients."
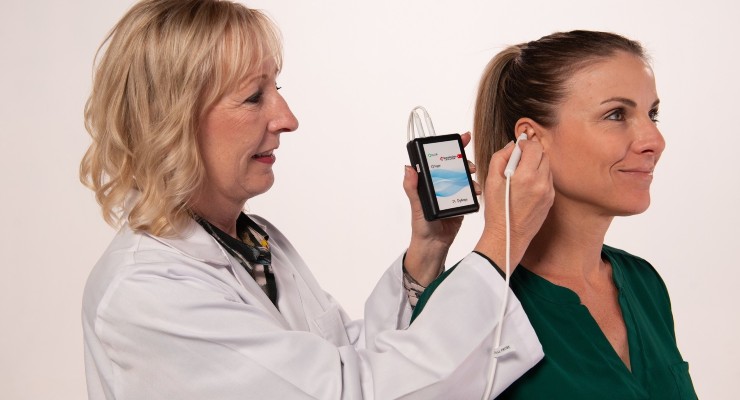
Wearable and battery operated, Primary Relief administers periodic low level electrical pulses to the ear over 72 hours. These are delivered to the cranial nerves on the ear via wire assembly and stimulation needles.
A 44-patient trial of C-section patients should the Primary Relief device reduced pain score more quickly than standard analgesics. No further opioids were needed, and no complications or adverse events were observed in the study.
It can be used for up to three days to relieve post-operative pain after the C-section delivery.
"This device has been shown to make a difference for patients, effectively relieving pain without reliance on opioids or other analgesics. This is a significant advancement in providing options to women," DyAnsys chief executive Srini Nageshwar told the press. "We look forward to connecting with physicians and hospitals to providing this alternative to their patients."
Wearable and battery operated, Primary Relief administers periodic low level electrical pulses to the ear over 72 hours. These are delivered to the cranial nerves on the ear via wire assembly and stimulation needles.
A 44-patient trial of C-section patients should the Primary Relief device reduced pain score more quickly than standard analgesics. No further opioids were needed, and no complications or adverse events were observed in the study.