Prasad Akella, Founder/CEO, Drishti05.03.21
In an industry like medical device manufacturing, where the stakes of errors and defects are patient health and mortality, quality is everyone’s first language. And with McKinsey finding manufacturers invest (on average) 7 to 9 percent of their sales dollars in quality control and assurance measures, it’s proof companies are hyper-focused on knowing and following current good manufacturing practices (cGMP).
But there’s a trade-off with that focus. If you’re following the same practices as the rest of the industry, what differentiates you from your competitors from a manufacturing and quality perspective?
Medical device manufacturers that make cGMP a critical rung on the ladder to extraordinary manufacturing practices are the ones who stand out in the marketplace. They recoup some of that investment because they find more efficient ways to ensure quality practices. They accelerate productivity and make their kaizen events more impactful. Further, they focus on training practices that drive standardized work adherence, improve employee satisfaction, and reach a global workforce.
In short, the medical device manufacturers who differentiate themselves in the market take manufacturing well beyond compliance to excellence. Instead of settling for cGMP, they strive to become extraordinary (cEMP, if you will).
Two ways to accomplish extraordinary manufacturing practices are through video and analytics.
Quality
Quality is the one facet of medical device manufacturing that cannot be compromised. Few manufacturers are as susceptible to risk as medical device makers. Not only is the human cost of a faulty or errant device—not to mention packaging errors or nonconformance—high, but the regulatory fines and penalties (including costly shutdowns) for quality issues are hefty. For example, in 2018, the FDA shut down Keystone Laboratories Inc. of Memphis, Tenn., for manufacturing practices that were in violation of cGMP requirements.1
Obviously, only looking at quality from the standpoint of avoiding fines is short-sighted. Infusing quality into every stage of the manufacturing process—and ensuring everyone on the team lives and breathes quality, not just those with quality in their titles—benefits the bottom line. Empowering them with insightful data as they are going about their job further impacts the bottom line. For example, if you can reduce time spent on root cause analysis and corrective action, minimize scrap, and reduce rework, you’ll have happier customers, happier employees, and bigger contracts. That’s where standardized work comes into play.
Standardized Work
Best created by the people who know the process most intimately—the line associate, the team leader, and the process engineer—standardized work represents the best possible way known at that point in time to build a quality product most efficiently. That’s why manufacturers—particularly in the medical device space—are maniacal about ensuring adherence happens. If the line associates are following standardized work, they are not producing defects. When changes are made, the baseline is clear: standardized work. If quality or productivity do not improve, one reverts to the last standardized work on record. If it does improve, we update the standardized work definition.
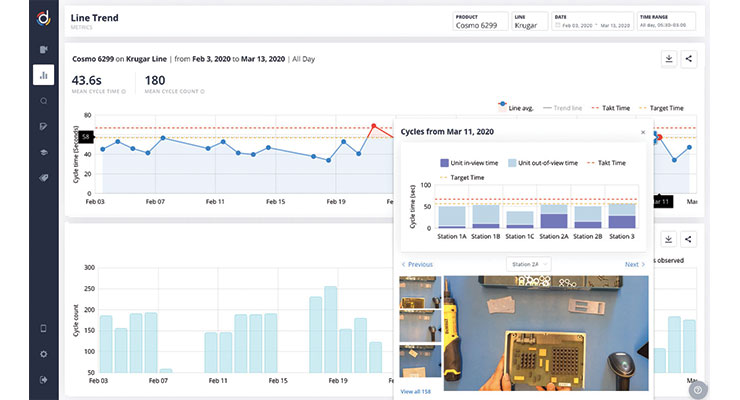
It’s really hard, however—maybe even impossible—to gauge standardized work continuously on every station on the line without help. New manufacturing practices like video, both in real time and stored, then processed and assigned metadata, are used to highlight deviations. The quality engineer can then prioritize and address those deviations.
How is this better than one-off visual inspection by the engineer? Because, while a human can only watch a handful of units being produced, AI can look at hundreds of thousands of units, then flag the ones that need attention.
Relying Less on Manual Visual Inspection
Traditionally, improving quality on manual assembly lines has required spot-checking the work of line associates, then setting up quality inspection stations at the end of the assembly process. But there’s a finite amount of quality inspection stations you can add before the effectiveness wanes (imagine having six different line associates inspecting a finished stent—yikes, the wasted expense!). The key is to bake quality measures deeper into the assembly process versus hoping to catch any issues at the end of the line.
One highly underrated and extremely effective tool for accomplishing that is video. Nothing is a stronger arbiter of truth than video footage of what’s happening in real time, what happened just a few minutes ago, or what happened last month during the third shift. Further, it’s this source of truth—video traceability—that provides significant savings.
Think about it: If you have video evidence in real time, you can prevent a standardized work deviation that caused a defect, or flag an issue for review before it ever hits the inspection stage. You can all but eliminate inspection from the back end of your processes. In addition, if you have past footage that shows the cause of a defect (e.g., a specific line associate who missed a consistent step and needs additional training), you can limit rework or scrap to those specific units. Also, the team can learn from these observations while the standardized work can be updated so the line improves.
That’s a much bigger risk-reducing scenario than the typical response to an FDA Form 3500A, which requires tedious root cause analysis (RCA) and corrective and preventive action (CAPA) that are time consuming, imperfect, require a lot of resources, and often end with your best guess that the true root cause of the defect was discovered and fixed.
Productivity Matters
There’s a misconception that many medical device manufacturers have: Productivity can only be improved at the expense of quality. Unfortunately, this faulty assumption too often resigns manufacturers to settle for tiny productivity gains. But it doesn’t have to be a trade-off; if you elevate your manufacturing practices beyond cGMP, you can use new technology like video and AI to drive both productivity and quality. Here’s how.
The key to improving productivity without sacrificing quality is (you guessed it): standardized work. That’s why most medical device manufacturers take painstaking measures to ensure standardized work is universally followed. But here’s a thought that will make you shudder: Even with your best efforts, you’re missing things on the line that make you less productive than you have the potential to be. You simply can’t be everywhere at once.
That’s where video comes back into play. It’s much more feasible to watch multiple live video streams than it is to walk from line to line and hope to catch standardized work deviations. But even that is hard. So if that video can be converted to analytics, you can quickly spot lines or stations that need more of your attention, and focus your gemba efforts for the day on that locale.
Shorter, More Impactful Kaizens Are Better for Business
Another key tool for productivity improvements is the kaizen, which is the mechanism by which medical device manufacturers make continuous improvement happen on the line. If you’re like most manufacturers following cGMP, a typical kaizen event looks like this:
Second, you can implement your kaizen recommendations while keeping another line unchanged, and continuously gather footage and data on both. So instead of guessing about the results of your kaizen’s success, you can control for any other variations and truly gauge the success of your recommendations.
Now, if you really want to up your game, why not give your line associates—the folks who really know your process—the freedom to make changes? Using the data gathered, one can quickly do an A/B comparison to determine if the change worked. If it did, keep it. Publish it as the new standardized work. If not, go back to your previous standard.
When you elevate your manufacturing practices beyond compliance, you can achieve greater levels of productivity than ever before. Better still, it doesn’t have to come at the cost of quality.
Line Associate Training Doesn’t Have To Be Boring
According to the Bureau of Labor Statistics, the average annual churn in manufacturing hovers around 30 percent. On top of that, the average onboarding program for a new line associate is four weeks. So in any given week, at least one of your line associates is likely to be brand new.
How do you quickly and effectively train line associates, decrease churn, and scale that training program across factories in multiple countries with varying languages and cultures? The answer is changing the way you train. By elevating your training game above cGMP and trying something extraordinary.
As of 2016, millennials are the dominant generation in the workforce. With the oldest millennials only turning 40 this year, they’ll be the dominant group for many years to come. Further, millennials like video. They watch videos in their free time on YouTube and other social media channels. They learn from how-to and DIY videos, and many of them take video classes online. That’s why using video for training purposes can be a much more effective method for training new employees, or implementing spot training with existing employees, than reading a handbook or listening to a trainer.
Think about it this way: When Tom Brady throws a rare interception, he doesn’t flip through the playbook to see what went wrong. He picks up the Microsoft Surface tablet on the sideline and watches the replay.
Leading manufacturers are bringing that same level of extraordinary training to their factories. They take video from their manual assembly lines, tag the footage that shows best practices, and play those clips for their trainees.
Another bonus of video: consistency. If you have Alissa training the new first-shift employees and Manuel training the second shift and Jerome training the third shift, chances are high one of your trainers may deviate in his or her instructions. Maybe Alissa didn’t have enough coffee before work. Maybe Jerome’s son was feeling sick when he left the house and he’s distracted. Video keeps the training consistent every single time, no matter what external factors may be influencing the trainers and line associates.
Video and Analytics Are Culturally and Linguistically Agnostic
Video can help you scale. Think about what happens when you share standardized work instructions from your plant outside Knoxville, Tenn., with your sister plant in Ensenada, Mexico, or Dong Nai, Vietnam. Now you’re relying on local translators to nail the distribution of your work instructions, understanding the cultural nuances and communicating clearly about something they don’t completely understand.
However, if you pair those work instructions, or even replace them with video, suddenly the room for error is much smaller. Video is culturally and linguistically agnostic, and it’s much more difficult for a video to get lost in translation than it is the written word. Further, analytics derived from video feeds don’t change regardless of whether a line associate wears culturally diverse clothing, hairstyles, skin colors, and the like.
Empowering Line Associates
There’s one more element to having video and analytics available to the line associates themselves. In the true embodiment of the Toyota Production System (TPS), the line associate who has access to information becomes empowered, and therefore, one of your most valuable assets.
In “The Toyota Way” by Dr. Jeffrey Liker, Alex Warren, former executive vice president of Toyota Motor Corporation in Kentucky, said, “In the case of humans, we give them the power to push buttons or pull cords—called ‘andon cords’—which can bring our entire assembly line to a halt. Every team member has the responsibility to stop the line every time they see something that is out of standard. That’s how we put the responsibility for quality in the hands of our team members. They feel the responsibility—they feel the power. They know they count.”
Now translate that to a line of medical devices, like catheters or infusion pumps, where an error can be fatal to the patient and cripplingly damaging to your business. If your line associates have been properly onboarded using video, and effectively spot-trained with video and analytics, they’ll be equipped to spot anomalies in real time and empowered to “pull the andon cord” to stop production.
Imagine how your line associates would feel about being a part of the problem-solving team, about being empowered to make key decisions on the line. It’s a potential game-changing opportunity to elevate your brand above the competition.
Conclusion
When it comes to medical device manufacturing, more data—provided at the right time, to the right person—can have a serious positive impact on quality, productivity, and training. Using video to capture footage from manual assembly lines is an effective way to secure those analytics and insights, while backing each data point with video-based evidence.
Particularly in a segment of the market like medical device manufacturing, which is going to see a lot of turnover soon as older workers retire and millennials become the dominant players in the market, manufacturers need to rethink training delivery and effectiveness and find new ways to drive productivity and ensure quality adherence.
New technologies like artificial intelligence can help manufacturers who want to strive beyond compliance by providing video and analytics from manual assembly operations. It’s the manufacturer who sees the value in moving beyond compliance and setting extraordinary standards for manufacturing excellence who will rise above the competition.
References
Dr. Prasad Akella, founder and CEO of Drishti, is creating his third massive market category that uses technology to extend human capabilities. In the 1990s, Dr. Akella led the General Motors team that built the world’s first collaborative robots (“cobots,” projected to be a $12 billion market by 2025). In the early 2000s, as cofounder of the social networking pioneer Spoke, he envisioned and helped build the first massive social graph—a category now worth trillions of dollars. Today, at Drishti, he is working to combine the cognition of AI with the flexibility of humans in factories in the form of AI-powered production.
But there’s a trade-off with that focus. If you’re following the same practices as the rest of the industry, what differentiates you from your competitors from a manufacturing and quality perspective?
Medical device manufacturers that make cGMP a critical rung on the ladder to extraordinary manufacturing practices are the ones who stand out in the marketplace. They recoup some of that investment because they find more efficient ways to ensure quality practices. They accelerate productivity and make their kaizen events more impactful. Further, they focus on training practices that drive standardized work adherence, improve employee satisfaction, and reach a global workforce.
In short, the medical device manufacturers who differentiate themselves in the market take manufacturing well beyond compliance to excellence. Instead of settling for cGMP, they strive to become extraordinary (cEMP, if you will).
Two ways to accomplish extraordinary manufacturing practices are through video and analytics.
Quality
Quality is the one facet of medical device manufacturing that cannot be compromised. Few manufacturers are as susceptible to risk as medical device makers. Not only is the human cost of a faulty or errant device—not to mention packaging errors or nonconformance—high, but the regulatory fines and penalties (including costly shutdowns) for quality issues are hefty. For example, in 2018, the FDA shut down Keystone Laboratories Inc. of Memphis, Tenn., for manufacturing practices that were in violation of cGMP requirements.1
Obviously, only looking at quality from the standpoint of avoiding fines is short-sighted. Infusing quality into every stage of the manufacturing process—and ensuring everyone on the team lives and breathes quality, not just those with quality in their titles—benefits the bottom line. Empowering them with insightful data as they are going about their job further impacts the bottom line. For example, if you can reduce time spent on root cause analysis and corrective action, minimize scrap, and reduce rework, you’ll have happier customers, happier employees, and bigger contracts. That’s where standardized work comes into play.
Standardized Work
Best created by the people who know the process most intimately—the line associate, the team leader, and the process engineer—standardized work represents the best possible way known at that point in time to build a quality product most efficiently. That’s why manufacturers—particularly in the medical device space—are maniacal about ensuring adherence happens. If the line associates are following standardized work, they are not producing defects. When changes are made, the baseline is clear: standardized work. If quality or productivity do not improve, one reverts to the last standardized work on record. If it does improve, we update the standardized work definition.
It’s really hard, however—maybe even impossible—to gauge standardized work continuously on every station on the line without help. New manufacturing practices like video, both in real time and stored, then processed and assigned metadata, are used to highlight deviations. The quality engineer can then prioritize and address those deviations.
How is this better than one-off visual inspection by the engineer? Because, while a human can only watch a handful of units being produced, AI can look at hundreds of thousands of units, then flag the ones that need attention.
Relying Less on Manual Visual Inspection
Traditionally, improving quality on manual assembly lines has required spot-checking the work of line associates, then setting up quality inspection stations at the end of the assembly process. But there’s a finite amount of quality inspection stations you can add before the effectiveness wanes (imagine having six different line associates inspecting a finished stent—yikes, the wasted expense!). The key is to bake quality measures deeper into the assembly process versus hoping to catch any issues at the end of the line.
One highly underrated and extremely effective tool for accomplishing that is video. Nothing is a stronger arbiter of truth than video footage of what’s happening in real time, what happened just a few minutes ago, or what happened last month during the third shift. Further, it’s this source of truth—video traceability—that provides significant savings.
Think about it: If you have video evidence in real time, you can prevent a standardized work deviation that caused a defect, or flag an issue for review before it ever hits the inspection stage. You can all but eliminate inspection from the back end of your processes. In addition, if you have past footage that shows the cause of a defect (e.g., a specific line associate who missed a consistent step and needs additional training), you can limit rework or scrap to those specific units. Also, the team can learn from these observations while the standardized work can be updated so the line improves.
That’s a much bigger risk-reducing scenario than the typical response to an FDA Form 3500A, which requires tedious root cause analysis (RCA) and corrective and preventive action (CAPA) that are time consuming, imperfect, require a lot of resources, and often end with your best guess that the true root cause of the defect was discovered and fixed.
Productivity Matters
There’s a misconception that many medical device manufacturers have: Productivity can only be improved at the expense of quality. Unfortunately, this faulty assumption too often resigns manufacturers to settle for tiny productivity gains. But it doesn’t have to be a trade-off; if you elevate your manufacturing practices beyond cGMP, you can use new technology like video and AI to drive both productivity and quality. Here’s how.
The key to improving productivity without sacrificing quality is (you guessed it): standardized work. That’s why most medical device manufacturers take painstaking measures to ensure standardized work is universally followed. But here’s a thought that will make you shudder: Even with your best efforts, you’re missing things on the line that make you less productive than you have the potential to be. You simply can’t be everywhere at once.
That’s where video comes back into play. It’s much more feasible to watch multiple live video streams than it is to walk from line to line and hope to catch standardized work deviations. But even that is hard. So if that video can be converted to analytics, you can quickly spot lines or stations that need more of your attention, and focus your gemba efforts for the day on that locale.
Shorter, More Impactful Kaizens Are Better for Business
Another key tool for productivity improvements is the kaizen, which is the mechanism by which medical device manufacturers make continuous improvement happen on the line. If you’re like most manufacturers following cGMP, a typical kaizen event looks like this:
- Weeks 1 and 2: A team of industrial engineers spends the bulk of its days gathering data on the focus lines and stations. The data is gathered by hand, usually using a stopwatch or smartphone timer. Chances are, your IEs have missed a few cycles (or a few dozen).
- Week 3: Kaizen event takes place. Your team spends several days creating work boards, mapping cycle times, calculating takt time, mapping the value stream, and accounting for, or potentially throwing out, outlier cycle data.
- Weeks 4 and 5: Your team implements the recommendations from the kaizen and continues to conduct time studies in order to compare before and after on the line. You attribute changes in the data to the kaizen, and not to a coincidental modification in the line associates’ moods or the materials flow process or any number of other variables.
Second, you can implement your kaizen recommendations while keeping another line unchanged, and continuously gather footage and data on both. So instead of guessing about the results of your kaizen’s success, you can control for any other variations and truly gauge the success of your recommendations.
Now, if you really want to up your game, why not give your line associates—the folks who really know your process—the freedom to make changes? Using the data gathered, one can quickly do an A/B comparison to determine if the change worked. If it did, keep it. Publish it as the new standardized work. If not, go back to your previous standard.
When you elevate your manufacturing practices beyond compliance, you can achieve greater levels of productivity than ever before. Better still, it doesn’t have to come at the cost of quality.
Line Associate Training Doesn’t Have To Be Boring
According to the Bureau of Labor Statistics, the average annual churn in manufacturing hovers around 30 percent. On top of that, the average onboarding program for a new line associate is four weeks. So in any given week, at least one of your line associates is likely to be brand new.
How do you quickly and effectively train line associates, decrease churn, and scale that training program across factories in multiple countries with varying languages and cultures? The answer is changing the way you train. By elevating your training game above cGMP and trying something extraordinary.
As of 2016, millennials are the dominant generation in the workforce. With the oldest millennials only turning 40 this year, they’ll be the dominant group for many years to come. Further, millennials like video. They watch videos in their free time on YouTube and other social media channels. They learn from how-to and DIY videos, and many of them take video classes online. That’s why using video for training purposes can be a much more effective method for training new employees, or implementing spot training with existing employees, than reading a handbook or listening to a trainer.
Think about it this way: When Tom Brady throws a rare interception, he doesn’t flip through the playbook to see what went wrong. He picks up the Microsoft Surface tablet on the sideline and watches the replay.
Leading manufacturers are bringing that same level of extraordinary training to their factories. They take video from their manual assembly lines, tag the footage that shows best practices, and play those clips for their trainees.
Another bonus of video: consistency. If you have Alissa training the new first-shift employees and Manuel training the second shift and Jerome training the third shift, chances are high one of your trainers may deviate in his or her instructions. Maybe Alissa didn’t have enough coffee before work. Maybe Jerome’s son was feeling sick when he left the house and he’s distracted. Video keeps the training consistent every single time, no matter what external factors may be influencing the trainers and line associates.
Video and Analytics Are Culturally and Linguistically Agnostic
Video can help you scale. Think about what happens when you share standardized work instructions from your plant outside Knoxville, Tenn., with your sister plant in Ensenada, Mexico, or Dong Nai, Vietnam. Now you’re relying on local translators to nail the distribution of your work instructions, understanding the cultural nuances and communicating clearly about something they don’t completely understand.
However, if you pair those work instructions, or even replace them with video, suddenly the room for error is much smaller. Video is culturally and linguistically agnostic, and it’s much more difficult for a video to get lost in translation than it is the written word. Further, analytics derived from video feeds don’t change regardless of whether a line associate wears culturally diverse clothing, hairstyles, skin colors, and the like.
Empowering Line Associates
There’s one more element to having video and analytics available to the line associates themselves. In the true embodiment of the Toyota Production System (TPS), the line associate who has access to information becomes empowered, and therefore, one of your most valuable assets.
In “The Toyota Way” by Dr. Jeffrey Liker, Alex Warren, former executive vice president of Toyota Motor Corporation in Kentucky, said, “In the case of humans, we give them the power to push buttons or pull cords—called ‘andon cords’—which can bring our entire assembly line to a halt. Every team member has the responsibility to stop the line every time they see something that is out of standard. That’s how we put the responsibility for quality in the hands of our team members. They feel the responsibility—they feel the power. They know they count.”
Now translate that to a line of medical devices, like catheters or infusion pumps, where an error can be fatal to the patient and cripplingly damaging to your business. If your line associates have been properly onboarded using video, and effectively spot-trained with video and analytics, they’ll be equipped to spot anomalies in real time and empowered to “pull the andon cord” to stop production.
Imagine how your line associates would feel about being a part of the problem-solving team, about being empowered to make key decisions on the line. It’s a potential game-changing opportunity to elevate your brand above the competition.
Conclusion
When it comes to medical device manufacturing, more data—provided at the right time, to the right person—can have a serious positive impact on quality, productivity, and training. Using video to capture footage from manual assembly lines is an effective way to secure those analytics and insights, while backing each data point with video-based evidence.
Particularly in a segment of the market like medical device manufacturing, which is going to see a lot of turnover soon as older workers retire and millennials become the dominant players in the market, manufacturers need to rethink training delivery and effectiveness and find new ways to drive productivity and ensure quality adherence.
New technologies like artificial intelligence can help manufacturers who want to strive beyond compliance by providing video and analytics from manual assembly operations. It’s the manufacturer who sees the value in moving beyond compliance and setting extraordinary standards for manufacturing excellence who will rise above the competition.
References
Dr. Prasad Akella, founder and CEO of Drishti, is creating his third massive market category that uses technology to extend human capabilities. In the 1990s, Dr. Akella led the General Motors team that built the world’s first collaborative robots (“cobots,” projected to be a $12 billion market by 2025). In the early 2000s, as cofounder of the social networking pioneer Spoke, he envisioned and helped build the first massive social graph—a category now worth trillions of dollars. Today, at Drishti, he is working to combine the cognition of AI with the flexibility of humans in factories in the form of AI-powered production.