Kevin Holland, Chief Executive, Invest Northern Ireland02.03.21
The re-evaluation of our global supply chains has been long overdue.
Back in 2011, the FDA published Reliance on Foreign Sourcing in the Healthcare and Public Sector, a report that highlighted many of the underlying risks lurking within the medical device and pharmaceutical industries. They discovered that for over 43 percent of critical components, materials, and finished products intended for incorporation into medical devices and surgical equipment, no alternate source of those materials—in or outside the United States—was available. The report also found that while the most common medical manufacturing components were provided by suppliers in 32 different companies, the top single-source locations were all located in Asia.
Cut to 2021; it’s now 10 years later, and over the last several months, the medical manufacturing industry has had to rapidly respond to a global health crisis that has upended not just life sciences and healthcare, but every other industry as well.
On the one hand, it has been impressive to witness the sector’s heroic response to this once-in-a-lifetime challenge. Companies have quickly adapted, developing new technologies and producing supply to meet new or increased demand. From Northern Ireland, I’ve watched our highly flexible medtech sector quickly pivot to develop COVID-19 related PPE, medical equipment, testing, and diagnostics.
On the other hand, it didn’t take long for COVID-19 to shine a spotlight on long-known, inherent supply chain weaknesses. In March 2020, the Coronavirus Aid, Relief, and Economic Security (CARES) Act provided the FDA—for the first time—with “new authority to help prevent or mitigate negative public health impacts of medical device supply chain disruptions.”
Given the findings of the previously mentioned 2011 report alongside what the FDA classifies as “unprecedented increased demand for some medical devices, as well as significant disruptions to global medical device manufacturing and supply chain operations,” it’s clear that effective global supply chains in this sector are more critical than ever.
From discussions with global manufacturers, it is evident that a profound and lasting change to how medical devices and pharmaceuticals are sourced and distributed is already underway. These companies are actively investigating how near-shoring and dual-source pipelines can protect the on-time manufacturing and distribution of critical devices and supplies.
This shift to diversified sourcing is poised to happen much quicker for medical product manufacturing than it will for pharma. The FDA found nearly half of medical devices/surgical equipment sourcing contracts are only maintained for three years or less. Flexibility is key due to ever-changing innovations and upgrades to medical products. By contrast, pharmaceutical companies operate with a higher level of regulation and the composition of medicines do not change, so supplier contracts are typically maintained for a much longer time.
So in the complex calculus of identifying alternative source locations outside the U.S., what are the top considerations? The FDA identified six factors in its research: cost, compliance (with regulatory requirements), product quality, product availability, IP protection, and market access.
It is in this context Northern Ireland presents several unique value propositions to U.S. medical product companies evaluating second-source manufacturing locations. One of them is an entirely new post-Brexit development, and another has long existed: a truly connected life sciences ecosystem.
A New EU-UK Trade Deal
Ever since the Brexit referendum was held in 2016, the world has been speculating about the global ramifications of the U.K. leaving the EU. In particular, there have been questions about Northern Ireland, given the region’s geopolitical circumstances.
Northern Ireland is unique because it’s part of the United Kingdom but geographically it’s located across the Irish Sea, where it shares a land border with the Republic of Ireland, which is part of the European Union.
Last year, special consideration was made to address this geography’s impact on post-Brexit trade through a pre-deal agreement called the Northern Ireland Protocol. The two aims of the agreement were 1) to ensure Northern Ireland businesses could continue trading with the rest of the U.K. without interruption, and 2) to make sure any agreed trade deal did not result in a hard border on the island of Ireland.
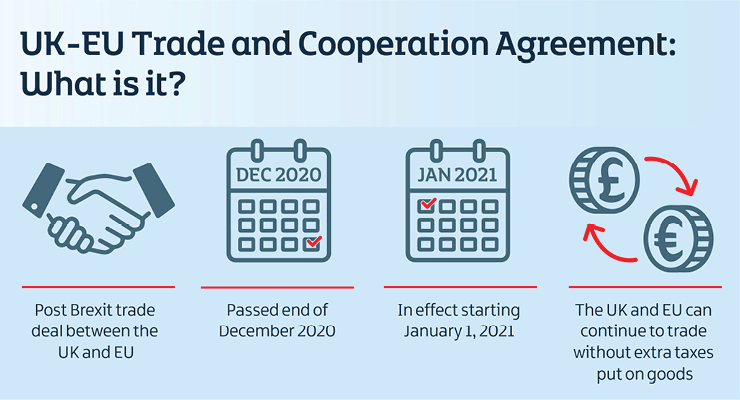
Though the U.K. (and Northern Ireland with it) formally left the EU in January 2020, a transition period was granted through the end of December 2020. During those 12 months, the U.K. and EU worked to negotiate a trade deal against the backdrop of a global pandemic. After more than four years since the initial referendum vote, the two parties agreed on the long-sought deal on Christmas Eve 2020, just a week before the year-end deadline. The deal was officially passed just before the ball dropped on 2021.
Coming in at around 2,000 pages, there is an extraordinary amount of detail in the EU-U.K. Trade and Cooperation Agreement. There will be much to process in terms of moving goods, people, and data across borders. However, the key initial takeaway is that since Jan. 1, 2021, and moving forward, the U.K. and EU can continue to trade without extra taxes being put on goods, which benefits companies in both markets and gives them certainty that finally allows them to plan for the future.
The finer detail of the Northern Ireland Protocol was agreed upon on Dec. 18 as part of the negotiations. The protocol ensures Northern Ireland-based businesses can continue trading in Great Britain, but also uniquely keeps Northern Ireland in the EU’s single market for goods given its direct EU land border via the Republic of Ireland.
Dual UK and EU Market Access
This means Northern Ireland will be the only part of the U.K. where local manufacturing standards will meet EU regulations. Because of this, the EU has agreed there will be no additional checks or controls on goods crossing between Northern Ireland and the Republic of Ireland, or into the European Union.
This matters for international trade and business because post-Brexit, Northern Ireland is the only location where U.K. and EU regulations will intersect, meaning companies will be able to trial, manufacture, and commercialize products there for both the U.K. and EU markets. Nowhere else will that be possible.
In short, Northern Ireland offers a dual, two-for-one market access opportunity for companies looking to export into the U.K. and EU from a single European manufacturing base.
From a medical manufacturing standpoint, this means EU laws with respect to medicinal and medical products will continue to apply in Northern Ireland and any QP release in Northern Ireland will be accepted in the EU. Movement of medical products from Northern Ireland into the EU will remain as it did before and will not be considered as an import. No customs controls or declarations will be required. No other location in the U.K. will have such special treatment.
This dual-market value proposition will most certainly increase Northern Ireland’s potential for second-source manufacturing growth.
Medical Manufacturing in Northern Ireland
Regardless of this new commercial advantage, the region already offers scientifically advanced manufacturing capabilities, zero-defect manufacturing standards, and a long history of advanced engineering and manufacturing excellence.
Traditionally this strength has manifested in the aerospace, automotive, technology, energy, and space industries. Northern Ireland already produces parts in 1/5 of the world’s computer hard drives (thanks to Seagate’s local operation), 40 percent of the world’s tracked mobile stone crushing and gravel screening equipment, over 30 percent of the world’s aircraft seats and high-tech composite-based aircraft wings.
The manufacturing industry in Northern Ireland in recent years has grown almost three times faster than in the rest of the U.K., and part of that is due to medical manufacturers taking advantage of regional strengths in plastics and polymers, composites design and manufacturing, and precision manufacturing.
Take for instance Denroy Plastics, an innovator in the design and manufacture of engineered polymer components and solutions. Most of their products are designed for aerospace, automotive, and defense, but when the pandemic hit, they applied their plastics manufacturing expertise to medical products, making special PPE masks through their Denpro line, including the bubl—a supple, see-through mask with 60 replaceable filters—and the Hero Shield.
Paragon Health made a similar pivot. They may be better known for their SeatingMatters line, which designs and manufactures clinical, therapeutic seating for patients and caregivers to help prevent injury, increase comfort, and reduce the cost of care. However, the company ramped up efforts to become a manufacturer and international exporter of Type IIR medical masks, helping meet the global demand for disposable PPE.
Axial3D, a Northern Irish medical 3D modeling company, partnered with Chicago digital manufacturing company Fast Radius last summer to provide a DICOM-to-print service for surgeons and hospitals across North America. The partnership allowed clinicians in the U.S., Canada, and Mexico to access 3D anatomical modeling to create precision surgical plans in as little as 48 hours, helping to clear through the backlog of surgeries that came as a result of COVID-19.
And the medical products being manufactured from the region go well beyond those being developed in response to the pandemic.
Lisburn’s Marturion specializes in advanced electronic design for a variety of medical devices including digital asthma monitors, negative pressure wound therapy devices, wearable light skincare products, and clinical chemistry analyzers, just to name a few.
Also in County Antrim, CMASS is a leader in providing flexible manufacturing services including complete product build assembly and testing, printed circuit board assembly, wiring harness and cable assembly, and software media duplication services. They are engaged in an ongoing basis in connected health research and development programs, providing prototype builds and design for a range of clients.
And while Boyce Precision Engineering is best-known around the globe for their aircraft furnishings, they also apply their component machining and machine prototyping for the medical device industry. They are a leading manufacturer of tools that allow customers to create blister packs, recover damaged blister packs, or test a pack’s integrity.
Unique Life Sciences Ecosystem
Much of the local industry’s success is attributed to the region's universities, companies, regulatory authorities, and healthcare systems being connected in a way that fosters great collaboration. It's the type of integration that simply doesn’t occur in larger countries or regions of a similar size. The companies operating in Northern Ireland can support all aspects of the development cycle, from discovery through preclinical and clinical research, to commercial manufacturing and lifecycle management.
Companies operating there have direct access to the U.K.’s National Health Service (NHS) as well as Northern Ireland’s data-driven closed-loop healthcare system, which has full longitudinal electronic health records for the population of 1.9 million, covering from birth through old age. Data of this scope does not exist in many places and offers opportunities for comparative efficacy research and data mining. Through ethical collaboration agreements, researchers can access this information to find ideal patient pools—even for more complicated diseases—creating an ideal environment for the co-creation, testing, evidence generation, and adoption of healthcare innovations.
Today, over 250 life and health sciences companies operate in Northern Ireland, including global names like Randox, Almac, Norbrook, G&L Scientific, and Teva Pharmaceutical Industries.
This includes medtech manufacturers such as Stryker, Armstrong Medical, and Elite Electronic Systems. Each of these companies employs more than 150 people in the region to design and manufacture medical devices that are exported across the globe.
As an example, Northern Ireland’s HeartSine Technologies—part of Stryker—deploys life-saving defibrillation therapy for the treatment of sudden cardiac arrest in non-traditional areas of care. The company invented the first mobile defibrillator in 1967 and its flagship product—the HeartSine samaritan PAD (Public Access Defibrillator)—is currently available in over 30 languages in 70 countries.
Together, Northern Ireland companies and investors of all sizes are focused on advancing medical devices, pharma, diagnostics, precision medicine, and connected health. Last year, the industry generated a turnover of more than $1.41 billion. Collectively, it employs more than 8,500 people.
Expertise in AI, Digital, and Connected Health
Another compelling attribute to the region is the emerging cross-sector strengths in areas like data analytics and AI, and world-class expertise in areas like cybersecurity and software development. The intersection of so many fields, made possible because of the intimate business environment, fosters innovation and collaboration in the life sciences.
Ulster University and Queen’s University Belfast, Northern Ireland’s two major universities, provide globally competitive research expertise, including 17 life sciences-related research centers and a proven track record in commercializing healthcare technologies.
In fact, Ulster University’s Connected Health Innovation Centre is a leader in connected health research, with a focus on the management of long-term chronic conditions. They are making significant advancements in the area of sensors, from those that monitor a patient’s health status and identify early signs of illness to those that track electrical responses in the brains of patients with neurodegenerative diseases, providing real-time feedback during clinical studies.
Working closely with the universities is the Health Innovation Research Alliance Northern Ireland (HIRANI). HIRANI is an alliance of universities, health organizations, and other industry bodies, established to drive and support ambitious growth in the sector. HIRANI acts as a “shop window” for Northern Ireland’s health and life sciences sector, offering partners and investors easy access to industry resources and expertise.
Strengthening the industry is Northern Ireland's global leadership position in cybersecurity, never more important given recent cyber threats targeting life science developers. Northern Ireland is home to the U.K.’s national Centre for Secure Information Technologies, located at Queen’s University Belfast, and has one of the highest concentrations of cyber employment in Europe with a talent pipeline to match, beneficial for companies looking to safeguard intellectual property and trade secrets.
Lower Operating Costs, Great Talent, and Excellent Quality of Life
The Northern Ireland workforce is highly educated and English-speaking, and business operating costs are on average 20 to 30 percent lower than the rest of the U.K. and Europe. Employee costs may be up to 50 percent lower than in the U.S., but with science workers maintaining an excellent quality of life in a truly green environment. In the age of COVID-19, the highly connected workers have adapted to working remotely and are supported by the infrastructure necessary for effective digital operations.
U.S. companies investing in the region can access expertise and grants for product development, employment, training, and R&D. For life and health science companies in particular, R&D and manufacturing activities within Northern Ireland have remained robust throughout the pandemic thanks to its infrastructure and ability to manage its own COVID-19 measures outside of the wider U.K. and EU response, enabling companies to maintain and even enhance their competitive position on a global stage.
In 2019, investment in Northern Ireland grew by more than 60 percent, the largest increase ever, and the current business investment pipeline continues to grow. More and more, despite and sometimes because of COVID-19, we are hearing from interested life science companies looking to develop healthcare products and technologies in the region.
Strong Government Support
The Northern Ireland government proactively supports the life sciences sector. As the region’s economic development agency, Invest Northern Ireland helps companies looking to establish operations in the region by brokering collaborations with local business, academia, and health organizations; providing research and entrepreneurship funding; and advising on how best to scale. Essentially, the organization serves as a catalyst helping companies identify and connect with the locations, people, and resources that will ensure their success.
The recently launched “City Deals” program will inject $1.54 billion in funding—an investment of approximately $1,000 per person—in economic growth projects. Much of it is earmarked for enhancing the life and health science capabilities, including new institutes for clinical research, digital healthcare technology, and longitudinal healthcare data capture and analysis.
As part of the initiative, Ulster University will create a center for digital technology in the areas of cardiology, diabetes, respiratory, and stroke. The project is a $70.4 million investment focused on enhancing medical device design and is software/prototyping-centric, with a strong clinical-industry pull toward creating digital healthcare technologies empowered by software engineering, AI, Internet of Things, and nanotechnology. It will include a world-class open innovation Living Lab Hub.
Construction is also underway at Kings Hall Health and Wellbeing Park to create a healthcare hub of the future. The first phase of the $128 million development will see the construction of Dataworks, a new precision medicine hub designed to attract data-focused medical companies to a secure and collaborative space. Medtech pioneer Diaceutics, which is listed on the London Stock Exchange, has already signed up as an anchor tenant, with others expected to follow suit.
Conclusion
When companies consider talent, cost, regulatory compliance, product quality and availability, IP protection, and now unique dual-market access, Northern Ireland is poised to play an essential and ever-growing role as medical manufacturers actively take steps to diversify their global supply chains.
Kevin Holland is CEO of Invest Northern Ireland. He previously served as Minister Counsellor for Life Sciences, Health and Social Care at the British Embassy in Beijing from 2016 to 2019. Previously, Holland was with Baxter for 15 years, where he led the company’s businesses in Nordic, Russia, Turkey, the Middle East, and Africa. Prior to that, he held European R&D management roles at Unilever. Holland graduated from Oxford University with an MA in chemistry and received his MBA from the IMD Business School in Switzerland.
Back in 2011, the FDA published Reliance on Foreign Sourcing in the Healthcare and Public Sector, a report that highlighted many of the underlying risks lurking within the medical device and pharmaceutical industries. They discovered that for over 43 percent of critical components, materials, and finished products intended for incorporation into medical devices and surgical equipment, no alternate source of those materials—in or outside the United States—was available. The report also found that while the most common medical manufacturing components were provided by suppliers in 32 different companies, the top single-source locations were all located in Asia.
Cut to 2021; it’s now 10 years later, and over the last several months, the medical manufacturing industry has had to rapidly respond to a global health crisis that has upended not just life sciences and healthcare, but every other industry as well.
On the one hand, it has been impressive to witness the sector’s heroic response to this once-in-a-lifetime challenge. Companies have quickly adapted, developing new technologies and producing supply to meet new or increased demand. From Northern Ireland, I’ve watched our highly flexible medtech sector quickly pivot to develop COVID-19 related PPE, medical equipment, testing, and diagnostics.
On the other hand, it didn’t take long for COVID-19 to shine a spotlight on long-known, inherent supply chain weaknesses. In March 2020, the Coronavirus Aid, Relief, and Economic Security (CARES) Act provided the FDA—for the first time—with “new authority to help prevent or mitigate negative public health impacts of medical device supply chain disruptions.”
Given the findings of the previously mentioned 2011 report alongside what the FDA classifies as “unprecedented increased demand for some medical devices, as well as significant disruptions to global medical device manufacturing and supply chain operations,” it’s clear that effective global supply chains in this sector are more critical than ever.
From discussions with global manufacturers, it is evident that a profound and lasting change to how medical devices and pharmaceuticals are sourced and distributed is already underway. These companies are actively investigating how near-shoring and dual-source pipelines can protect the on-time manufacturing and distribution of critical devices and supplies.
This shift to diversified sourcing is poised to happen much quicker for medical product manufacturing than it will for pharma. The FDA found nearly half of medical devices/surgical equipment sourcing contracts are only maintained for three years or less. Flexibility is key due to ever-changing innovations and upgrades to medical products. By contrast, pharmaceutical companies operate with a higher level of regulation and the composition of medicines do not change, so supplier contracts are typically maintained for a much longer time.
So in the complex calculus of identifying alternative source locations outside the U.S., what are the top considerations? The FDA identified six factors in its research: cost, compliance (with regulatory requirements), product quality, product availability, IP protection, and market access.
It is in this context Northern Ireland presents several unique value propositions to U.S. medical product companies evaluating second-source manufacturing locations. One of them is an entirely new post-Brexit development, and another has long existed: a truly connected life sciences ecosystem.
A New EU-UK Trade Deal
Ever since the Brexit referendum was held in 2016, the world has been speculating about the global ramifications of the U.K. leaving the EU. In particular, there have been questions about Northern Ireland, given the region’s geopolitical circumstances.
Northern Ireland is unique because it’s part of the United Kingdom but geographically it’s located across the Irish Sea, where it shares a land border with the Republic of Ireland, which is part of the European Union.
Last year, special consideration was made to address this geography’s impact on post-Brexit trade through a pre-deal agreement called the Northern Ireland Protocol. The two aims of the agreement were 1) to ensure Northern Ireland businesses could continue trading with the rest of the U.K. without interruption, and 2) to make sure any agreed trade deal did not result in a hard border on the island of Ireland.
Though the U.K. (and Northern Ireland with it) formally left the EU in January 2020, a transition period was granted through the end of December 2020. During those 12 months, the U.K. and EU worked to negotiate a trade deal against the backdrop of a global pandemic. After more than four years since the initial referendum vote, the two parties agreed on the long-sought deal on Christmas Eve 2020, just a week before the year-end deadline. The deal was officially passed just before the ball dropped on 2021.
Coming in at around 2,000 pages, there is an extraordinary amount of detail in the EU-U.K. Trade and Cooperation Agreement. There will be much to process in terms of moving goods, people, and data across borders. However, the key initial takeaway is that since Jan. 1, 2021, and moving forward, the U.K. and EU can continue to trade without extra taxes being put on goods, which benefits companies in both markets and gives them certainty that finally allows them to plan for the future.
The finer detail of the Northern Ireland Protocol was agreed upon on Dec. 18 as part of the negotiations. The protocol ensures Northern Ireland-based businesses can continue trading in Great Britain, but also uniquely keeps Northern Ireland in the EU’s single market for goods given its direct EU land border via the Republic of Ireland.
Dual UK and EU Market Access
This means Northern Ireland will be the only part of the U.K. where local manufacturing standards will meet EU regulations. Because of this, the EU has agreed there will be no additional checks or controls on goods crossing between Northern Ireland and the Republic of Ireland, or into the European Union.
This matters for international trade and business because post-Brexit, Northern Ireland is the only location where U.K. and EU regulations will intersect, meaning companies will be able to trial, manufacture, and commercialize products there for both the U.K. and EU markets. Nowhere else will that be possible.
In short, Northern Ireland offers a dual, two-for-one market access opportunity for companies looking to export into the U.K. and EU from a single European manufacturing base.
From a medical manufacturing standpoint, this means EU laws with respect to medicinal and medical products will continue to apply in Northern Ireland and any QP release in Northern Ireland will be accepted in the EU. Movement of medical products from Northern Ireland into the EU will remain as it did before and will not be considered as an import. No customs controls or declarations will be required. No other location in the U.K. will have such special treatment.
This dual-market value proposition will most certainly increase Northern Ireland’s potential for second-source manufacturing growth.
Medical Manufacturing in Northern Ireland
Regardless of this new commercial advantage, the region already offers scientifically advanced manufacturing capabilities, zero-defect manufacturing standards, and a long history of advanced engineering and manufacturing excellence.
Traditionally this strength has manifested in the aerospace, automotive, technology, energy, and space industries. Northern Ireland already produces parts in 1/5 of the world’s computer hard drives (thanks to Seagate’s local operation), 40 percent of the world’s tracked mobile stone crushing and gravel screening equipment, over 30 percent of the world’s aircraft seats and high-tech composite-based aircraft wings.
The manufacturing industry in Northern Ireland in recent years has grown almost three times faster than in the rest of the U.K., and part of that is due to medical manufacturers taking advantage of regional strengths in plastics and polymers, composites design and manufacturing, and precision manufacturing.
Take for instance Denroy Plastics, an innovator in the design and manufacture of engineered polymer components and solutions. Most of their products are designed for aerospace, automotive, and defense, but when the pandemic hit, they applied their plastics manufacturing expertise to medical products, making special PPE masks through their Denpro line, including the bubl—a supple, see-through mask with 60 replaceable filters—and the Hero Shield.
Paragon Health made a similar pivot. They may be better known for their SeatingMatters line, which designs and manufactures clinical, therapeutic seating for patients and caregivers to help prevent injury, increase comfort, and reduce the cost of care. However, the company ramped up efforts to become a manufacturer and international exporter of Type IIR medical masks, helping meet the global demand for disposable PPE.
Axial3D, a Northern Irish medical 3D modeling company, partnered with Chicago digital manufacturing company Fast Radius last summer to provide a DICOM-to-print service for surgeons and hospitals across North America. The partnership allowed clinicians in the U.S., Canada, and Mexico to access 3D anatomical modeling to create precision surgical plans in as little as 48 hours, helping to clear through the backlog of surgeries that came as a result of COVID-19.
And the medical products being manufactured from the region go well beyond those being developed in response to the pandemic.
Lisburn’s Marturion specializes in advanced electronic design for a variety of medical devices including digital asthma monitors, negative pressure wound therapy devices, wearable light skincare products, and clinical chemistry analyzers, just to name a few.
Also in County Antrim, CMASS is a leader in providing flexible manufacturing services including complete product build assembly and testing, printed circuit board assembly, wiring harness and cable assembly, and software media duplication services. They are engaged in an ongoing basis in connected health research and development programs, providing prototype builds and design for a range of clients.
And while Boyce Precision Engineering is best-known around the globe for their aircraft furnishings, they also apply their component machining and machine prototyping for the medical device industry. They are a leading manufacturer of tools that allow customers to create blister packs, recover damaged blister packs, or test a pack’s integrity.
Unique Life Sciences Ecosystem
Much of the local industry’s success is attributed to the region's universities, companies, regulatory authorities, and healthcare systems being connected in a way that fosters great collaboration. It's the type of integration that simply doesn’t occur in larger countries or regions of a similar size. The companies operating in Northern Ireland can support all aspects of the development cycle, from discovery through preclinical and clinical research, to commercial manufacturing and lifecycle management.
Companies operating there have direct access to the U.K.’s National Health Service (NHS) as well as Northern Ireland’s data-driven closed-loop healthcare system, which has full longitudinal electronic health records for the population of 1.9 million, covering from birth through old age. Data of this scope does not exist in many places and offers opportunities for comparative efficacy research and data mining. Through ethical collaboration agreements, researchers can access this information to find ideal patient pools—even for more complicated diseases—creating an ideal environment for the co-creation, testing, evidence generation, and adoption of healthcare innovations.
Today, over 250 life and health sciences companies operate in Northern Ireland, including global names like Randox, Almac, Norbrook, G&L Scientific, and Teva Pharmaceutical Industries.
This includes medtech manufacturers such as Stryker, Armstrong Medical, and Elite Electronic Systems. Each of these companies employs more than 150 people in the region to design and manufacture medical devices that are exported across the globe.
As an example, Northern Ireland’s HeartSine Technologies—part of Stryker—deploys life-saving defibrillation therapy for the treatment of sudden cardiac arrest in non-traditional areas of care. The company invented the first mobile defibrillator in 1967 and its flagship product—the HeartSine samaritan PAD (Public Access Defibrillator)—is currently available in over 30 languages in 70 countries.
Together, Northern Ireland companies and investors of all sizes are focused on advancing medical devices, pharma, diagnostics, precision medicine, and connected health. Last year, the industry generated a turnover of more than $1.41 billion. Collectively, it employs more than 8,500 people.
Expertise in AI, Digital, and Connected Health
Another compelling attribute to the region is the emerging cross-sector strengths in areas like data analytics and AI, and world-class expertise in areas like cybersecurity and software development. The intersection of so many fields, made possible because of the intimate business environment, fosters innovation and collaboration in the life sciences.
Ulster University and Queen’s University Belfast, Northern Ireland’s two major universities, provide globally competitive research expertise, including 17 life sciences-related research centers and a proven track record in commercializing healthcare technologies.
In fact, Ulster University’s Connected Health Innovation Centre is a leader in connected health research, with a focus on the management of long-term chronic conditions. They are making significant advancements in the area of sensors, from those that monitor a patient’s health status and identify early signs of illness to those that track electrical responses in the brains of patients with neurodegenerative diseases, providing real-time feedback during clinical studies.
Working closely with the universities is the Health Innovation Research Alliance Northern Ireland (HIRANI). HIRANI is an alliance of universities, health organizations, and other industry bodies, established to drive and support ambitious growth in the sector. HIRANI acts as a “shop window” for Northern Ireland’s health and life sciences sector, offering partners and investors easy access to industry resources and expertise.
Strengthening the industry is Northern Ireland's global leadership position in cybersecurity, never more important given recent cyber threats targeting life science developers. Northern Ireland is home to the U.K.’s national Centre for Secure Information Technologies, located at Queen’s University Belfast, and has one of the highest concentrations of cyber employment in Europe with a talent pipeline to match, beneficial for companies looking to safeguard intellectual property and trade secrets.
Lower Operating Costs, Great Talent, and Excellent Quality of Life
The Northern Ireland workforce is highly educated and English-speaking, and business operating costs are on average 20 to 30 percent lower than the rest of the U.K. and Europe. Employee costs may be up to 50 percent lower than in the U.S., but with science workers maintaining an excellent quality of life in a truly green environment. In the age of COVID-19, the highly connected workers have adapted to working remotely and are supported by the infrastructure necessary for effective digital operations.
U.S. companies investing in the region can access expertise and grants for product development, employment, training, and R&D. For life and health science companies in particular, R&D and manufacturing activities within Northern Ireland have remained robust throughout the pandemic thanks to its infrastructure and ability to manage its own COVID-19 measures outside of the wider U.K. and EU response, enabling companies to maintain and even enhance their competitive position on a global stage.
In 2019, investment in Northern Ireland grew by more than 60 percent, the largest increase ever, and the current business investment pipeline continues to grow. More and more, despite and sometimes because of COVID-19, we are hearing from interested life science companies looking to develop healthcare products and technologies in the region.
Strong Government Support
The Northern Ireland government proactively supports the life sciences sector. As the region’s economic development agency, Invest Northern Ireland helps companies looking to establish operations in the region by brokering collaborations with local business, academia, and health organizations; providing research and entrepreneurship funding; and advising on how best to scale. Essentially, the organization serves as a catalyst helping companies identify and connect with the locations, people, and resources that will ensure their success.
The recently launched “City Deals” program will inject $1.54 billion in funding—an investment of approximately $1,000 per person—in economic growth projects. Much of it is earmarked for enhancing the life and health science capabilities, including new institutes for clinical research, digital healthcare technology, and longitudinal healthcare data capture and analysis.
As part of the initiative, Ulster University will create a center for digital technology in the areas of cardiology, diabetes, respiratory, and stroke. The project is a $70.4 million investment focused on enhancing medical device design and is software/prototyping-centric, with a strong clinical-industry pull toward creating digital healthcare technologies empowered by software engineering, AI, Internet of Things, and nanotechnology. It will include a world-class open innovation Living Lab Hub.
Construction is also underway at Kings Hall Health and Wellbeing Park to create a healthcare hub of the future. The first phase of the $128 million development will see the construction of Dataworks, a new precision medicine hub designed to attract data-focused medical companies to a secure and collaborative space. Medtech pioneer Diaceutics, which is listed on the London Stock Exchange, has already signed up as an anchor tenant, with others expected to follow suit.
Conclusion
When companies consider talent, cost, regulatory compliance, product quality and availability, IP protection, and now unique dual-market access, Northern Ireland is poised to play an essential and ever-growing role as medical manufacturers actively take steps to diversify their global supply chains.
Kevin Holland is CEO of Invest Northern Ireland. He previously served as Minister Counsellor for Life Sciences, Health and Social Care at the British Embassy in Beijing from 2016 to 2019. Previously, Holland was with Baxter for 15 years, where he led the company’s businesses in Nordic, Russia, Turkey, the Middle East, and Africa. Prior to that, he held European R&D management roles at Unilever. Holland graduated from Oxford University with an MA in chemistry and received his MBA from the IMD Business School in Switzerland.