Maria Shepherd, Medi-Vantage05.01.20
If you’re like me in our current circumstances, you are unfortunately finding it impossible to focus on much beyond the coronavirus pandemic. To their credit, many involved within our industry are fixated on determining the best ways to protect the public health. Meanwhile, the happenings of business outside of COVID-19 such as in the M&A space continue, but garner much less attention. For example, GE recently completed the sale of its BioPharma business unit to Danaher (Table 1) for more than $20 billion1—few noticed. Abbvie’s $63 billion buyout of Allergan is projected to close in May after they divest three product lines2; a tough thing to do when the SEC, as well as the financial and legal world, is focused on coronavirus. Further, Stryker’s $5.4 billion bid for Wright Medical is getting chewed up by the FTC, which is unhappy with the combined strengths of Stryker and Wright Medical in lower extremity joint implants. Since Wright Medical has a 70 percent share of market in the total ankle replacement market,3 a divestiture may be required. Smith+Nephew may be the real beneficiary in the situation, but again, has anyone noticed?
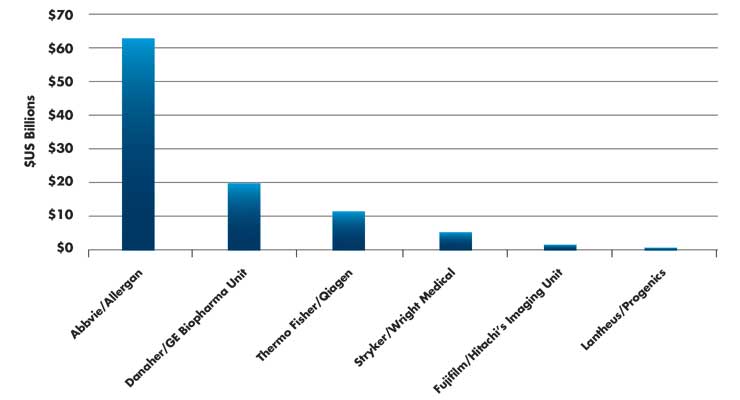
Table 1: Recent acquisitions impacting the medtech space1,2
Why This Is Important
Medical device innovation and policymaking not directly linked to COVID-19 isn’t completely frozen, but it is definitely experiencing a spring frost. While this may spell trouble for startups seeking funding, it could be an opportunity for medtech companies with something to offer to help combat the COVID-19 pandemic. The early stage telehealth industry is getting a shot in the arm from the FCC, which announced a $200 million program that will fund telecommunications equipment and services for healthcare providers.4 It will relieve the enormous strain on our healthcare providers as they deal with the surge in patients from the pandemic.
Meanwhile, other medical device manufacturers are taking unprecedented steps to help. For example, 3M has published its price lists to help customers avoid inflated prices and to help fight PPE counterfeits during COVID-19.5
In a unique and previously unheard of partnership, Tesla will make Medtronic ventilators in a New York factory repurposed from the manufacture of solar power cells.6 Medtronic is trying to increase production of ventilators critical for treating the sickest COVID-19 patients. This pairing is the result of great minds working together; in this case, those of Omar Ishrak and Elon Musk.
Smiths Medical will produce 10,000 ventilators for the British government.7 They have announced production of ventilators will surge from hundreds of ventilators per month to thousands to meet the need.
Medtronic shared its design specifications for the Puritan Bennett 560 to accelerate efforts to increase global ventilator production.8 The Puritan Bennett 560 is a small, lightweight, mobile ventilator that delivers airway support for adults and children in the clinical environment and for home care.
In a survey published in the journal Disaster Medicine and Public Health Preparedness, the supply and distribution of mechanical ventilators was noted to be of critical importance in planning for acute public health emergencies.9 The study’s goal was to estimate the quantity of pediatric and adult ventilators at acute care hospitals in the U.S. Of the 5,752 U.S. acute care hospitals included in the 2007 American Hospital Association database surveyed, 4,305 (74.8 percent), accounting for 83.8 percent of U.S. ICU beds, responded, suggesting the clinicians in ICUs recognize the perils of a shortage. The study revealed of the 52,118 full-featured mechanical ventilators owned by respondent hospitals, 24,204 (46.4 percent) were for pediatric/neonatal use (Table 2). Accounting for non-respondents, the study authors estimated there were 62,188 full-featured mechanical ventilators owned by acute care hospitals in the U.S. at the time. In addition, respondent hospitals reported having 82,755 ventilators other than full-featured mechanical ventilators. The authors also found while the number of mechanical ventilators per capita is greater than those reported in other developed countries, there is broad variation across U.S. states. In addition, the ratio of infant/pediatric-capable ventilators to those designed for adults is large.
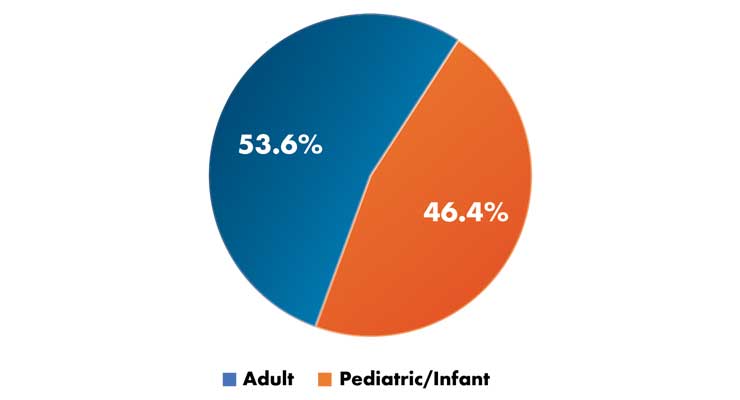
Table 2: In 2010, a survey of 4,305 U.S. acute care hospitals (which accounted for 83.8 percent of U.S. intensive care unit beds) was published on mechanical ventilation capacity. The pie chart shows the total breakdown between the two types of full-feature mechanical ventilators the facilities owned.9
The Medical Device Supply Chain
The ability of healthcare organizations to respond to COVID-19 is diminished by the immediate and increasing disruption to the supply of personal protective products and medical products. The surge in demand and dearth of supplies was predictable. China manufactures approximately 50 percent more medical and pharmaceutical supplies than the U.S. (Table 3), according to data supplied by Euromonitor International.10 But China now needs those valuable supplies for its affected population, at a time when manufacturing ground to a halt across the country.
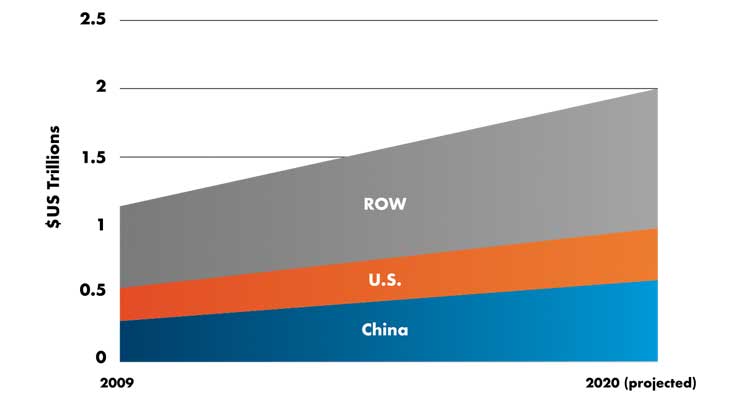
Table 3: Worldwide production of pharmaceuticals and medical equipment10
Medical Device Industry Post-COVID-19
Expect telehealth to dominate a much larger segment of direct patient care. Hospitals are desperate to free up beds for only the sickest COVID-19 patients, and clinicians across the nation are considering the use of new technologies to monitor patients from their homes.11
Doctors at multiple hospitals said their discussions with manufacturers of telehealth devices grew substantially after the FDA issued guidance indicating telemedicine tools could be used during the COVID-19 outbreak.11 Most healthcare organizations are expanding the use of video and telephone appointments with patients, but remote monitoring of vital signs was considered the “next phase.” Many barriers exist—supply of vital signs monitors for home use, training for clinicians and patients, integration with hospital IT and EMRs.
Also, stockpiles will be larger, as our healthcare workers must be protected. Be assured we will not underestimate the power of a pandemic for some time to come. Current ratios for managing a surge of patients is largely focused on human-caused disasters, such as mass shootings, and most hospitals that have planned for a surge calculate it as 120 percent of average patient load.12
Expect innovation to emerge from the pandemic. Many medical device concepts are being dusted off to help address the coronavirus; it is exciting to see this response from technology providers.
The Medi-Vantage Perspective
Bravo to all these medical devices companies adopting strategies of collaboration and transparency to fight this global pandemic. What can your medical device company do to help?
References
Maria Shepherd has more than 20 years of experience in medical device marketing in small startups and top-tier companies. After her industry career, including her role as VP of marketing for Oridion Medical, where she boosted the company valuation prior to its acquisition, director of marketing for Philips Medical, and senior management roles at Boston Scientific Corp., she founded Medi-Vantage. Medi-Vantage provides marketing, business strategy, and innovation research for the medical device, diagnostic, and digital health industries. The firm quantitatively and qualitatively sizes and segments opportunities, evaluates new technologies, provides marketing services, and assesses prospective acquisitions. Shepherd has taught marketing and product development courses and is a member of the Aligo Medtech Investment Committee (www.aligo.com). She can be reached at 855-343-3100. Visit her website at www.medi-vantage.com.

Table 1: Recent acquisitions impacting the medtech space1,2
Why This Is Important
Medical device innovation and policymaking not directly linked to COVID-19 isn’t completely frozen, but it is definitely experiencing a spring frost. While this may spell trouble for startups seeking funding, it could be an opportunity for medtech companies with something to offer to help combat the COVID-19 pandemic. The early stage telehealth industry is getting a shot in the arm from the FCC, which announced a $200 million program that will fund telecommunications equipment and services for healthcare providers.4 It will relieve the enormous strain on our healthcare providers as they deal with the surge in patients from the pandemic.
Meanwhile, other medical device manufacturers are taking unprecedented steps to help. For example, 3M has published its price lists to help customers avoid inflated prices and to help fight PPE counterfeits during COVID-19.5
In a unique and previously unheard of partnership, Tesla will make Medtronic ventilators in a New York factory repurposed from the manufacture of solar power cells.6 Medtronic is trying to increase production of ventilators critical for treating the sickest COVID-19 patients. This pairing is the result of great minds working together; in this case, those of Omar Ishrak and Elon Musk.
Smiths Medical will produce 10,000 ventilators for the British government.7 They have announced production of ventilators will surge from hundreds of ventilators per month to thousands to meet the need.
Medtronic shared its design specifications for the Puritan Bennett 560 to accelerate efforts to increase global ventilator production.8 The Puritan Bennett 560 is a small, lightweight, mobile ventilator that delivers airway support for adults and children in the clinical environment and for home care.
In a survey published in the journal Disaster Medicine and Public Health Preparedness, the supply and distribution of mechanical ventilators was noted to be of critical importance in planning for acute public health emergencies.9 The study’s goal was to estimate the quantity of pediatric and adult ventilators at acute care hospitals in the U.S. Of the 5,752 U.S. acute care hospitals included in the 2007 American Hospital Association database surveyed, 4,305 (74.8 percent), accounting for 83.8 percent of U.S. ICU beds, responded, suggesting the clinicians in ICUs recognize the perils of a shortage. The study revealed of the 52,118 full-featured mechanical ventilators owned by respondent hospitals, 24,204 (46.4 percent) were for pediatric/neonatal use (Table 2). Accounting for non-respondents, the study authors estimated there were 62,188 full-featured mechanical ventilators owned by acute care hospitals in the U.S. at the time. In addition, respondent hospitals reported having 82,755 ventilators other than full-featured mechanical ventilators. The authors also found while the number of mechanical ventilators per capita is greater than those reported in other developed countries, there is broad variation across U.S. states. In addition, the ratio of infant/pediatric-capable ventilators to those designed for adults is large.

Table 2: In 2010, a survey of 4,305 U.S. acute care hospitals (which accounted for 83.8 percent of U.S. intensive care unit beds) was published on mechanical ventilation capacity. The pie chart shows the total breakdown between the two types of full-feature mechanical ventilators the facilities owned.9
The Medical Device Supply Chain
The ability of healthcare organizations to respond to COVID-19 is diminished by the immediate and increasing disruption to the supply of personal protective products and medical products. The surge in demand and dearth of supplies was predictable. China manufactures approximately 50 percent more medical and pharmaceutical supplies than the U.S. (Table 3), according to data supplied by Euromonitor International.10 But China now needs those valuable supplies for its affected population, at a time when manufacturing ground to a halt across the country.

Table 3: Worldwide production of pharmaceuticals and medical equipment10
Medical Device Industry Post-COVID-19
Expect telehealth to dominate a much larger segment of direct patient care. Hospitals are desperate to free up beds for only the sickest COVID-19 patients, and clinicians across the nation are considering the use of new technologies to monitor patients from their homes.11
Doctors at multiple hospitals said their discussions with manufacturers of telehealth devices grew substantially after the FDA issued guidance indicating telemedicine tools could be used during the COVID-19 outbreak.11 Most healthcare organizations are expanding the use of video and telephone appointments with patients, but remote monitoring of vital signs was considered the “next phase.” Many barriers exist—supply of vital signs monitors for home use, training for clinicians and patients, integration with hospital IT and EMRs.
Also, stockpiles will be larger, as our healthcare workers must be protected. Be assured we will not underestimate the power of a pandemic for some time to come. Current ratios for managing a surge of patients is largely focused on human-caused disasters, such as mass shootings, and most hospitals that have planned for a surge calculate it as 120 percent of average patient load.12
Expect innovation to emerge from the pandemic. Many medical device concepts are being dusted off to help address the coronavirus; it is exciting to see this response from technology providers.
The Medi-Vantage Perspective
Bravo to all these medical devices companies adopting strategies of collaboration and transparency to fight this global pandemic. What can your medical device company do to help?
References
- bit.ly/mpo200501
- bit.ly/mpo200502
- bit.ly/mpo200503
- bit.ly/mpo200504
- bit.ly/mpo200505
- bit.ly/mpo200506
- bit.ly/mpo200507
- bit.ly/mpo200508
- bit.ly/mpo200509
- bit.ly/mpo200510
- bit.ly/mpo200511
- Medi-Vantage research
Maria Shepherd has more than 20 years of experience in medical device marketing in small startups and top-tier companies. After her industry career, including her role as VP of marketing for Oridion Medical, where she boosted the company valuation prior to its acquisition, director of marketing for Philips Medical, and senior management roles at Boston Scientific Corp., she founded Medi-Vantage. Medi-Vantage provides marketing, business strategy, and innovation research for the medical device, diagnostic, and digital health industries. The firm quantitatively and qualitatively sizes and segments opportunities, evaluates new technologies, provides marketing services, and assesses prospective acquisitions. Shepherd has taught marketing and product development courses and is a member of the Aligo Medtech Investment Committee (www.aligo.com). She can be reached at 855-343-3100. Visit her website at www.medi-vantage.com.