Maria Shepherd, President and Founder, Medi-Vantage09.06.19
The shift in U.S. healthcare delivery over the past decade has been dramatic. The impact on the medical device space has been substantial, especially in the methods used to make medtech purchasing decisions. Centralization has been a key strategy that drives hospital economies of scale. The amount of control an Integrated Delivery Network (IDN) has is extraordinary and covers a range of pharmaceuticals, new medtech product review, health IT (including EMRs), and the management of the hospital facility like a business. Pity the physician that is just starting their practice today! Ten years ago, a physician had autonomy and almost limitless financial rewards. All that has changed.
Why This Is Important
The top five IDNs employ 135,913 MDs and DOs in the U.S., approximately 15 percent of all active physicians in the country.1 According to Statista, in 2015, there were 870,900 active physicians in the U.S.2 The U.S. has a high concentration of physicians as compared to other countries and the Washington, D.C. area, Massachusetts, and Maryland have the greatest number of physicians per patient population.2 The healthcare industry is no place for xenophobia; almost 25 percent of all active U.S. physicians were educated outside of the U.S.2
Top 10 IDNs in the U.S.
Take a look at the ratios of the total number of hospitals relative to the number of facilities as presented in Table 1. This is a testament to the financial and clinical commitments these IDNs have made to the continuum of care.
Most of these facilities are outpatient centers, lead by Ascension Health (1,887), HCA (1,576), Catholic Health Initiatives (1,334), Providence St. Joseph Health (1,270), and VA (1,271).1 Trinity Health is the leader in the number of residential facilities (125), while the rest of the top 10 vary between only one facility through to 107.1
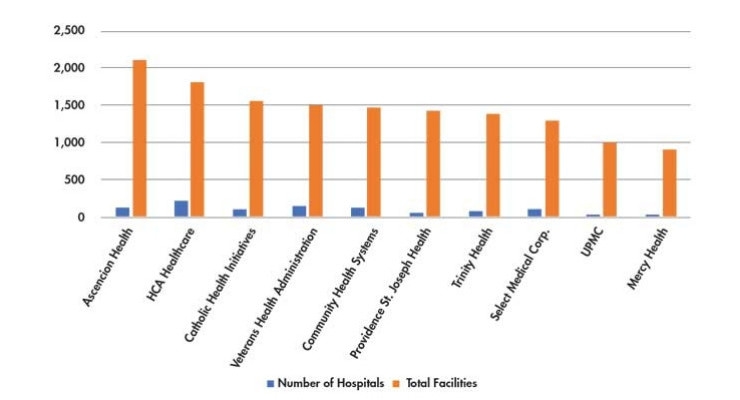
Table 1: The ratio of the total number of hospitals relative to the number of facilities at the Top 10 IDNs in the U.S.1
Physicians Turn to Medtech to Supplement Their Incomes
The change in IDN growth has had little impact on physician compensation levels (Table 2), which remained relatively flat in 2017 as compared with 2016.3 As such, many physicians take advantage of other opportunities to supplement their incomes.
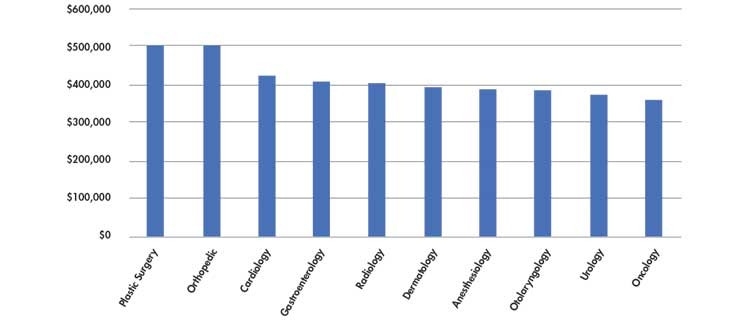
Table 2: Annual salaries of physicians by specialty (2018 salary survey results).4
It is still routine for physicians to get paid for consulting with medical device companies. However, a study published in JAMA Surgery reported that full disclosure of compensation from medical device companies is still not the norm when the doctors have authored articles appearing in medical journals.5 In fact, the JAMA Surgery study determined that a minority of publications written by a doctor (37.3 percent) disclosed compensation from device companies as a relevant conflict of interest. In the JAMA study, researchers focused on 10 larger medical device companies such as Medtronic, Stryker, Intuitive Surgical, Edwards, J&J, Olympus, W.L. Gore, and Baxter. They selected the 10 highest paid physicans from each of these companies, creating a sample of 100 physicians. The investigators noted greater than 7,000 journal articles written by the 100 physicans in the sample. Researchers compared the conflict of interest reports in the publications to compensation reported on the CMS Open Payment Database and arrived at their conclusion—only 37.3 percent of physicians disclosed their medtech relationships via disclosure in the article. In the sample of 100 doctors, the median annual payment came to $95,993.
Here are the rules: when a physician is authoring a research study or article, the International Committee of Medical Journal Editors compels the author(s) to reveal any pertinent conflict of interest that has taken place in the prior three years. The JAMA research also showed that, up until 2010, there was no global reporting mechanism to disclose conflicts of interest.
In 2010, CMS introduced the Open Payment Database.6 Open Payments is a disclosure program that sponsors more transparency in accountable healthcare systems by making the financial relationships between manufacturers, healthcare providers (physicians and teaching hospitals), and group purchasing organizations (GPOs) open to the public. Open Payments must report annually to Congress and the last report was issued in April 2019.7
The report makes for an interesting read. In the latest report, it is stated that in 2017, medtech manufacturers and GPOs reported $8.4 billion in payments. The payments were comprised of 11.54 million records paid to more than 600,000 physicians and over 1,100 teaching hospitals. Since 2010, CMS has accounted for $33.4 billion in payments and ownership, as well as investment interests (Table 3).
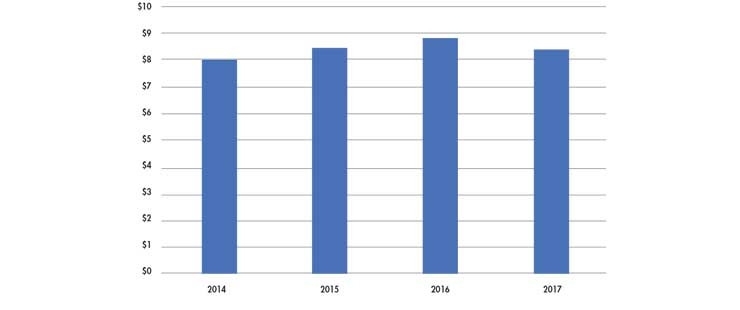
Table 3: Total value of payments to physicians and teaching hospitals (in billions) as reported on the CMS Open Payment Database.7 The program began recording payment data for 2013, but only did so from August through end of year, noting $4.61 billion in payments.
The Medi-Vantage Perspective
Physicians can still be compensated if they are rendering a true service. Is your medical device one that has been engineered to provide better outcomes or to reduce costs? If your device meets these goals, you can still reimburse physicians for their consultative services.
References
Maria Shepherd has more than 20 years of leadership experience in medical device/life science marketing in small startups and top-tier companies. After her industry career, including her role as vice president of marketing for Oridion Medical, where she boosted the company valuation prior to its acquisition by Covidien/Medtronic, director of marketing for Philips Medical, and senior management roles at Boston Scientific Corp., she founded Medi-Vantage. Medi-Vantage provides marketing, business strategy, and innovation research for the medical device, diagnostic, and digital health industries. The firm quantitatively and qualitatively sizes and segments opportunities, evaluates new technologies, provides marketing services, and assesses prospective acquisitions. Shepherd has taught marketing and product development courses and is a member of the Aligo Medtech Investment Committee (www.aligo.com). She can be reached at 855-343-3100. Visit her website at www.medi-vantage.com.
Why This Is Important
The top five IDNs employ 135,913 MDs and DOs in the U.S., approximately 15 percent of all active physicians in the country.1 According to Statista, in 2015, there were 870,900 active physicians in the U.S.2 The U.S. has a high concentration of physicians as compared to other countries and the Washington, D.C. area, Massachusetts, and Maryland have the greatest number of physicians per patient population.2 The healthcare industry is no place for xenophobia; almost 25 percent of all active U.S. physicians were educated outside of the U.S.2
Top 10 IDNs in the U.S.
Take a look at the ratios of the total number of hospitals relative to the number of facilities as presented in Table 1. This is a testament to the financial and clinical commitments these IDNs have made to the continuum of care.
Most of these facilities are outpatient centers, lead by Ascension Health (1,887), HCA (1,576), Catholic Health Initiatives (1,334), Providence St. Joseph Health (1,270), and VA (1,271).1 Trinity Health is the leader in the number of residential facilities (125), while the rest of the top 10 vary between only one facility through to 107.1

Table 1: The ratio of the total number of hospitals relative to the number of facilities at the Top 10 IDNs in the U.S.1
Physicians Turn to Medtech to Supplement Their Incomes
The change in IDN growth has had little impact on physician compensation levels (Table 2), which remained relatively flat in 2017 as compared with 2016.3 As such, many physicians take advantage of other opportunities to supplement their incomes.

Table 2: Annual salaries of physicians by specialty (2018 salary survey results).4
It is still routine for physicians to get paid for consulting with medical device companies. However, a study published in JAMA Surgery reported that full disclosure of compensation from medical device companies is still not the norm when the doctors have authored articles appearing in medical journals.5 In fact, the JAMA Surgery study determined that a minority of publications written by a doctor (37.3 percent) disclosed compensation from device companies as a relevant conflict of interest. In the JAMA study, researchers focused on 10 larger medical device companies such as Medtronic, Stryker, Intuitive Surgical, Edwards, J&J, Olympus, W.L. Gore, and Baxter. They selected the 10 highest paid physicans from each of these companies, creating a sample of 100 physicians. The investigators noted greater than 7,000 journal articles written by the 100 physicans in the sample. Researchers compared the conflict of interest reports in the publications to compensation reported on the CMS Open Payment Database and arrived at their conclusion—only 37.3 percent of physicians disclosed their medtech relationships via disclosure in the article. In the sample of 100 doctors, the median annual payment came to $95,993.
Here are the rules: when a physician is authoring a research study or article, the International Committee of Medical Journal Editors compels the author(s) to reveal any pertinent conflict of interest that has taken place in the prior three years. The JAMA research also showed that, up until 2010, there was no global reporting mechanism to disclose conflicts of interest.
In 2010, CMS introduced the Open Payment Database.6 Open Payments is a disclosure program that sponsors more transparency in accountable healthcare systems by making the financial relationships between manufacturers, healthcare providers (physicians and teaching hospitals), and group purchasing organizations (GPOs) open to the public. Open Payments must report annually to Congress and the last report was issued in April 2019.7
The report makes for an interesting read. In the latest report, it is stated that in 2017, medtech manufacturers and GPOs reported $8.4 billion in payments. The payments were comprised of 11.54 million records paid to more than 600,000 physicians and over 1,100 teaching hospitals. Since 2010, CMS has accounted for $33.4 billion in payments and ownership, as well as investment interests (Table 3).

Table 3: Total value of payments to physicians and teaching hospitals (in billions) as reported on the CMS Open Payment Database.7 The program began recording payment data for 2013, but only did so from August through end of year, noting $4.61 billion in payments.
The Medi-Vantage Perspective
Physicians can still be compensated if they are rendering a true service. Is your medical device one that has been engineered to provide better outcomes or to reduce costs? If your device meets these goals, you can still reimburse physicians for their consultative services.
References
- http://bit.ly/mpo190901
- http://bit.ly/mpo190902
- http://bit.ly/mpo190903
- http://bit.ly/mpo190904
- http://bit.ly/mpo190905
- http://bit.ly/mpo190906
- http://bit.ly/mpo190907 [PDF]
Maria Shepherd has more than 20 years of leadership experience in medical device/life science marketing in small startups and top-tier companies. After her industry career, including her role as vice president of marketing for Oridion Medical, where she boosted the company valuation prior to its acquisition by Covidien/Medtronic, director of marketing for Philips Medical, and senior management roles at Boston Scientific Corp., she founded Medi-Vantage. Medi-Vantage provides marketing, business strategy, and innovation research for the medical device, diagnostic, and digital health industries. The firm quantitatively and qualitatively sizes and segments opportunities, evaluates new technologies, provides marketing services, and assesses prospective acquisitions. Shepherd has taught marketing and product development courses and is a member of the Aligo Medtech Investment Committee (www.aligo.com). She can be reached at 855-343-3100. Visit her website at www.medi-vantage.com.




























