Allyssa Eclarin and Ray Hein, Propel03.07.19
The medical device sector is an estimated $147.7 billion dollar industry1 that, up until recently, followed a very strict “product-centric” model. But the rise of Amazon, social media, and personalized healthcare are causing the end customer, or patient, to demand a more personalized experience. In order to thrive in this new digital world, it is crucial the industry pivots toward a more customer-centric approach.
Social media and patient (customer) satisfaction are pushing companies to embrace and incorporate patient needs and engagement into their product lifecycle, which can be used to create more relevant products for their users. As the industry is projected to grow to an estimated $173 billion in 2019, it makes business sense for companies to embrace this customer-centric strategy going forward.
When it comes to medical devices, such as orthopedics, pacemakers, or even 3D-printed prosthetic limbs, obtaining and retaining patients or customers is vital to maintaining a successful and profitable business. Further, in order to stay competitive and retain customers in any industry, especially the growing and very competitive medical device industry, companies need to implement a customer-centric strategy within the organization.
Customer centricity can be quickly defined as allowing the customer’s or patient’s needs to drive the product offerings. Customer-centric strategies can increase patient satisfaction and help a firm retain those customers going forward despite the emergence of competing products and/or as shifts in healthcare continue to take hold.
Patient satisfaction should be the priority of any organization, regardless of market size. According to a study by McKinsey,2 70 percent of buying experiences are based on how the customer feels they’re being treated. Today’s customers or patients expect to be treated with white gloves. They aren’t just asking for a great “customer experience,” they are asking for products that fit their needs and lifestyle.
Customers want to be heard, as well as properly cared for even after the “sale.” Customer service in medicine (or any industry for that matter) should not be limited to the original “sale.” It must incorporate the entire lifecycle of the product. Many companies have been operating under the notion that customer service and the product experience start and end at the point of purchase of a product or medical device. Rather, the truth is, good product experience and customer service encompass every interaction a customer has with an organization, from the patient’s first introduction to the product through to the point at which it is no longer used. This includes the moment they purchase the medical device to even their experience with a firm’s customer complaint department.
The New Generation of Patients
Today’s new generation of patients don’t just want customized solutions or better experiences, they’re demanding them. Patients and customers are feeling more empowered than ever. From custom-printed prosthetics to IoT-powered precision medical devices, customer expectations are at an all-time high. Further, with non-medical industries dictating change, and consumer subscription services booming to the likes of Netflix, Blue Apron, and other “custom box” services, customers from all types of industries are expecting their other purchasing experiences to operate the same, from their car dealership to grocery stores to even healthcare.
How Does That Look in Healthcare?
No longer are we creating products that fit what someone in a boardroom decided was important for their bottom line or what shareholders suggest a company makes to appease investors. Now, medical device manufacturers must listen to and continuously incorporate patient feedback into the product lifecycle.
What does that mean?
That means engaging and surveying customers or patients in the field. Find out what they want, how they respond to the products and experiences being offered, how they feel about the company, and what needs are not currently being fulfilled.
Then, the patient feedback should be incorporated into the product lifecycle via an automated means—preferably a product lifecycle management (PLM) or quality management system (QMS), and ultimately incorporated into the product lifecycle of future iterations. Patient engagement and feedback should drive future products and work toward eliminating design issues.
3D Printing and Customer-Centricity—A Real-World Example
Additive manufacturing (sometimes referred to as 3D printing) represents a great example of a medical device industry change that has the customer or patient in mind. Instead of taking months to get a metal part (e.g., create a plastic prototype, build out the tool for a metal prototype, and then go to volume production), additive manufacturing can eliminate significant time and costs from the process.
When 3D printing first emerged within the mainstream, few could have predicted how it had the potential to grow as a manufacturing option and revolutionize the medical device industry, let alone almost every other industry in virtually every corner of the world. While some may have viewed 3D printing as a solution for custom toys or trinkets, it is actually a perfect driver of customer-centricity in medicine.
For example, within the healthcare space, 3D printing can be used to produce prosthetic limbs customized to fit a specific patient in a relatively short period of time, especially when compared to more traditional manufacturing methods. Typically, it can be quite common for amputees to wait months or longer for a new prosthetic limb.
CSIRO, Australia’s premier national research organization, partners with Anatomics3 to deliver 3D-printed prosthetics (Figure 1). This novel approach greatly speeds up creation and delivery of new limbs, sternums, and other body parts at a lower premium. The ability to make a custom-designed body part in hours is truly a customer-centric approach to medical devices.
Prescription Glasses Reinvented
Another excellent example of a successful customer-centricity project in healthcare is Warby Parker, an American online retailer of prescription glasses and sunglasses. Warby Parker flipped the traditional model of prescription glasses on its head by serving the patient directly—a desire of prescription glasses users, but one that was going unfulfilled by the industry.
Warby Parker listened and, as a result, discovered the frustration patients found with the existing prescription glasses purchasing model. Customers were seeking affordable options to counter the expensive prescription glasses they were purchasing through traditional methods. The company also found some patients didn’t want to go to a brick-and-mortar store just to buy a pair of glasses.
Warby Parker listened to customers’ feedback and opened an online marketplace to serve as a lower cost, high-quality alternative. Warby Parker even works to serve those who prefer to go into a physical location to try on glasses, but still want a lower cost; the company will send those customers five pairs for free to try on. Still another option for those customers, Warby Parker has set up stores across the country while still offering a lower cost than traditional retailers.
In order to further distinguish itself against competitors, Warby Parker donates a pair of glasses for each pair purchased through its “Buy a Pair, Give a Pair” program. The firm is using its business to help with philanthropic efforts across the world—another example of customer centricity, but on a humanitarian level.
Hearing on Demand by Eargo
Another example of a successful customer-centricity strategy in healthcare is Eargo, an online retailer of beautifully designed hearing aids for the United States. As the population continues to age, hearing loss is affecting more and more people. Like Warby Parker, Eargo established a sales model of selling the company’s offerings—hearing aids—directly to patients, specifically those with mild to moderate hearing loss (Figure 2).
Frustrated by high costs and inconvenience, consumers were unhappy with available buying options and methods. They didn’t want to travel to brick-and-mortar stores; rather, consumers much more preferred shopping from the convenience of their homes. Eargo listened to these requests and responded appropriately. The company sells award-winning hearing aids online and by phone at a reasonable price by cutting out the middlemen.
Healthcare is an undeniable need for all now and perhaps even more so in the future as people begin to live longer and require different devices to enable a better quality of life. Just as Apple and Amazon innovated and, as a result, impacted the world, healthcare and medical device companies will need to consider the models of Warby Parker and Eargo. Medtech firms need to innovate while also listening to the needs and requests of patients and potential customers, instead of trying to be the driver of the technology.
Driving Customer-Centricity
For organizations to successfully employ a customer-centric strategy, they need to do so from the inside out. That means getting each department and team member on board with the methodology. Then, the question remains, “What exactly does that look like?”
Engineering and product teams need to design with the customer’s needs in mind. They need to consider feedback gathered in the field and use that as a guide to dictate product features and materials used. Marketing and sales teams should understand the potential customer and coordinate efforts accordingly. If customers are responding to certain products or offerings, that should drive a product campaign and/or sales channel strategy. Quality teams should gather feedback from customers using a quality management system that allows them to feed complaints and comments back into a CRM for engineers to eliminate product issues and drive iterations and changes of future offerings.
This cycle will create a closed loop for engineering and allow them to create products customers will use and value. Traditionally, engineering has been closed off from feedback from the field, but in a customer-centric model, engineering is incorporated into the conversation and able to address suggested changes and customer comments via the next product version. Further, in a customer-centric model, customer feedback should dictate each phase of the product lifecycle.
The Technology to Drive Customer Centricity
The importance of enabling clear communication between internal teams as well as between the customer (e.g., feedback, complaints) and the company has been well established. Now, it’s vital to ensure the right technology is in place to facilitate this exchange.
Quality management software (QMS) is a “must have” for all medical device companies. Quality management software allows key personnel to manage the quality of the company’s products through various tools and dashboards. In the age of the customer and for those organizations who look to follow a customer-centric strategy, however, a QMS system that provides more than the standard set of features is required.
Just as open collaboration between team members is necessary, so is the same type of functionality in the software solution. A collaborative QMS that seamlessly interfaces with other systems is the first factor to consider. Siloed departmental processes will need to be placed into a secure and robust system. Ideally, the QMS will store customer, product, and quality data all in one place. This enables all customer data to be connected and referenced from one central location, including (and perhaps most importantly) customer complaints and feedback. Whether a company is dealing with those customer complaints, supplier incidents, or non-conformances, there needs to be an integrated process that can quickly identify and resolve issues.
A good QMS solution would ideally eliminate the challenges of manual entry. When a company adopts a customer-centric approach, all customer, product, and quality data should be stored in one centralized location. Manually entering complaints, related items, and feedback from one system to another can lead to errors and incorrect information being attached to the wrong customers or products, or not being transferred at all. Customer feedback or complaints should flow seamlessly into a company’s QMS and PLM systems; engineering can then quickly resolve product issues in the appropriate product for later versions. Only through direct integration between the QMS and PLM systems can a company be certain the right quality issues are being flagged against the correct product revisions and engineering changes.
Another benefit of a single system for quality is that everyone—engineering, operations, and QA—can collaborate on quality management processes such as CAPAs (corrective and preventive actions, or corrective action preventive actions), NCMRs (non-conforming material reports), and SCARs (supplier corrective action requests). A QMS system should help improve compliance across the board, not hinder it. A compliant organization not only helps reduce business risk, it also helps keep customers and patients safe.
An ideal QMS solution should also have pre-configured workflows to help companies reduce errors and remain compliant. With pre-configured workflows, built-in best practices can be established and collaboration is better facilitated across teams. For companies in the medical device industry, quality management systems are held to a much higher bar. Medical device companies need to comply with standards like 21 CFR Part 11 and ensure their systems are fully validated.
With regard to the manufacturing of certain products, medical device companies might need to track environmental and regulatory requirements for each component (e.g., RoHS, WEEE, and REACH). To manage those product specifications, organizations need to be able to identify and rollup how products comply with these specifications. The QMS should help companies remain compliant by helping to track the requirements for each component of a product.
In addition, medical devices require proper training and compliance for more than the products themselves, but also, for the employees of the organization manufacturing the device. A good QMS system should help companies track training records, identify which employees are non-compliant, and update records based on new procedures.
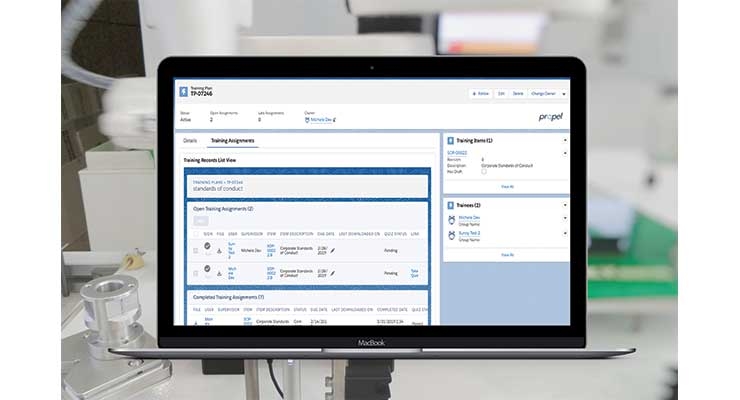
Proper training is key for the safety of customers or patients. Some companies prefer to put training procedures into the learning management system (LMS). This is not a recommended approach. When an LMS is utilized in this context, the training assignments in that system are completely separate from the products, documents, and procedures being updated and versioned. These training assignments should be managed in the QMS system (Figure 3). This missing link between systems creates the potential for training plans that don’t match the latest products and procedures, resulting in non-compliant training and increased risk to the organization and customers.
When a collaborative QMS is implemented, anytime a training protocol is created or updated, appropriate team members should be immediately notified. As key personnel complete the required training, the information is tracked—those who have finished, those having trouble, and those who are delinquent. Further, periodic re-training sessions can be scheduled to ensure everyone is up to date on the latest information, helping a company minimize risk and oversight.
In the medical device industry, audit management is an absolute must so a proper QMS system is required that helps execute good audit management. In order to employ a strong quality management approach, companies should have periodic audits to comply with regulations and ensure product safety, quality, and customer satisfaction. Companies should be able to plan, schedule, and conduct both internal and external audits within the QMS system. Audits can or should include internal processes, supplier procedures, training records, and more.
While many organizations may think any software solution will work to achieve a customer-centric strategy, or they can piece together any set of systems to meet needs, due diligence should be conducted to ensure a good QMS is in place. The benefits of having the right solution for a company can be the difference in achieving success in business, compliance, and training, while also ensuring customers are safe.
What’s Next?
While other industries have begun embracing customers in their product development and lifecycle processes in some shape or form, the healthcare world (specifically the medical device industry) has lagged behind.
The benefits of embracing customer centricity are evident for medical device companies, especially as the industry continues to grow. With the rapidly changing landscape of healthcare and medicine, companies need to start pivoting and embracing customer-focused strategies that make sense for their organizations and their products. Customer centricity requires commitment across the organization, from the firm’s leadership to team members to even the software being implemented. It also requires focus and attention to be paid to each stage of a product’s lifecycle. In order for medical device companies to survive and thrive, they need to focus on customer centricity from the original concept of the medical device to the moment it is being used by the patient. Customer centricity should be a closed loop that puts the patient’s needs and wants at the forefront of product decisions. Companies that employ a customer-centricity strategy set themselves up to experience financial rewards and a happier organization.
References
Allyssa Eclairin is the senior marketing manager at Propel. She is a seasoned designer turned marketer who has worked in the tech and startup world for almost a decade. From cybersecurity to eCommerce to SaaS to even medical devices, Eclairin has continued to work in different markets and hone her craft at digital marketing and design. She enjoys writing relatable content to explain high-level concepts.
Ray Hein is the founder and CEO of Propel. He is a SaaS veteran with 20+ years of PLM, development, and product launch experience in both hardware and enterprise software organizations. Hein served in multiple executive positions at companies including Agile Software, Apttus, Vendavo, and Centric Software.
Social media and patient (customer) satisfaction are pushing companies to embrace and incorporate patient needs and engagement into their product lifecycle, which can be used to create more relevant products for their users. As the industry is projected to grow to an estimated $173 billion in 2019, it makes business sense for companies to embrace this customer-centric strategy going forward.
When it comes to medical devices, such as orthopedics, pacemakers, or even 3D-printed prosthetic limbs, obtaining and retaining patients or customers is vital to maintaining a successful and profitable business. Further, in order to stay competitive and retain customers in any industry, especially the growing and very competitive medical device industry, companies need to implement a customer-centric strategy within the organization.
Customer centricity can be quickly defined as allowing the customer’s or patient’s needs to drive the product offerings. Customer-centric strategies can increase patient satisfaction and help a firm retain those customers going forward despite the emergence of competing products and/or as shifts in healthcare continue to take hold.
Patient satisfaction should be the priority of any organization, regardless of market size. According to a study by McKinsey,2 70 percent of buying experiences are based on how the customer feels they’re being treated. Today’s customers or patients expect to be treated with white gloves. They aren’t just asking for a great “customer experience,” they are asking for products that fit their needs and lifestyle.
Customers want to be heard, as well as properly cared for even after the “sale.” Customer service in medicine (or any industry for that matter) should not be limited to the original “sale.” It must incorporate the entire lifecycle of the product. Many companies have been operating under the notion that customer service and the product experience start and end at the point of purchase of a product or medical device. Rather, the truth is, good product experience and customer service encompass every interaction a customer has with an organization, from the patient’s first introduction to the product through to the point at which it is no longer used. This includes the moment they purchase the medical device to even their experience with a firm’s customer complaint department.
The New Generation of Patients
Today’s new generation of patients don’t just want customized solutions or better experiences, they’re demanding them. Patients and customers are feeling more empowered than ever. From custom-printed prosthetics to IoT-powered precision medical devices, customer expectations are at an all-time high. Further, with non-medical industries dictating change, and consumer subscription services booming to the likes of Netflix, Blue Apron, and other “custom box” services, customers from all types of industries are expecting their other purchasing experiences to operate the same, from their car dealership to grocery stores to even healthcare.
How Does That Look in Healthcare?
No longer are we creating products that fit what someone in a boardroom decided was important for their bottom line or what shareholders suggest a company makes to appease investors. Now, medical device manufacturers must listen to and continuously incorporate patient feedback into the product lifecycle.
What does that mean?
That means engaging and surveying customers or patients in the field. Find out what they want, how they respond to the products and experiences being offered, how they feel about the company, and what needs are not currently being fulfilled.
Then, the patient feedback should be incorporated into the product lifecycle via an automated means—preferably a product lifecycle management (PLM) or quality management system (QMS), and ultimately incorporated into the product lifecycle of future iterations. Patient engagement and feedback should drive future products and work toward eliminating design issues.
3D Printing and Customer-Centricity—A Real-World Example
Additive manufacturing (sometimes referred to as 3D printing) represents a great example of a medical device industry change that has the customer or patient in mind. Instead of taking months to get a metal part (e.g., create a plastic prototype, build out the tool for a metal prototype, and then go to volume production), additive manufacturing can eliminate significant time and costs from the process.
When 3D printing first emerged within the mainstream, few could have predicted how it had the potential to grow as a manufacturing option and revolutionize the medical device industry, let alone almost every other industry in virtually every corner of the world. While some may have viewed 3D printing as a solution for custom toys or trinkets, it is actually a perfect driver of customer-centricity in medicine.
For example, within the healthcare space, 3D printing can be used to produce prosthetic limbs customized to fit a specific patient in a relatively short period of time, especially when compared to more traditional manufacturing methods. Typically, it can be quite common for amputees to wait months or longer for a new prosthetic limb.
CSIRO, Australia’s premier national research organization, partners with Anatomics3 to deliver 3D-printed prosthetics (Figure 1). This novel approach greatly speeds up creation and delivery of new limbs, sternums, and other body parts at a lower premium. The ability to make a custom-designed body part in hours is truly a customer-centric approach to medical devices.
Prescription Glasses Reinvented
Another excellent example of a successful customer-centricity project in healthcare is Warby Parker, an American online retailer of prescription glasses and sunglasses. Warby Parker flipped the traditional model of prescription glasses on its head by serving the patient directly—a desire of prescription glasses users, but one that was going unfulfilled by the industry.
Warby Parker listened and, as a result, discovered the frustration patients found with the existing prescription glasses purchasing model. Customers were seeking affordable options to counter the expensive prescription glasses they were purchasing through traditional methods. The company also found some patients didn’t want to go to a brick-and-mortar store just to buy a pair of glasses.
Warby Parker listened to customers’ feedback and opened an online marketplace to serve as a lower cost, high-quality alternative. Warby Parker even works to serve those who prefer to go into a physical location to try on glasses, but still want a lower cost; the company will send those customers five pairs for free to try on. Still another option for those customers, Warby Parker has set up stores across the country while still offering a lower cost than traditional retailers.
In order to further distinguish itself against competitors, Warby Parker donates a pair of glasses for each pair purchased through its “Buy a Pair, Give a Pair” program. The firm is using its business to help with philanthropic efforts across the world—another example of customer centricity, but on a humanitarian level.
Hearing on Demand by Eargo
Another example of a successful customer-centricity strategy in healthcare is Eargo, an online retailer of beautifully designed hearing aids for the United States. As the population continues to age, hearing loss is affecting more and more people. Like Warby Parker, Eargo established a sales model of selling the company’s offerings—hearing aids—directly to patients, specifically those with mild to moderate hearing loss (Figure 2).
Frustrated by high costs and inconvenience, consumers were unhappy with available buying options and methods. They didn’t want to travel to brick-and-mortar stores; rather, consumers much more preferred shopping from the convenience of their homes. Eargo listened to these requests and responded appropriately. The company sells award-winning hearing aids online and by phone at a reasonable price by cutting out the middlemen.
Healthcare is an undeniable need for all now and perhaps even more so in the future as people begin to live longer and require different devices to enable a better quality of life. Just as Apple and Amazon innovated and, as a result, impacted the world, healthcare and medical device companies will need to consider the models of Warby Parker and Eargo. Medtech firms need to innovate while also listening to the needs and requests of patients and potential customers, instead of trying to be the driver of the technology.
Driving Customer-Centricity
For organizations to successfully employ a customer-centric strategy, they need to do so from the inside out. That means getting each department and team member on board with the methodology. Then, the question remains, “What exactly does that look like?”
Engineering and product teams need to design with the customer’s needs in mind. They need to consider feedback gathered in the field and use that as a guide to dictate product features and materials used. Marketing and sales teams should understand the potential customer and coordinate efforts accordingly. If customers are responding to certain products or offerings, that should drive a product campaign and/or sales channel strategy. Quality teams should gather feedback from customers using a quality management system that allows them to feed complaints and comments back into a CRM for engineers to eliminate product issues and drive iterations and changes of future offerings.
This cycle will create a closed loop for engineering and allow them to create products customers will use and value. Traditionally, engineering has been closed off from feedback from the field, but in a customer-centric model, engineering is incorporated into the conversation and able to address suggested changes and customer comments via the next product version. Further, in a customer-centric model, customer feedback should dictate each phase of the product lifecycle.
The Technology to Drive Customer Centricity
The importance of enabling clear communication between internal teams as well as between the customer (e.g., feedback, complaints) and the company has been well established. Now, it’s vital to ensure the right technology is in place to facilitate this exchange.
Quality management software (QMS) is a “must have” for all medical device companies. Quality management software allows key personnel to manage the quality of the company’s products through various tools and dashboards. In the age of the customer and for those organizations who look to follow a customer-centric strategy, however, a QMS system that provides more than the standard set of features is required.
Just as open collaboration between team members is necessary, so is the same type of functionality in the software solution. A collaborative QMS that seamlessly interfaces with other systems is the first factor to consider. Siloed departmental processes will need to be placed into a secure and robust system. Ideally, the QMS will store customer, product, and quality data all in one place. This enables all customer data to be connected and referenced from one central location, including (and perhaps most importantly) customer complaints and feedback. Whether a company is dealing with those customer complaints, supplier incidents, or non-conformances, there needs to be an integrated process that can quickly identify and resolve issues.
A good QMS solution would ideally eliminate the challenges of manual entry. When a company adopts a customer-centric approach, all customer, product, and quality data should be stored in one centralized location. Manually entering complaints, related items, and feedback from one system to another can lead to errors and incorrect information being attached to the wrong customers or products, or not being transferred at all. Customer feedback or complaints should flow seamlessly into a company’s QMS and PLM systems; engineering can then quickly resolve product issues in the appropriate product for later versions. Only through direct integration between the QMS and PLM systems can a company be certain the right quality issues are being flagged against the correct product revisions and engineering changes.
Another benefit of a single system for quality is that everyone—engineering, operations, and QA—can collaborate on quality management processes such as CAPAs (corrective and preventive actions, or corrective action preventive actions), NCMRs (non-conforming material reports), and SCARs (supplier corrective action requests). A QMS system should help improve compliance across the board, not hinder it. A compliant organization not only helps reduce business risk, it also helps keep customers and patients safe.
An ideal QMS solution should also have pre-configured workflows to help companies reduce errors and remain compliant. With pre-configured workflows, built-in best practices can be established and collaboration is better facilitated across teams. For companies in the medical device industry, quality management systems are held to a much higher bar. Medical device companies need to comply with standards like 21 CFR Part 11 and ensure their systems are fully validated.
With regard to the manufacturing of certain products, medical device companies might need to track environmental and regulatory requirements for each component (e.g., RoHS, WEEE, and REACH). To manage those product specifications, organizations need to be able to identify and rollup how products comply with these specifications. The QMS should help companies remain compliant by helping to track the requirements for each component of a product.
In addition, medical devices require proper training and compliance for more than the products themselves, but also, for the employees of the organization manufacturing the device. A good QMS system should help companies track training records, identify which employees are non-compliant, and update records based on new procedures.
Proper training is key for the safety of customers or patients. Some companies prefer to put training procedures into the learning management system (LMS). This is not a recommended approach. When an LMS is utilized in this context, the training assignments in that system are completely separate from the products, documents, and procedures being updated and versioned. These training assignments should be managed in the QMS system (Figure 3). This missing link between systems creates the potential for training plans that don’t match the latest products and procedures, resulting in non-compliant training and increased risk to the organization and customers.
When a collaborative QMS is implemented, anytime a training protocol is created or updated, appropriate team members should be immediately notified. As key personnel complete the required training, the information is tracked—those who have finished, those having trouble, and those who are delinquent. Further, periodic re-training sessions can be scheduled to ensure everyone is up to date on the latest information, helping a company minimize risk and oversight.
In the medical device industry, audit management is an absolute must so a proper QMS system is required that helps execute good audit management. In order to employ a strong quality management approach, companies should have periodic audits to comply with regulations and ensure product safety, quality, and customer satisfaction. Companies should be able to plan, schedule, and conduct both internal and external audits within the QMS system. Audits can or should include internal processes, supplier procedures, training records, and more.
While many organizations may think any software solution will work to achieve a customer-centric strategy, or they can piece together any set of systems to meet needs, due diligence should be conducted to ensure a good QMS is in place. The benefits of having the right solution for a company can be the difference in achieving success in business, compliance, and training, while also ensuring customers are safe.
What’s Next?
While other industries have begun embracing customers in their product development and lifecycle processes in some shape or form, the healthcare world (specifically the medical device industry) has lagged behind.
The benefits of embracing customer centricity are evident for medical device companies, especially as the industry continues to grow. With the rapidly changing landscape of healthcare and medicine, companies need to start pivoting and embracing customer-focused strategies that make sense for their organizations and their products. Customer centricity requires commitment across the organization, from the firm’s leadership to team members to even the software being implemented. It also requires focus and attention to be paid to each stage of a product’s lifecycle. In order for medical device companies to survive and thrive, they need to focus on customer centricity from the original concept of the medical device to the moment it is being used by the patient. Customer centricity should be a closed loop that puts the patient’s needs and wants at the forefront of product decisions. Companies that employ a customer-centricity strategy set themselves up to experience financial rewards and a happier organization.
References
Allyssa Eclairin is the senior marketing manager at Propel. She is a seasoned designer turned marketer who has worked in the tech and startup world for almost a decade. From cybersecurity to eCommerce to SaaS to even medical devices, Eclairin has continued to work in different markets and hone her craft at digital marketing and design. She enjoys writing relatable content to explain high-level concepts.
Ray Hein is the founder and CEO of Propel. He is a SaaS veteran with 20+ years of PLM, development, and product launch experience in both hardware and enterprise software organizations. Hein served in multiple executive positions at companies including Agile Software, Apttus, Vendavo, and Centric Software.