Sean Fenske, Editor-in-Chief11.13.23
For virtually everyone in the U.S., November marks the beginning of winter, shorter days, decreasing temperatures, and Thanksgiving. For those involved with medical device development and manufacturing, it’s also the month for the Medica and CompaMed trade shows in Dusseldorf, Germany. While last year’s event seemed to bring the crowd’s back following the COVID-induced hiatus, this year may be a more true return to form.
With this in mind, I reached out to a number of companies who will be exhibiting at the show to find out directly from them what they are showing off or focusing on at the event, what challenges customers have brought them, and where they see their role within the industry in aiding medical device manufacturers. Tackling these questions as well as a couple others, Fergus Higgins, vice president, product marketing at Integer, shared a few insights to help you determine if the firm should be a potential services partner for you in 2024 or beyond.
Sean Fenske: What technology or service are you emphasizing at Medica/CompaMed this year?
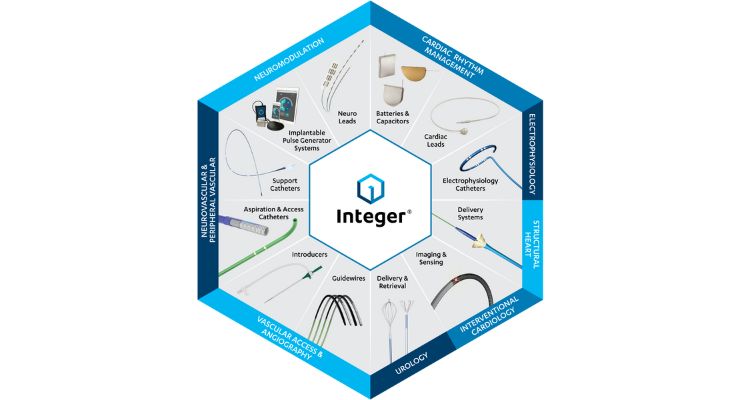
Fergus Higgins: Integer is executing on our European expansion plans announced last year, including investments in an 80,000 sq. ft. expansion of our New Ross, Ireland guidewire manufacturing facility to support growing demand for OEM and custom-developed guidewire products. Also nearing completion is the new Integer Galway, Ireland facility, which will provide customers across Europe with co-located R&D and manufacturing for catheters and delivery systems, as well as specialized capabilities in complex delivery wire grinding and assembly. We continue to develop our team in Galway with skillsets to support customers in our core markets, including neurovascular, structural heart, electrophysiology, and peripheral vascular.
In addition, we continue to expand our capabilities and product offerings in medical textiles and complex braiding, as well as implant covering and coatings such as ePTFE.
These investments in our facilities in Ireland significantly increase our capacity and capabilities to serve our European customer base.
We are also investing in adding capacity to our global network in support of our customers for critical components such as marker bands, electrodes (including proprietary automated Ring on Wire), and other precision machined precious metal components producing best-in-class products for electrophysiology and other core markets.
Fenske: What’s the most common challenge customers inquire about and how do you address it?
Higgins: One common theme witnessed in the industry during the past year is trust in suppliers. Through post-COVID times and recent inflation challenges, Integer has continued to gain market share as a direct result of our efforts to support our customers through these difficult periods.
In fact, despite industry challenges in recent years, Integer has increased investment in capacity and capability each year, specifically to support our customers.
We’re proud that, during these difficult periods, we’ve been able to address our customers’ needs and concerns with candor and transparency.
Fenske: If you could give one piece of advice to companies seeking a manufacturing partner before they make a decision, what would it be?
Higgins: The medical device supplier space is evolving. M&A and ownership changes are accelerating, and competition is increasing with leading players pursuing high-growth market segments and developing a range of capabilities. As the largest player in the market, Integer not only welcomes but is invigorated by this competition.
Companies seeking development or manufacturing partners should consider value provided by suppliers beyond just the sticker price. Key considerations should include, for example, what a company’s track record says about how they will perform and manage a project when it comes to execution and cost management; past performance on quality; service and project execution; and whether the supplier can be trusted to support a project through all stages.
Fenske: What are the forces driving medical device manufacturers to seek your technology/services over doing it in-house?
Higgins: The major drivers are technology and innovation, increasing speed to market, manufacturing scale and capacity, and cost.
Fenske: In what ways is your company able to aid in getting a product (project) to market faster?
Higgins: Integer’s depth and breadth of knowledge in R&D, quality, and regulatory sets us apart from other suppliers in the space. Over the years, Integer has honed our product development process to be flexible and support customers with different approaches, while leveraging our in-house functional teams for support.
Additionally, Integer’s strong core competencies in key components such as precision machined precious metals and stainless steel components, wire and tube processing, specialist extrusions, medical textiles, and complex braided components supports our ability to quickly iterate through development cycles.
Integer is located at Medica/Compamed in Hall 8B, Booth/Stand N29.
With this in mind, I reached out to a number of companies who will be exhibiting at the show to find out directly from them what they are showing off or focusing on at the event, what challenges customers have brought them, and where they see their role within the industry in aiding medical device manufacturers. Tackling these questions as well as a couple others, Fergus Higgins, vice president, product marketing at Integer, shared a few insights to help you determine if the firm should be a potential services partner for you in 2024 or beyond.
Sean Fenske: What technology or service are you emphasizing at Medica/CompaMed this year?
Fergus Higgins: Integer is executing on our European expansion plans announced last year, including investments in an 80,000 sq. ft. expansion of our New Ross, Ireland guidewire manufacturing facility to support growing demand for OEM and custom-developed guidewire products. Also nearing completion is the new Integer Galway, Ireland facility, which will provide customers across Europe with co-located R&D and manufacturing for catheters and delivery systems, as well as specialized capabilities in complex delivery wire grinding and assembly. We continue to develop our team in Galway with skillsets to support customers in our core markets, including neurovascular, structural heart, electrophysiology, and peripheral vascular.
In addition, we continue to expand our capabilities and product offerings in medical textiles and complex braiding, as well as implant covering and coatings such as ePTFE.
These investments in our facilities in Ireland significantly increase our capacity and capabilities to serve our European customer base.
We are also investing in adding capacity to our global network in support of our customers for critical components such as marker bands, electrodes (including proprietary automated Ring on Wire), and other precision machined precious metal components producing best-in-class products for electrophysiology and other core markets.
Fenske: What’s the most common challenge customers inquire about and how do you address it?
Higgins: One common theme witnessed in the industry during the past year is trust in suppliers. Through post-COVID times and recent inflation challenges, Integer has continued to gain market share as a direct result of our efforts to support our customers through these difficult periods.
In fact, despite industry challenges in recent years, Integer has increased investment in capacity and capability each year, specifically to support our customers.
We’re proud that, during these difficult periods, we’ve been able to address our customers’ needs and concerns with candor and transparency.
Fenske: If you could give one piece of advice to companies seeking a manufacturing partner before they make a decision, what would it be?
Higgins: The medical device supplier space is evolving. M&A and ownership changes are accelerating, and competition is increasing with leading players pursuing high-growth market segments and developing a range of capabilities. As the largest player in the market, Integer not only welcomes but is invigorated by this competition.
Companies seeking development or manufacturing partners should consider value provided by suppliers beyond just the sticker price. Key considerations should include, for example, what a company’s track record says about how they will perform and manage a project when it comes to execution and cost management; past performance on quality; service and project execution; and whether the supplier can be trusted to support a project through all stages.
Fenske: What are the forces driving medical device manufacturers to seek your technology/services over doing it in-house?
Higgins: The major drivers are technology and innovation, increasing speed to market, manufacturing scale and capacity, and cost.
Fenske: In what ways is your company able to aid in getting a product (project) to market faster?
Higgins: Integer’s depth and breadth of knowledge in R&D, quality, and regulatory sets us apart from other suppliers in the space. Over the years, Integer has honed our product development process to be flexible and support customers with different approaches, while leveraging our in-house functional teams for support.
Additionally, Integer’s strong core competencies in key components such as precision machined precious metals and stainless steel components, wire and tube processing, specialist extrusions, medical textiles, and complex braided components supports our ability to quickly iterate through development cycles.
Integer is located at Medica/Compamed in Hall 8B, Booth/Stand N29.