Tim Stakenborg, Principal member of technical staff at imec10.03.22
Belgian researchers have developed a new SARS-CoV-2 test that aims to reliably identify positive cases at the point of care. Contrary to existing solutions based on saliva or a nasopharyngeal swab, the new test directly samples viral particles in one’s exhaled breath – making a potential test for infectiousness and highly convenient for frequent (even daily) and widespread use.
Imec developed the solution in collaboration with Belgium’s UZ Leuven University Hospital to conduct the clinical validation.
SARS-CoV-2, the virus at the basis of COVID-19, has had the world in a stranglehold already for 2 years now. With close to 600 million confirmed COVID-19 cases to date (and global mortality numbers exceeding 6 million), its impact on worldwide healthcare systems is enormous. Even after several successful vaccination campaigns, the need for easy-to-use, reliable and rapid SARS-CoV-2 tests will remain for the foreseeable future. However, that might prove a challenge since today’s diagnostic tools to test for SARS-CoV-2 have their drawbacks.
The most sensitive and reliable test—a molecular test—uses the polymerase chain reaction (PCR) to detect the virus’ genetic material in a nasopharyngeal or saliva sample and to quantify the viral load. Molecular tests performed on nasopharyngeal swabs show a viral load that peaks on day 4 or 5. They come with the advantage that only a small amount of viral material is needed to detect the virus. On the downside, the sample collection must be done by trained medical staff and the process is described by many as highly uncomfortable and false-negatives can occur if the test is not taken correctly. Additionally, PCR-tests can remain positive for weeks and even months after infection. Moreover, the test can take up to one day including sample shipment, batch analysis and processing time in a clinical lab.
To counter the longer processing time of PCR tests, several companies have introduced rapid antigen tests that detect the presence of a specific viral protein, which implies a viral infection. Antigen tests are relatively inexpensive and can be used at the point of care. Current authorized devices return results in approximately 15 minutes. Similar to PCR tests, however, most rapid antigen tests are performed on nasopharyngeal or nasal swab specimens, which is perceived as uncomfortable. They are also less reliable than PCR tests. They are especially less sensitive in the early stages of an infection with SARS-CoV-2, i.e., when the viral load is considered the highest.
As a matter of fact, none of those tools can help quickly evaluate whether someone can still pass on the virus. And that is a significant limitation since that should be the very factor to decide when – and for how long – someone should be quarantined.
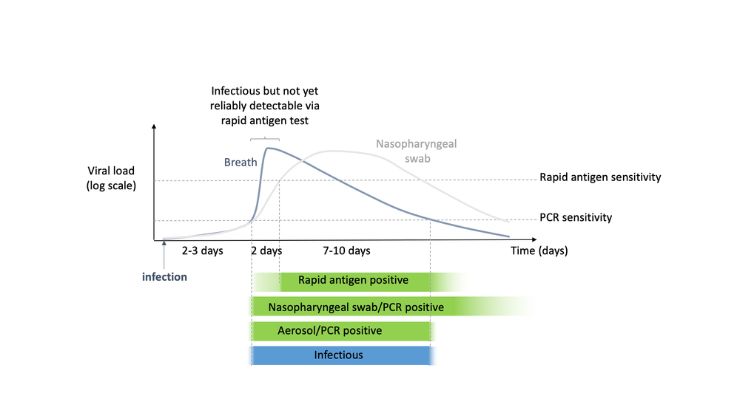
On day 6, X has developed several symptoms and decides to take another rapid antigen test. This time, the amount of viral material in the sample is high enough to result in a positive test – even though X’s immune system is well underway to suppress the virus. Although X has moved beyond his peak transmission and has become less contagious, he or she is required to quarantine in line with today's regulations. And even worse, the actual transmission window has gone by largely unnoticed.
That is why we developed a convenient point-of-care test that is able to confirm whether or not someone is carrying the virus even in the first days after infection and to give an indication of their infectiousness.
Contrary to rapid antigen tests, our test is based on the highly reliable PCR methods used in clinical labs. Our test delivers results in less than twenty minutes taking into account a sampling time of less than a minute and a subsequent fast PCR of under 15 min. We already have results that the PCR time can be lowered to less than 5 min. Unlike current tests – which work with saliva or a nasopharyngeal swab – our test detects viral particles in one’s exhaled breath. As such, there is a substantially lower threshold for using our test. This means our solution can be used on a more frequent and even on a daily basis.
Research has revealed that SARS-CoV-2 virus particles in one’s exhaled breath, cough or sneeze – called aerosols – appear to be the virus’ dominant transmission method, and that is a significant finding. For although resorting to one’s exhaled breath is perhaps the most logical and convenient way of testing, it is a sampling method that has largely been ignored so far. Moreover, the direct quantification of viral shedding in exhaled particles can potentially measure contagiousness directly.
A large longitudinal study shows that our new technology is on par with the golden-standard nasopharyngeal-based PCR tests and even supersedes the rapid antigen-tests in the first week of infection. The exhaled viral load – the purported measurement of contagiousness – peaks before the antigen tests are reliably positive. This would imply that people can spread the virus early on in the infection, even when they just performed a negative self-test. The breath-based test is also the first to consistently report a negative result, suggesting that the person is not infectious anymore.
Thanks to our technology, I am convinced that it will become a lot easier to test for SARS-CoV-2 faster, more comfortably, and on a much larger scale. Since SARS-CoV-2 is believed to spread mostly by means of exhaled particles, the exhaled viral load may also allow for a more accurate measurement of the actual transmission window. Moreover, we are designing our test to flexibly cope with the rise of other viruses and germs that spread via exhaled particles – such as influenza, RSV, and tuberculosis.
The sample collector’s function is twofold: to capture the aerosol (and virus) particles in one’s exhaled breath and to allow for PCR directly on the same collector.
Designing such a solution using conventional technologies is extremely hard to do – but it works using silicon. We created a chip featuring thousands of microscale PCR cavities that that capture the viral particles from exhaled breath.
When the sample collector is inserted into the analysis unit, reagents are added fully automatically to collect the virus present in the aerosol particles. Next, the necessary heat cycles are generated to facilitate the thermal PCR reaction, and to measure the amount of viral material. Our current PCR takes 15 min. The process is fully automated, cutting the time from sample to result to less than 20 minutes. First results also indicate it can be sped up to 5 minutes – compared to the 30 minutes required by conventional lab solutions. Key to this innovation is silicon technology and because the processes are standardized the tests can be produced on a large scale and at a relatively low cost.
Tim Stakenborg graduated in 1998 at the University of Leuven as a master in engineering in chemistry and biochemistry. After being a project engineer at Alcon-Novartis and his PhD in molecular biology, he joined imec. At imec, Stakenborg is currently a principal member of the technical staff and group leader in the life science technology department. He has been a project lead in several funded and industrial research projects especially focusing on merging biology with technology. In his current role of group leader, he is heavily involved in the transfer and integration of key technical components in fully functional devices.
Imec developed the solution in collaboration with Belgium’s UZ Leuven University Hospital to conduct the clinical validation.
SARS-CoV-2, the virus at the basis of COVID-19, has had the world in a stranglehold already for 2 years now. With close to 600 million confirmed COVID-19 cases to date (and global mortality numbers exceeding 6 million), its impact on worldwide healthcare systems is enormous. Even after several successful vaccination campaigns, the need for easy-to-use, reliable and rapid SARS-CoV-2 tests will remain for the foreseeable future. However, that might prove a challenge since today’s diagnostic tools to test for SARS-CoV-2 have their drawbacks.
Current SARS-CoV-2 Tests
There are two types of SARS-CoV-2 tests available today that detect the virus: molecular (DNA) tests and antigen (protein) tests.The most sensitive and reliable test—a molecular test—uses the polymerase chain reaction (PCR) to detect the virus’ genetic material in a nasopharyngeal or saliva sample and to quantify the viral load. Molecular tests performed on nasopharyngeal swabs show a viral load that peaks on day 4 or 5. They come with the advantage that only a small amount of viral material is needed to detect the virus. On the downside, the sample collection must be done by trained medical staff and the process is described by many as highly uncomfortable and false-negatives can occur if the test is not taken correctly. Additionally, PCR-tests can remain positive for weeks and even months after infection. Moreover, the test can take up to one day including sample shipment, batch analysis and processing time in a clinical lab.
To counter the longer processing time of PCR tests, several companies have introduced rapid antigen tests that detect the presence of a specific viral protein, which implies a viral infection. Antigen tests are relatively inexpensive and can be used at the point of care. Current authorized devices return results in approximately 15 minutes. Similar to PCR tests, however, most rapid antigen tests are performed on nasopharyngeal or nasal swab specimens, which is perceived as uncomfortable. They are also less reliable than PCR tests. They are especially less sensitive in the early stages of an infection with SARS-CoV-2, i.e., when the viral load is considered the highest.
As a matter of fact, none of those tools can help quickly evaluate whether someone can still pass on the virus. And that is a significant limitation since that should be the very factor to decide when – and for how long – someone should be quarantined.
Imecs breathalyzer (Dutch subs) from imec on Vimeo.
Starting point: The Typical Course of a SARS-CoV-2 Infection
Contrary to what most people think, the SARS-CoV-2 virus can only be passed for a limited period of time. Let us have a look at a concrete example. In Figure 1, person X gets infected on day 0. X has an important meeting and takes a precautionary self-test that same day. However, as the number of viral particles in the sample (viral load) still ranges below the lower detection limit, X gets presented with a negative test result. Still, shortly after that, X enters the period in which the viral load soars and in which a person can pass the virus to others (the transmission window).On day 6, X has developed several symptoms and decides to take another rapid antigen test. This time, the amount of viral material in the sample is high enough to result in a positive test – even though X’s immune system is well underway to suppress the virus. Although X has moved beyond his peak transmission and has become less contagious, he or she is required to quarantine in line with today's regulations. And even worse, the actual transmission window has gone by largely unnoticed.
That is why we developed a convenient point-of-care test that is able to confirm whether or not someone is carrying the virus even in the first days after infection and to give an indication of their infectiousness.
Contrary to rapid antigen tests, our test is based on the highly reliable PCR methods used in clinical labs. Our test delivers results in less than twenty minutes taking into account a sampling time of less than a minute and a subsequent fast PCR of under 15 min. We already have results that the PCR time can be lowered to less than 5 min. Unlike current tests – which work with saliva or a nasopharyngeal swab – our test detects viral particles in one’s exhaled breath. As such, there is a substantially lower threshold for using our test. This means our solution can be used on a more frequent and even on a daily basis.
Detecting SARS-CoV-2 Virus Particles in One’s Exhaled Breath
It is a well-known fact that respiratory diseases – such as SARS-CoV-2 – are typically spread via respiratory droplets formed during sneezing, coughing and even normal breathing.Research has revealed that SARS-CoV-2 virus particles in one’s exhaled breath, cough or sneeze – called aerosols – appear to be the virus’ dominant transmission method, and that is a significant finding. For although resorting to one’s exhaled breath is perhaps the most logical and convenient way of testing, it is a sampling method that has largely been ignored so far. Moreover, the direct quantification of viral shedding in exhaled particles can potentially measure contagiousness directly.
A large longitudinal study shows that our new technology is on par with the golden-standard nasopharyngeal-based PCR tests and even supersedes the rapid antigen-tests in the first week of infection. The exhaled viral load – the purported measurement of contagiousness – peaks before the antigen tests are reliably positive. This would imply that people can spread the virus early on in the infection, even when they just performed a negative self-test. The breath-based test is also the first to consistently report a negative result, suggesting that the person is not infectious anymore.
Thanks to our technology, I am convinced that it will become a lot easier to test for SARS-CoV-2 faster, more comfortably, and on a much larger scale. Since SARS-CoV-2 is believed to spread mostly by means of exhaled particles, the exhaled viral load may also allow for a more accurate measurement of the actual transmission window. Moreover, we are designing our test to flexibly cope with the rise of other viruses and germs that spread via exhaled particles – such as influenza, RSV, and tuberculosis.
Columbus’ Egg: Harnessing the Power of Silicon
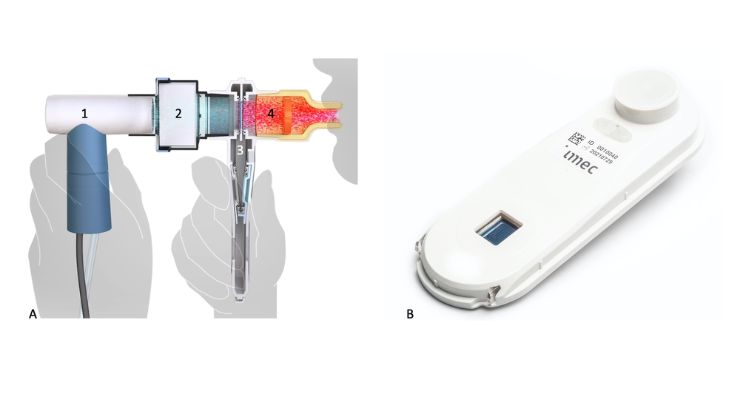
Imec has developed a prototype to demonstrate an integrated workflow. It consists of a sample collector and an analysis unit.The sample collector’s function is twofold: to capture the aerosol (and virus) particles in one’s exhaled breath and to allow for PCR directly on the same collector.
Designing such a solution using conventional technologies is extremely hard to do – but it works using silicon. We created a chip featuring thousands of microscale PCR cavities that that capture the viral particles from exhaled breath.
When the sample collector is inserted into the analysis unit, reagents are added fully automatically to collect the virus present in the aerosol particles. Next, the necessary heat cycles are generated to facilitate the thermal PCR reaction, and to measure the amount of viral material. Our current PCR takes 15 min. The process is fully automated, cutting the time from sample to result to less than 20 minutes. First results also indicate it can be sped up to 5 minutes – compared to the 30 minutes required by conventional lab solutions. Key to this innovation is silicon technology and because the processes are standardized the tests can be produced on a large scale and at a relatively low cost.
Want to Know More?
The breath sampler technology and science behind it are presented in three recent papers:- Stakenborg T et al., Molecular detection of SARS-CoV-2 in exhaled breath using a portable sampler Biosensors and Bioelectronics (2022). https://doi.org/10.1016/j.bios.2022.114663
- Raymenants J et al., Exhaled breath SARS-CoV-2 shedding patterns across variants of concern, International Journal of Infectious Diseases (2022). https://doi.org/10.1016/j.ijid.2022.07.069
- Paeps F et al., Development of an integrated breath analysis technology for on-chip aerosol capture and molecular analysis. MNE Eurosensors 2022 conference paper (2022).
Tim Stakenborg graduated in 1998 at the University of Leuven as a master in engineering in chemistry and biochemistry. After being a project engineer at Alcon-Novartis and his PhD in molecular biology, he joined imec. At imec, Stakenborg is currently a principal member of the technical staff and group leader in the life science technology department. He has been a project lead in several funded and industrial research projects especially focusing on merging biology with technology. In his current role of group leader, he is heavily involved in the transfer and integration of key technical components in fully functional devices.