Jason Middleton, VP of Sales & Development, Ray Products07.10.19
As the medtech industry continually evolves, so must the plastics manufacturing industry that supports it. Ray Products frequently works with partners in the medtech space to create a wide array of medical supplies and devices; and, as a result, must adapt their manufacturing approaches to meet the industry’s ever-changing needs. That’s why each year, Ray Products conducts an annual industry-wide survey asking thousands of professionals about their preferred processes, customers, and more. Now in its fifth year, the survey has proven to be an excellent tool for learning about the landscape of the current industry and what’s to come.
Survey respondents hold titles in engineering, manufacturing, quality, purchasing and industrial design from diverse industries such as medical device manufacturing, transportation, green energy and contract manufacturing. This year, medical device manufacturers made up just over half of survey respondents.
Medical Device Manufacturers Prefer Domestic
Last year’s survey told us that medical device manufacturers are more likely to use domestic manufacturing than other plastics manufacturing customers. They are also more likely to require a prototype prior to production than engineers in other industries.
Process Diversity Is on the Rise
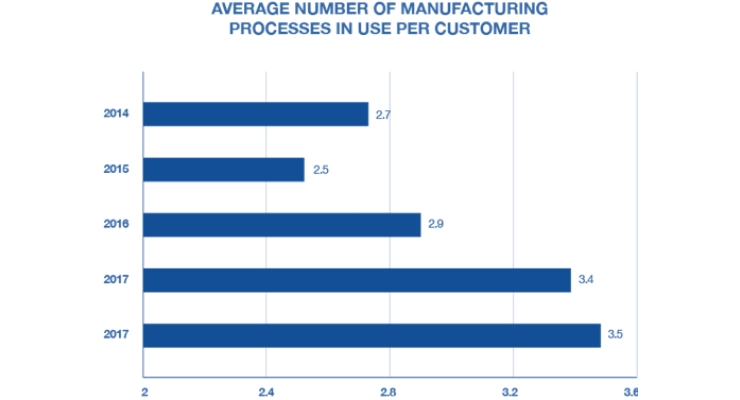
This year’s survey found that process diversity—or using multiple types of plastics manufacturing processes, like pressure forming, injection molding, RIM, and fiberglass—is higher than ever. This is likely because manufacturers are realizing that there is no “one-size-fits-all” approach when it comes to developing high-quality, cost-effective plastic enclosures.
For example, pressure forming (one of two thermoforming processes) and injection molding are typically presented as competing options. And while there are some instances in which one process is better than the other—the large-part capability of pressure forming, and mid-volume production is ideal for many device manufacturers—there are instances when the two processes can be used for different elements of the same project to maximize cost efficiency and maintain excellent quality.
The Future of Reshoring
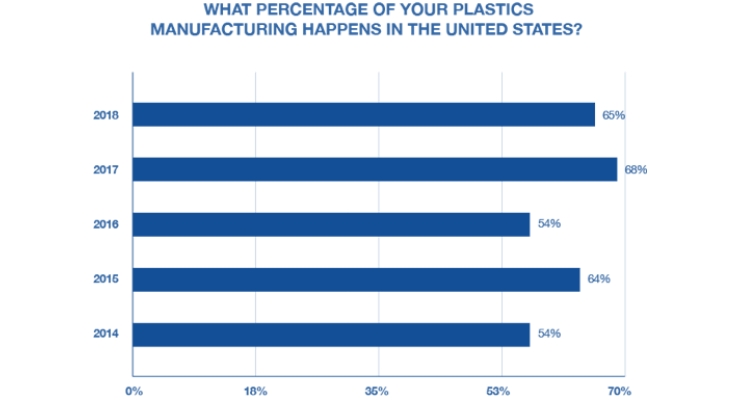
This year’s survey also found that 65% of respondents’ plastics manufacturing is based in the U.S. While this percentage is slightly lower than the previous year (68%), it has grown significantly since 2016. Historically, outsourcing plastics manufacturing has made sense economically; however, given the ongoing trade war with China, it seems likely that plastics manufacturers may be reshoring their processes in the coming year.
As medical device manufacturers continue to develop innovative equipment and products for the medical community, the insights and expertise of customized plastics manufacturers like Ray Products will be increasingly important and useful.
Jason Middleton is vice president of Sales & Development at Ray Products, a custom heavy gauge thermoforming manufacturer founded in 1949 and located in Ontario, California. With more than 15 years of plastics manufacturing industry experience, Middleton plans and executes successful pressure forming and vacuum forming projects for clients in a wide range of industries.
Survey respondents hold titles in engineering, manufacturing, quality, purchasing and industrial design from diverse industries such as medical device manufacturing, transportation, green energy and contract manufacturing. This year, medical device manufacturers made up just over half of survey respondents.
Medical Device Manufacturers Prefer Domestic
Last year’s survey told us that medical device manufacturers are more likely to use domestic manufacturing than other plastics manufacturing customers. They are also more likely to require a prototype prior to production than engineers in other industries.
Process Diversity Is on the Rise
This year’s survey found that process diversity—or using multiple types of plastics manufacturing processes, like pressure forming, injection molding, RIM, and fiberglass—is higher than ever. This is likely because manufacturers are realizing that there is no “one-size-fits-all” approach when it comes to developing high-quality, cost-effective plastic enclosures.
For example, pressure forming (one of two thermoforming processes) and injection molding are typically presented as competing options. And while there are some instances in which one process is better than the other—the large-part capability of pressure forming, and mid-volume production is ideal for many device manufacturers—there are instances when the two processes can be used for different elements of the same project to maximize cost efficiency and maintain excellent quality.
The Future of Reshoring
This year’s survey also found that 65% of respondents’ plastics manufacturing is based in the U.S. While this percentage is slightly lower than the previous year (68%), it has grown significantly since 2016. Historically, outsourcing plastics manufacturing has made sense economically; however, given the ongoing trade war with China, it seems likely that plastics manufacturers may be reshoring their processes in the coming year.
As medical device manufacturers continue to develop innovative equipment and products for the medical community, the insights and expertise of customized plastics manufacturers like Ray Products will be increasingly important and useful.
Jason Middleton is vice president of Sales & Development at Ray Products, a custom heavy gauge thermoforming manufacturer founded in 1949 and located in Ontario, California. With more than 15 years of plastics manufacturing industry experience, Middleton plans and executes successful pressure forming and vacuum forming projects for clients in a wide range of industries.