Ajay Devgire, Senior Research Analyst, Global Market Insights03.14.19
The global medical device vigilance market will surpass $91.5 billion by 2025, according to a new research report by Global Market Insights. Demand for medical device vigilance systems is increasing over recent years owing to a surge in the number of adverse events reported related to medical devices. High adoption of medical device vigilance systems to ensure the safety of patients, healthcare professionals, and other users will positively impact business growth. Furthermore, strict government regulations for manufacturers to develop safe and highly efficient devices will favor the medical device vigilance industry’s growth.
Companies’ focus on improvising existing medical device vigilance software will increase its adoption. Growing R&D investments by key industry players to develop technologically advanced vigilance systems will further augment business growth. Availability of vigilance software and increasing awareness among patients will also boost growth. Patient access to adverse event reporting that further enables exporting of files and publishing regulatory reports as well as handling daily medical device vigilance requirements should favor the medical device vigilance market’s growth. However, the failure of certain manufacturing companies to ensure product safety may impede the medical device vigilance business growth during the forthcoming years.
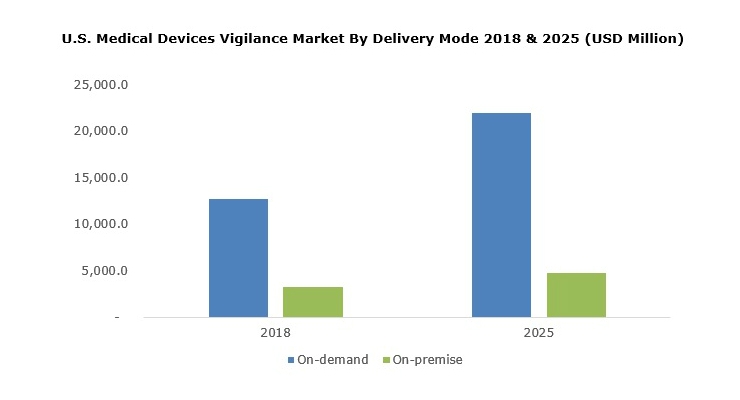
On-demand delivery mode segment dominated the medical device vigilance market with 80.2 percent revenue share in 2018 and is estimated to witness a similar trend over the forecast period. Segmental growth is attributed to companies’ growing focus on the development of a cloud-based software system to ensure the quality of medical devices. For instance, AssurX Cloud QMS developed by AssurX offers a quick, secure, and reliable quality management software solution over the internet.
The therapeutic application segment held significant revenue in 2018 and is estimated to show the fastest CAGR (8.9 percent) over the analysis years. The growing demand to ensure the safety of therapeutic medical devices exerting local effects such as wound covering, tissue cutting, or supporting open clogged arteries will surge adoption of medical device vigilance systems, thereby driving segmental growth.
The clinical research organizations (CROs) segment was valued at $6.1 billion in 2018 and is projected to grow considerably over the estimated timeframe. CROs offer real-time and accurate safety management of medical devices in all phases of development, including pre- and post-market studies. The ability of CROs to communicate directly with manufacturers, physicians, regulatory authorities, and clients to ensure full control at every stage of the project will upsurge segmental growth in the foreseeable future.
The U.S. medical device vigilance market dominated the North America region with more than 89 percent revenue share in 2018 and is projected to witness similar growth in the foreseeable future. High growth can be attributed to the rising number of medical device adverse events reported in the country. As per the FDA, more than 1 million adverse events related to medical devices were reported in the U.S. in 2015. Such factors will increase the adoption of medical device vigilance systems, thereby augmenting business growth.
The UK medical device vigilance market is anticipated to witness the fastest CAGR of 9.5 percent over the analysis timeframe. In line with the UK medical device regulations, the Medicines and Healthcare products Regulatory Agency (MHRA) strictly investigates any complaints about products with or without CE mark, examines manufacturing facilities in case of breach of regulations, and investigates results of vigilance reports. Favorable regulatory policies in the country will further spur the market size.
Some of the key industry players operating in the medical device vigilance market include AssurX, Oracle, AB-Cube, Sparta Systems, Xybion, Sarjen Systems, ZEINCRO, MDI Consultants, INTEL, Omnify Software, and Numerix. Industry players focus their efforts on new software development and strategic initiatives including collaborations, acquisitions, and partnerships that help them in strengthening market position. For instance, AB-Cube introduced SafetyEasy MD, a medical device vigilance software dedicated to the management of medical device adverse events, broadening the company’s customer base and strengthening its market position.
Ajay Devgire is a senior research analyst at Global Market Insights with a professional experience of over 4 years in Healthcare Market research industry. His work is focused on identifying growth opportunities in the life science and healthcare industry. He is well- adept in primary and secondary research; qualitative and quantitative studies; continuous research and market monitoring. Market assessment, data analysis, and growth forecasting are some of Ajay’s major work areas. Over the years, Ajay has gained expertise in veterinary healthcare, medical devices, and healthcare IT markets. Prior to working with Global Market Insights, Ajay has worked as a trainee analyst and been a part of various assignments in IMS Health (now IQVIA); post which he worked as an analyst at Mordor Intelligence.
Companies’ focus on improvising existing medical device vigilance software will increase its adoption. Growing R&D investments by key industry players to develop technologically advanced vigilance systems will further augment business growth. Availability of vigilance software and increasing awareness among patients will also boost growth. Patient access to adverse event reporting that further enables exporting of files and publishing regulatory reports as well as handling daily medical device vigilance requirements should favor the medical device vigilance market’s growth. However, the failure of certain manufacturing companies to ensure product safety may impede the medical device vigilance business growth during the forthcoming years.
On-demand delivery mode segment dominated the medical device vigilance market with 80.2 percent revenue share in 2018 and is estimated to witness a similar trend over the forecast period. Segmental growth is attributed to companies’ growing focus on the development of a cloud-based software system to ensure the quality of medical devices. For instance, AssurX Cloud QMS developed by AssurX offers a quick, secure, and reliable quality management software solution over the internet.
The therapeutic application segment held significant revenue in 2018 and is estimated to show the fastest CAGR (8.9 percent) over the analysis years. The growing demand to ensure the safety of therapeutic medical devices exerting local effects such as wound covering, tissue cutting, or supporting open clogged arteries will surge adoption of medical device vigilance systems, thereby driving segmental growth.
The clinical research organizations (CROs) segment was valued at $6.1 billion in 2018 and is projected to grow considerably over the estimated timeframe. CROs offer real-time and accurate safety management of medical devices in all phases of development, including pre- and post-market studies. The ability of CROs to communicate directly with manufacturers, physicians, regulatory authorities, and clients to ensure full control at every stage of the project will upsurge segmental growth in the foreseeable future.
The U.S. medical device vigilance market dominated the North America region with more than 89 percent revenue share in 2018 and is projected to witness similar growth in the foreseeable future. High growth can be attributed to the rising number of medical device adverse events reported in the country. As per the FDA, more than 1 million adverse events related to medical devices were reported in the U.S. in 2015. Such factors will increase the adoption of medical device vigilance systems, thereby augmenting business growth.
The UK medical device vigilance market is anticipated to witness the fastest CAGR of 9.5 percent over the analysis timeframe. In line with the UK medical device regulations, the Medicines and Healthcare products Regulatory Agency (MHRA) strictly investigates any complaints about products with or without CE mark, examines manufacturing facilities in case of breach of regulations, and investigates results of vigilance reports. Favorable regulatory policies in the country will further spur the market size.
Some of the key industry players operating in the medical device vigilance market include AssurX, Oracle, AB-Cube, Sparta Systems, Xybion, Sarjen Systems, ZEINCRO, MDI Consultants, INTEL, Omnify Software, and Numerix. Industry players focus their efforts on new software development and strategic initiatives including collaborations, acquisitions, and partnerships that help them in strengthening market position. For instance, AB-Cube introduced SafetyEasy MD, a medical device vigilance software dedicated to the management of medical device adverse events, broadening the company’s customer base and strengthening its market position.
Ajay Devgire is a senior research analyst at Global Market Insights with a professional experience of over 4 years in Healthcare Market research industry. His work is focused on identifying growth opportunities in the life science and healthcare industry. He is well- adept in primary and secondary research; qualitative and quantitative studies; continuous research and market monitoring. Market assessment, data analysis, and growth forecasting are some of Ajay’s major work areas. Over the years, Ajay has gained expertise in veterinary healthcare, medical devices, and healthcare IT markets. Prior to working with Global Market Insights, Ajay has worked as a trainee analyst and been a part of various assignments in IMS Health (now IQVIA); post which he worked as an analyst at Mordor Intelligence.