Peter Rose, Managing Director, Maetrics03.19.18
In May 2017, the new Medical Device Regulation (MDR) was published by the European Parliament. Not only is the MDR a revision of earlier Directives, but it was drafted with the aim of increasing the level of public health and safety by ensuring a transparent regulatory framework. The new regulation is an extremely complex document that will have a significant impact on medtech manufacturers and it is quite apparent manufacturers cannot afford to delay in complying.
The MDR is designed to improve both clinical safety and fair market access for all medtech players. The specific changes will affect:
It is understandable that manufacturers may be lured into a false sense of security by thinking the transition timeframe to completely comply with the new Regulation leaves a considerable amount of time to do so. However, this timescale has not incorporated the lack of available, suitably trained consultants to assist with the preparatory process for compliance nor does it address the diminishing amount of Notified Bodies (NBs) and the effect this will have on certification completion.
There is widespread rhetoric about the concerning lack of NBs to service the medtech industry’s requirements. Some NBs have advised that they are not currently providing quotations for CE marking services to new clients, and others are quoting extremely lengthy lead times for new CE mark clients. NBs are themselves under serious pressure from their Competent Authorities (CA) to heighten scrutiny of their registered medical device manufacturers. This is, in part, due to a number of notable safety issues that significantly contributed to the drafting of the new MDR.
In Europe, the number of NBs has reduced by a staggering 20 percent over the last two years and it will come as no surprise to some that this will only continue to increase as the new MDR is rolled out. There will be an increased demand on their services, which means manufacturers have to be time savvy and factor in long wait times for their NB to help them. To escalate this issue further, unannounced inspections are becoming mandatory and will have to take place every five years—another hindrance to an NB’s capacity to audit and certify its clients.
Capacity issues don’t just sit with the NBs but with the life sciences industry as a whole, whereby there is a highly publicized skills gap. The same is true in medtech all around the world. In fact, research shows that up to 80 percent of clinical research professionals work freelance for contract research organizations rather than companies1. This results in a catch-22 effect with the market lacking clinical research associates with enough experience or a wide-enough set of skills. This is especially poignant as widespread use of consultants became popular 10 to 15 years ago with many experienced researchers now retiring, the rate of clinical trials increasing, and the new burden of MDR compliance adding to the pressure.
Capacity issues are certainly an aspect that offers organized and prepared “early MDR adopters” market competitive advantage. There is going to be significant market access problems for manufacturers who delay in their preparations as the NBs will not be able to keep up and the top talent preparatory consultants will already be contracted. With this comes a hidden advantage for the early mover (MDR compliance pioneers); they will be equipped and certified ready to service the market.
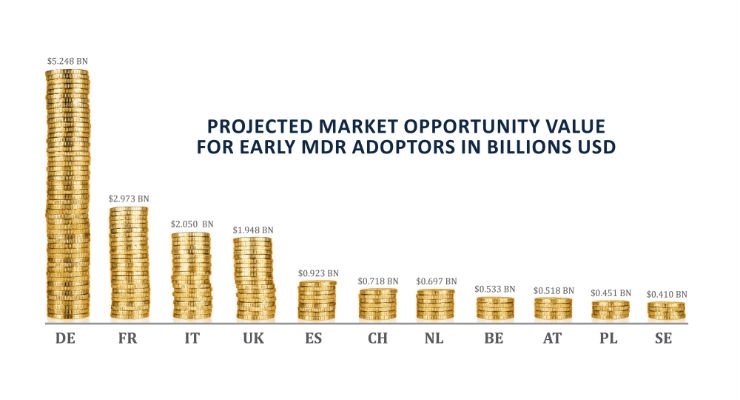
At Maetrics, we have drawn upon interviews with industry analysts and key players to build a model that demonstrates the Market Opportunity Value (MOV) that early compliance presents. It is essentially an estimate value on the medical device market that late-moving players could miss out on for the entire period they are restricted from trading while they wait to become MDR compliant. For pioneers, the MOV represents potential additional annual revenues until slow-moving competitors can come up to speed with compliance.
To investigate this further, an MOV model that demonstrates the potential annual revenues achievable for MDR-compliant manufacturers, or conversely, the scale of the market penalty for non-compliance is available. The figure sits at a staggering $16.5 billion2. This is based on the estimate—corroborated by interviews with major medtech businesses operating in Europe—that compliance under-capacity will be 20 percent of total market value.
The new MDR is going to be affecting so many critical processes for medical device manufacturers that it is critical manufacturers implement strategies to adopt compliance early. It is apparent there are going to be capacity bottlenecks, both from the NBs and the surging demand from compliance professionals, which will force late adopters to delay in complying. This will result in some market suppliers being unable to service their European markets, while the market gap is filled by early movers, thereby gaining access to additional Market Value, which they may well retain even after the late adopters are fully compliant.
References
1 Clinical Leader, Clinical Staff Shortage: “Growing Plague” For Pharma & CROs, 6th November 2016
2 The total market values from which it is derived are based on the latest verified medical market value estimates from a variety of established analysts. The countries selected represent the top ten EU countries by nominal GDP, plus Switzerland.
The MDR is designed to improve both clinical safety and fair market access for all medtech players. The specific changes will affect:
- Reclassification, including legacy products
- Reprocessing of single use devices
- Technical documentation standards
- Clinical evaluation/evidence standards
- Vigilance and post-market surveillance
- Mandatory product liability insurance
- Transparency through EUDAMED
- Labeling and the supply chain
- Unique device identification (UDI)
It is understandable that manufacturers may be lured into a false sense of security by thinking the transition timeframe to completely comply with the new Regulation leaves a considerable amount of time to do so. However, this timescale has not incorporated the lack of available, suitably trained consultants to assist with the preparatory process for compliance nor does it address the diminishing amount of Notified Bodies (NBs) and the effect this will have on certification completion.
There is widespread rhetoric about the concerning lack of NBs to service the medtech industry’s requirements. Some NBs have advised that they are not currently providing quotations for CE marking services to new clients, and others are quoting extremely lengthy lead times for new CE mark clients. NBs are themselves under serious pressure from their Competent Authorities (CA) to heighten scrutiny of their registered medical device manufacturers. This is, in part, due to a number of notable safety issues that significantly contributed to the drafting of the new MDR.
In Europe, the number of NBs has reduced by a staggering 20 percent over the last two years and it will come as no surprise to some that this will only continue to increase as the new MDR is rolled out. There will be an increased demand on their services, which means manufacturers have to be time savvy and factor in long wait times for their NB to help them. To escalate this issue further, unannounced inspections are becoming mandatory and will have to take place every five years—another hindrance to an NB’s capacity to audit and certify its clients.
Capacity issues don’t just sit with the NBs but with the life sciences industry as a whole, whereby there is a highly publicized skills gap. The same is true in medtech all around the world. In fact, research shows that up to 80 percent of clinical research professionals work freelance for contract research organizations rather than companies1. This results in a catch-22 effect with the market lacking clinical research associates with enough experience or a wide-enough set of skills. This is especially poignant as widespread use of consultants became popular 10 to 15 years ago with many experienced researchers now retiring, the rate of clinical trials increasing, and the new burden of MDR compliance adding to the pressure.
Capacity issues are certainly an aspect that offers organized and prepared “early MDR adopters” market competitive advantage. There is going to be significant market access problems for manufacturers who delay in their preparations as the NBs will not be able to keep up and the top talent preparatory consultants will already be contracted. With this comes a hidden advantage for the early mover (MDR compliance pioneers); they will be equipped and certified ready to service the market.
At Maetrics, we have drawn upon interviews with industry analysts and key players to build a model that demonstrates the Market Opportunity Value (MOV) that early compliance presents. It is essentially an estimate value on the medical device market that late-moving players could miss out on for the entire period they are restricted from trading while they wait to become MDR compliant. For pioneers, the MOV represents potential additional annual revenues until slow-moving competitors can come up to speed with compliance.
To investigate this further, an MOV model that demonstrates the potential annual revenues achievable for MDR-compliant manufacturers, or conversely, the scale of the market penalty for non-compliance is available. The figure sits at a staggering $16.5 billion2. This is based on the estimate—corroborated by interviews with major medtech businesses operating in Europe—that compliance under-capacity will be 20 percent of total market value.
The new MDR is going to be affecting so many critical processes for medical device manufacturers that it is critical manufacturers implement strategies to adopt compliance early. It is apparent there are going to be capacity bottlenecks, both from the NBs and the surging demand from compliance professionals, which will force late adopters to delay in complying. This will result in some market suppliers being unable to service their European markets, while the market gap is filled by early movers, thereby gaining access to additional Market Value, which they may well retain even after the late adopters are fully compliant.
References
1 Clinical Leader, Clinical Staff Shortage: “Growing Plague” For Pharma & CROs, 6th November 2016
2 The total market values from which it is derived are based on the latest verified medical market value estimates from a variety of established analysts. The countries selected represent the top ten EU countries by nominal GDP, plus Switzerland.