Maria Shepherd, President and Founder, Medi-Vantage02.28.24
You may not have been able to experience the 100,000-plus visitors to the Consumer Electronics Show (CES) in Las Vegas this year (after all, it conflicted with the JP Morgan Healthcare meeting—a grave mistake in my eyes), but do not despair, the reporting on healthcare devices is substantial. Most of the products at CES are over-promoted items that the majority of us really don’t need. There are, however, medtech pearls in the mix.
A Reusable COVID-19 Breath Analyzer
The past five years have witnessed a mounting worldwide concern over respiratory virus outbreaks, underscoring the critical need for rapid and accurate screening, diagnostics, and treatment strategies for virus-related diseases.2 Since COVID isn’t going away any time soon, new diagnostic technology from Opteev—the ViraWarn—is (according to the manufacturer) the first affordable respiratory virus detector for personal spaces.3
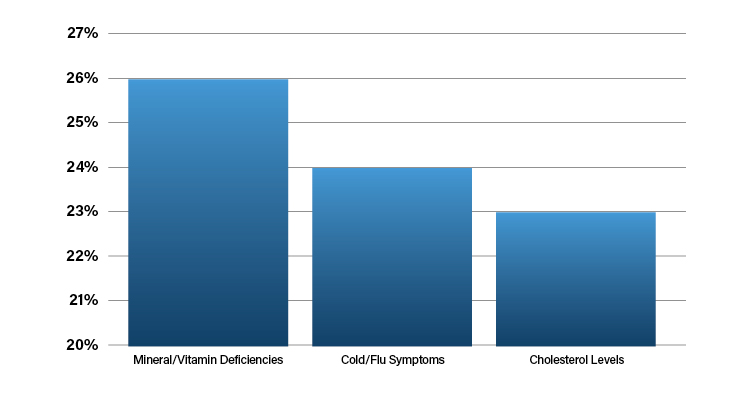
According to McKinsey, the pandemic made at-home testing kits a household item.4 Now, patients accustomed to home testing for COVID are showing more interest in other types of at-home kits (Table 2).
Patients have learned at-home diagnostic tests offer greater convenience than going to a doctor’s office, rapid results, and the ability to test frequently. Barriers to adoption of at-home diagnostics include a preference to see a doctor in person, poorly understood need, and price.
The current device from Opteev is a tabletop device that monitors personal or office spaces while discreetly and rapidly alerting users of the presence of respiratory viruses, including COVID, RSV, and influenza. A purse or pocket-sized breath analyzer that can detect COVID-19, RSV, and influenza in under 60 seconds using artificial intelligence is also in development. With the handheld device, users blow twice into the device and receive a positive or negative result through a red or green LED light. The handheld device is still awaiting FDA clearance. It is reusable and projected to cost approximately $99. Cartridge replacements will be priced at a few dollars each, continuing the focus on affordability cited by the company.
The Holy Grail of Blood Pressure Monitoring
According to the Mayo Clinic, some wrist blood pressure monitors will be accurate if used by patients exactly as directed and checked against measurements taken in the provider's office. However, the AHA recommends using the traditional blood pressure monitoring cuff-based device that goes around the upper arm. This may not work for patients with very large arms or those that do not have access to a blood pressure monitor with an arm cuff with a good fit.5
Enter Valencell, the developer of a fingertip blood pressure monitor that looks like an SpO2 monitor and is claimed by the company to be the world’s first cuffless blood pressure device using patented biometric sensor technology.6 The firm is a digital health company that uses sensors from Samsung and Bose, as well as leveraging more than 100 wearable biomedical sensors attributed to the CEO of Valencell. Its key technology utilizes an algorithm that measures blood pressure by analyzing photoplethysmography data on its fingertip device.
The medical device is interactive and uses an app to log all prior readings and chart trends over time, recording blood pressure and recommending diet and exercise plans to patients. The device is awaiting FDA clearance and Valencell expects the over-the-counter monitor to be available in the U.S. at a cost of $99.
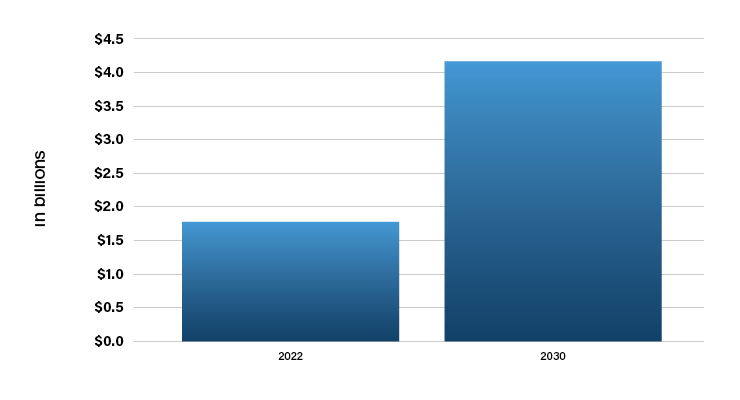
The remarkable CAGR of 10.93% predicted for the global blood pressure monitoring devices market size (Table 3) is primarily due to the rising incidence of hypertension. Per statistics published by the World Health Organization in 2019, nearly 1.13 billion people around the world suffer from hypertension.8 This factor, combined with the aging population and the rising incidence of sedentary lifestyles and obesity, may contribute to the growth in the use of blood pressure monitors.
References
Maria Shepherd has more than 20 years of experience in marketing in small startups and top-tier companies. She founded Medi-Vantage, which provides marketing and business strategy for the medtech industry. She can be reached at mshepherd@medi-vantage.com. Visit her website at www.medi-vantage.com.
Why This Is Important
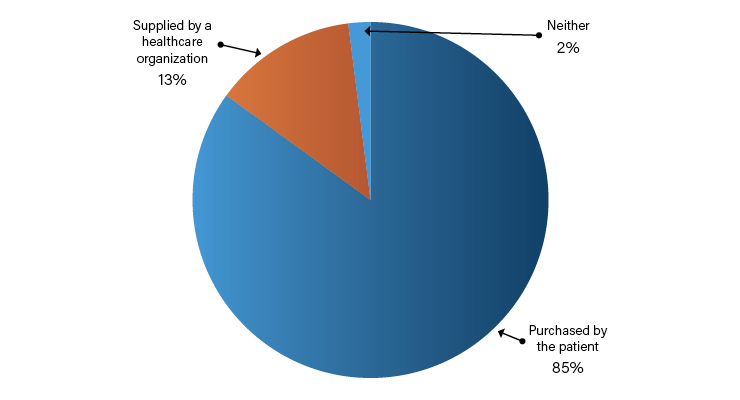
The big takeaway from CES is there are still plenty of places in medtech to disrupt, where technologies are old or not user friendly. In addition, many digital devices migrate from consumer use to healthcare, but sometimes, that migration is slow. In a survey, it was revealed the overwhelming majority of patients received their wearable as a gift or purchased it themselves. A much smaller percentage received a wearable from a healthcare organization of an employer, while a fraction stated none of these options fit their acquisition of a wearable (Table 1).1Top Rated Medtech on Display at CES
The Vegas event had several interesting medtech offerings among a sea of less intriguing products.A Reusable COVID-19 Breath Analyzer
The past five years have witnessed a mounting worldwide concern over respiratory virus outbreaks, underscoring the critical need for rapid and accurate screening, diagnostics, and treatment strategies for virus-related diseases.2 Since COVID isn’t going away any time soon, new diagnostic technology from Opteev—the ViraWarn—is (according to the manufacturer) the first affordable respiratory virus detector for personal spaces.3
According to McKinsey, the pandemic made at-home testing kits a household item.4 Now, patients accustomed to home testing for COVID are showing more interest in other types of at-home kits (Table 2).
Patients have learned at-home diagnostic tests offer greater convenience than going to a doctor’s office, rapid results, and the ability to test frequently. Barriers to adoption of at-home diagnostics include a preference to see a doctor in person, poorly understood need, and price.
The current device from Opteev is a tabletop device that monitors personal or office spaces while discreetly and rapidly alerting users of the presence of respiratory viruses, including COVID, RSV, and influenza. A purse or pocket-sized breath analyzer that can detect COVID-19, RSV, and influenza in under 60 seconds using artificial intelligence is also in development. With the handheld device, users blow twice into the device and receive a positive or negative result through a red or green LED light. The handheld device is still awaiting FDA clearance. It is reusable and projected to cost approximately $99. Cartridge replacements will be priced at a few dollars each, continuing the focus on affordability cited by the company.
The Holy Grail of Blood Pressure Monitoring
According to the Mayo Clinic, some wrist blood pressure monitors will be accurate if used by patients exactly as directed and checked against measurements taken in the provider's office. However, the AHA recommends using the traditional blood pressure monitoring cuff-based device that goes around the upper arm. This may not work for patients with very large arms or those that do not have access to a blood pressure monitor with an arm cuff with a good fit.5
Enter Valencell, the developer of a fingertip blood pressure monitor that looks like an SpO2 monitor and is claimed by the company to be the world’s first cuffless blood pressure device using patented biometric sensor technology.6 The firm is a digital health company that uses sensors from Samsung and Bose, as well as leveraging more than 100 wearable biomedical sensors attributed to the CEO of Valencell. Its key technology utilizes an algorithm that measures blood pressure by analyzing photoplethysmography data on its fingertip device.
The medical device is interactive and uses an app to log all prior readings and chart trends over time, recording blood pressure and recommending diet and exercise plans to patients. The device is awaiting FDA clearance and Valencell expects the over-the-counter monitor to be available in the U.S. at a cost of $99.
The remarkable CAGR of 10.93% predicted for the global blood pressure monitoring devices market size (Table 3) is primarily due to the rising incidence of hypertension. Per statistics published by the World Health Organization in 2019, nearly 1.13 billion people around the world suffer from hypertension.8 This factor, combined with the aging population and the rising incidence of sedentary lifestyles and obesity, may contribute to the growth in the use of blood pressure monitors.
The Medi-Vantage Perspective
The two devices described in this column are disrupting markets that rely on old technology paradigms that are not user friendly and may not contribute their full potential to improving patient outcomes or reducing costs. Mobile phones, wearables, and portable devices are gaining popularity due to their ease-of-use benefits and wireless transmission of patient information. Medtech is full of opportunities to disrupt the current technology model and improve patient lives worldwide. Improve your technology for your clinicians and patients before a disruptor competitor does it for you.References
- t.ly/_8oAy
- Meshesha, Mesfin, et al. "Development and Analytical Evaluation of a Point-of-Care Electrochemical Biosensor for Rapid and Accurate SARS-CoV-2 Detection." Sensors, vol. 23, no. 18, 2023, p. 8000.
- t.ly/rtJEC
- t.ly/acGzk
- t.ly/pZN6l
- t.ly/j03Cv
- t.ly/70BCt
- t.ly/KIoOX
Maria Shepherd has more than 20 years of experience in marketing in small startups and top-tier companies. She founded Medi-Vantage, which provides marketing and business strategy for the medtech industry. She can be reached at mshepherd@medi-vantage.com. Visit her website at www.medi-vantage.com.