Sam Brusco, Associate Editor04.22.24
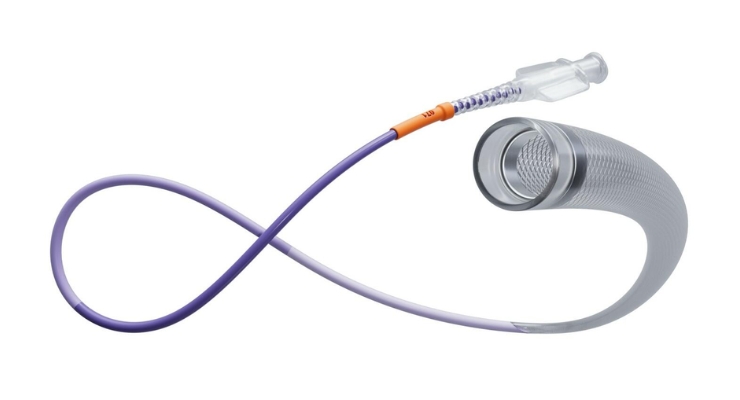
Cerenovus, a Johnson & Johnson MedTech company, launched its Cereglide 71 aspiration catheter in Europe. It is the latest innovation in the Cerenovus Stroke Solutions portfolio, which is developed by the company’s stroke science insights from its Neuro Theomboembolic Initiative (NTI).
Cereglide 71, equipped with the company’s TruCourse flexibility technology, is indicated for revascularizing patients suffering from ischemic stroke. Cerenovus said it’s optimized for effective, direct blood clot aspiration and delivering compatible stent retrievers. This includes the Embotrap III revascularization device and Nimbus geometric clot extractor.
By using aspiration catheters during thrombectomy, blood flow can be restored in the brain via direct blood clot withdrawal, or using the catheter with a stent retriever. Unfortunately, in nearly half of cases, difficult anatomical features present can impact clot access, procedure time, recanalization success, and outcomes.
Cereglide 71’s TruCourse feature adds flexibility to the device for smoother navigation and access to clots, according to Cerenovus, even in challenging anatomical conditions.
“The CEREGLIDE 71 Aspiration Catheter using TruCourse technology represents a great advancement in acute ischemic stroke treatment,” said Cerenovus worldwide president Mark Dickinson. “It provides physicians with a greater level of flexibility while offering reliable trackability, durable delivery, and versatility for both direct aspiration and stent-retriever use.”
The Cereglide 71 aspiration catheter will be included in the next phase of the Cerenovus Excellent Registry, a real-world registry that will study stroke-inducing blood clot removal by mechanical thrombectomy.
Earlier this month, Cerenovus also rolled out its Trufill n-BCA liquid embolic system procedural set for hemorrhagic stroke.
Cereglide 71, equipped with the company’s TruCourse flexibility technology, is indicated for revascularizing patients suffering from ischemic stroke. Cerenovus said it’s optimized for effective, direct blood clot aspiration and delivering compatible stent retrievers. This includes the Embotrap III revascularization device and Nimbus geometric clot extractor.
By using aspiration catheters during thrombectomy, blood flow can be restored in the brain via direct blood clot withdrawal, or using the catheter with a stent retriever. Unfortunately, in nearly half of cases, difficult anatomical features present can impact clot access, procedure time, recanalization success, and outcomes.
Cereglide 71’s TruCourse feature adds flexibility to the device for smoother navigation and access to clots, according to Cerenovus, even in challenging anatomical conditions.
“The CEREGLIDE 71 Aspiration Catheter using TruCourse technology represents a great advancement in acute ischemic stroke treatment,” said Cerenovus worldwide president Mark Dickinson. “It provides physicians with a greater level of flexibility while offering reliable trackability, durable delivery, and versatility for both direct aspiration and stent-retriever use.”
The Cereglide 71 aspiration catheter will be included in the next phase of the Cerenovus Excellent Registry, a real-world registry that will study stroke-inducing blood clot removal by mechanical thrombectomy.
Earlier this month, Cerenovus also rolled out its Trufill n-BCA liquid embolic system procedural set for hemorrhagic stroke.