Sam Brusco04.03.24
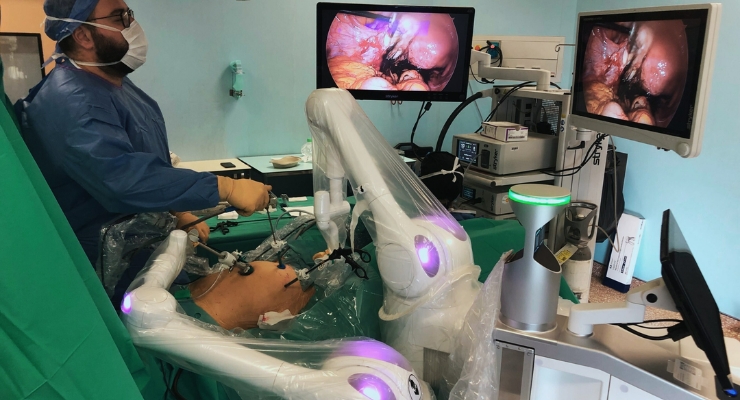
Moon Surgical and The Franche-Comté Polyclinic, part of ELSAN, the leading private hospital group in France, revealed Moon Surgical’s Maestro surgical robotic system is now undergoing clinical use in gynecological laparoscopic surgeries.
Over the last few weeks, eight surgeons across two French facilities used the commercial Maestro surgical robotic system routinely in 20 different clinical indications covering abdominal, bariatric, and most recently, gynecological surgery. Hysterectomy, cystectomy, adnexectomy and ovariectomy procedures were completed following the same onsite, one-hour long training curriculum, as for the previously completed surgical cases with Maestro.
The Maestro surgical robotic system earned U.S. Food and Drug Administration (FDA) clearance in 2022, and had its first U.S. surgical cases—sleeve gastrectomies—in 2023. Maestro also received CE mark certification in 2023.
Maestro features automated setup with bedside guidance and tailored surgeon configurations, as well as cloud connectivity. Its surgeon-guided, hands-free scope control offers optimized vision capabilities, Moon Surgical said.
Dr. Malek Chehab, an OB/GYN at the Franche-Comté Polyclinic, said performing gynecological procedures using the Maestro was natural.
"I could use the same instruments I operate with every day and docking them was very simple,” Dr. Chehab said. “I was even able to have the Maestro hold the uterine manipulator, which is typically done by my assistant holding a particularly uncomfortable pose."
"Maestro's adaptability was already demonstrated in the field of digestive surgery, where the system has been used for pretty much any indication successfully,” added Anne Osdoit, CEO of Moon Surgical. “Expanding our experience to gynecology is an exciting step towards our goal of providing unprecedented surgical assistance to soft tissue surgical teams everywhere."
Moon Surgical said it intends to execute limited market release in the EU and U.S. throughout 2024, expanding the Maestro system to further clinical indications in low-acuity settings
Over the last few weeks, eight surgeons across two French facilities used the commercial Maestro surgical robotic system routinely in 20 different clinical indications covering abdominal, bariatric, and most recently, gynecological surgery. Hysterectomy, cystectomy, adnexectomy and ovariectomy procedures were completed following the same onsite, one-hour long training curriculum, as for the previously completed surgical cases with Maestro.
The Maestro surgical robotic system earned U.S. Food and Drug Administration (FDA) clearance in 2022, and had its first U.S. surgical cases—sleeve gastrectomies—in 2023. Maestro also received CE mark certification in 2023.
Maestro features automated setup with bedside guidance and tailored surgeon configurations, as well as cloud connectivity. Its surgeon-guided, hands-free scope control offers optimized vision capabilities, Moon Surgical said.
Dr. Malek Chehab, an OB/GYN at the Franche-Comté Polyclinic, said performing gynecological procedures using the Maestro was natural.
"I could use the same instruments I operate with every day and docking them was very simple,” Dr. Chehab said. “I was even able to have the Maestro hold the uterine manipulator, which is typically done by my assistant holding a particularly uncomfortable pose."
"Maestro's adaptability was already demonstrated in the field of digestive surgery, where the system has been used for pretty much any indication successfully,” added Anne Osdoit, CEO of Moon Surgical. “Expanding our experience to gynecology is an exciting step towards our goal of providing unprecedented surgical assistance to soft tissue surgical teams everywhere."
Moon Surgical said it intends to execute limited market release in the EU and U.S. throughout 2024, expanding the Maestro system to further clinical indications in low-acuity settings