Sam Brusco, Associate Editor03.27.24
Medtronic has earned U.S. Food and Drug Administration (FDA) approval of its Evolut FX+ transcatheter aortic valve replacement (TAVR) system to treat severe, symptomatic aortic stenosis.
The system is indicated for these patients across all surgical risk categories: extreme, high, intermediate, and low.
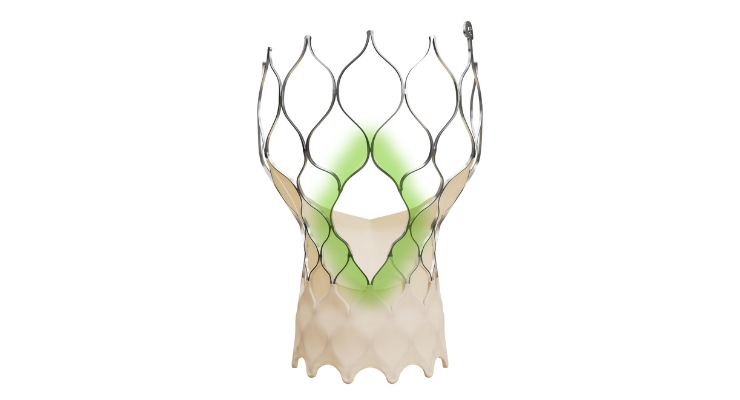
According to Medtronic, the Evolut FX+ TAVR has larger coronary access windows due to a modified, diamond-shaped frame design. This is four times larger than previous Evolut TAVR iterations, the company said.
There is increased space for catheter maneuverability for easier access to coronary arteries of differing patient anatomies. The company said the new design doesn’t compromise the valve performance, hemodynamics, and radial strength expected from the Evolut platform.
Medtronic expected early commercial launch in spring 2024, will full release anticipated in summer 2024.
"We are committed to consistently developing and advancing minimally invasive solutions for physicians to treat their patients with aortic stenosis. This is reinforced by our continued innovation of the Evolut TAVR platform, which has delivered proven valve performance and durability to physicians and patients for years. The Evolut FX+ TAVR system was designed to facilitate coronary access across a diverse range of patient anatomies with no compromise to valve performance," said Jeffrey Popma, M.D., vice president and chief medical officer for Medtronic’s Coronary & Renal Denervation business and the Structural Heart & Aortic business.
Earlier this year, the company shared the promising four-year results from its Evolut Low Risk Trial.
The system is indicated for these patients across all surgical risk categories: extreme, high, intermediate, and low.
According to Medtronic, the Evolut FX+ TAVR has larger coronary access windows due to a modified, diamond-shaped frame design. This is four times larger than previous Evolut TAVR iterations, the company said.
There is increased space for catheter maneuverability for easier access to coronary arteries of differing patient anatomies. The company said the new design doesn’t compromise the valve performance, hemodynamics, and radial strength expected from the Evolut platform.
Medtronic expected early commercial launch in spring 2024, will full release anticipated in summer 2024.
"We are committed to consistently developing and advancing minimally invasive solutions for physicians to treat their patients with aortic stenosis. This is reinforced by our continued innovation of the Evolut TAVR platform, which has delivered proven valve performance and durability to physicians and patients for years. The Evolut FX+ TAVR system was designed to facilitate coronary access across a diverse range of patient anatomies with no compromise to valve performance," said Jeffrey Popma, M.D., vice president and chief medical officer for Medtronic’s Coronary & Renal Denervation business and the Structural Heart & Aortic business.
Earlier this year, the company shared the promising four-year results from its Evolut Low Risk Trial.