Michael Barbella, Managing Editor03.22.24
Non-invasive cardiac assessment company Sensydia has completed its 50-subject development study at the University of Minnesota (UMN). The company conducted the study to collect data for its AI-powered, non-invasive Cardiac Performance System (CPS), which uses heart sound analysis to help clinicians detect heart failure and pulmonary hypertension earlier and treat the conditions more effectively.
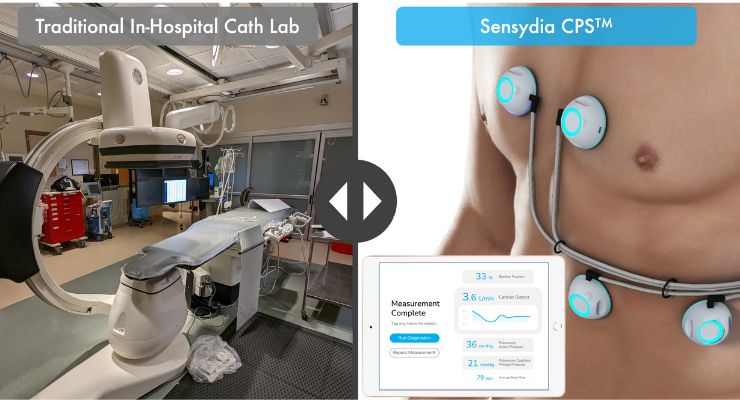
The CPS platform uses ultra-sensitive biosensors to provide clinicians with rapid, non-invasive measurements of ejection fraction, cardiac output, pulmonary artery pressure, and pulmonary capillary wedge pressure in a handheld device. Knowledge of these parameters is critical when caring for patients with heart failure and pulmonary hypertension. To obtain these measurements today, patients must undergo echocardiography and invasive right heart catheterization, which are resource intensive, restricted to medical facilities, and only provide snapshot data. In contrast, CPS measurements are fast, safe, may be repeated as frequently as indicated, and can be performed essentially anywhere with minimal training.
"This is Sensydia’s fifth successful study, and we will continue to collect data across leading cardiac care institutions to improve the performance and utility of the artificial intelligence algorithms that power our CPS platform," Sensydia President/CEO Anthony Arnold said. “We’re working to overcome today’s barriers in acquiring vital hemodynamic measures for monitoring and managing cardiac patients, and we appreciate the participation of cardiologists and staff at University of Minnesota in working toward this goal.”
Sensydia previously conducted a study at the Ronald Reagan UCLA Medical Center that contributed to the U.S. Food and Drug Administration (FDA) clearance of its ejection fraction algorithm, a study at the OHSU Knight Cardiovascular Institute for its cardiac output algorithm, and two studies at the University of Pittsburgh Medical Center for its pulmonary pressure algorithm and general clinical utility.
“The CPS platform shows promise as a non-invasive alternative to routine echocardiography and right-heart catheterization. It has the potential to positively impact the way patients with heart failure are monitored, managed, and ultimately to improve clinical outcomes,” said Tamas Alexy, MD, associate professor of Medicine at UMN Medical School and principal investigator of the latest study. “These measurements are essential in the diagnosis and ongoing management of patients with heart failure as well as pulmonary hypertension. We are pleased to help evaluate Sensydia’s platform utilizing acoustic sensing technology and advanced AI algorithms, reducing the need for repeat echocardiograms and invasive hemodynamic assessments.”
CPS received FDA Breakthrough Device Designation in January 2022. Sensydia plans to use data from this 50-subject study to develop the CPS pulmonary pressure algorithms.
The company won FDA 510(k) clearance in 2018 for the non-invasive measurement of ejection fraction using its first-generation hardware.
The 50-subject milestone achievement occurs as Sensydia executes Phase I of its NIH award. With its CPS device, Sensydia aims to make non-invasive, fast, and safe cardiac assessment broadly available to improve heart failure and pulmonary hypertension diagnosis and treatment.
Sensydia is working to expand access to cardiac performance assessment outside the hospital with its Cardiac Performance System. The United States currently spends more than $30 billion annually on diagnosing and managing heart failure, which requires costly and risky in-hospital catheterization procedures to obtain an accurate assessment of cardiac performance. Sensydia’s CPS platform delivers accurate, non-invasive assessment of cardiac performance (cardiac output, ejection fraction, and pulmonary pressures) almost anywhere in less than five minutes. CPS utilizes proprietary waveform machine learning methods that have been trained against gold-standard measurements from in-hospital catheterization lab data.
The CPS platform uses ultra-sensitive biosensors to provide clinicians with rapid, non-invasive measurements of ejection fraction, cardiac output, pulmonary artery pressure, and pulmonary capillary wedge pressure in a handheld device. Knowledge of these parameters is critical when caring for patients with heart failure and pulmonary hypertension. To obtain these measurements today, patients must undergo echocardiography and invasive right heart catheterization, which are resource intensive, restricted to medical facilities, and only provide snapshot data. In contrast, CPS measurements are fast, safe, may be repeated as frequently as indicated, and can be performed essentially anywhere with minimal training.
"This is Sensydia’s fifth successful study, and we will continue to collect data across leading cardiac care institutions to improve the performance and utility of the artificial intelligence algorithms that power our CPS platform," Sensydia President/CEO Anthony Arnold said. “We’re working to overcome today’s barriers in acquiring vital hemodynamic measures for monitoring and managing cardiac patients, and we appreciate the participation of cardiologists and staff at University of Minnesota in working toward this goal.”
Sensydia previously conducted a study at the Ronald Reagan UCLA Medical Center that contributed to the U.S. Food and Drug Administration (FDA) clearance of its ejection fraction algorithm, a study at the OHSU Knight Cardiovascular Institute for its cardiac output algorithm, and two studies at the University of Pittsburgh Medical Center for its pulmonary pressure algorithm and general clinical utility.
“The CPS platform shows promise as a non-invasive alternative to routine echocardiography and right-heart catheterization. It has the potential to positively impact the way patients with heart failure are monitored, managed, and ultimately to improve clinical outcomes,” said Tamas Alexy, MD, associate professor of Medicine at UMN Medical School and principal investigator of the latest study. “These measurements are essential in the diagnosis and ongoing management of patients with heart failure as well as pulmonary hypertension. We are pleased to help evaluate Sensydia’s platform utilizing acoustic sensing technology and advanced AI algorithms, reducing the need for repeat echocardiograms and invasive hemodynamic assessments.”
CPS received FDA Breakthrough Device Designation in January 2022. Sensydia plans to use data from this 50-subject study to develop the CPS pulmonary pressure algorithms.
The company won FDA 510(k) clearance in 2018 for the non-invasive measurement of ejection fraction using its first-generation hardware.
The 50-subject milestone achievement occurs as Sensydia executes Phase I of its NIH award. With its CPS device, Sensydia aims to make non-invasive, fast, and safe cardiac assessment broadly available to improve heart failure and pulmonary hypertension diagnosis and treatment.
Sensydia is working to expand access to cardiac performance assessment outside the hospital with its Cardiac Performance System. The United States currently spends more than $30 billion annually on diagnosing and managing heart failure, which requires costly and risky in-hospital catheterization procedures to obtain an accurate assessment of cardiac performance. Sensydia’s CPS platform delivers accurate, non-invasive assessment of cardiac performance (cardiac output, ejection fraction, and pulmonary pressures) almost anywhere in less than five minutes. CPS utilizes proprietary waveform machine learning methods that have been trained against gold-standard measurements from in-hospital catheterization lab data.