Sam Brusco, Associate Editor03.20.24
Fujifilm Healthcare Americas has earned U.S. Food and Drug Administration (FDA) 510(k) clearance for its CAD EYE artificial intelligence (AI) detection systems for endoscopic imaging.
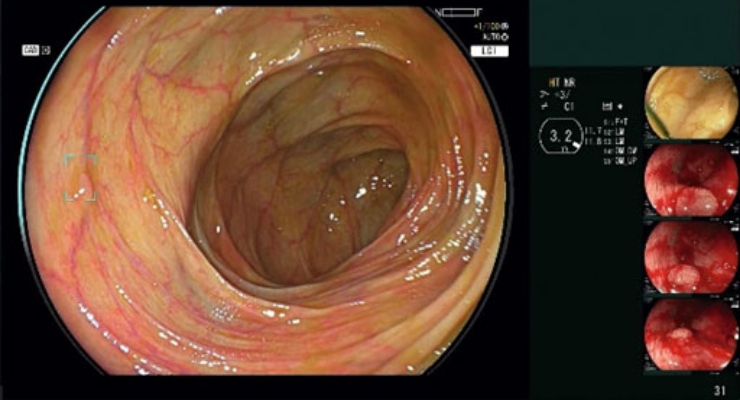
CAD EYE allows real-time detection of colonic mucosal lesions like polyps and adenomas during colonoscopies. It consists of the Fujifilm EX-1 compatible expansion unit and EW10-ECO2 endoscopy software.
CAD EYE is a progression from the company’s ELUXEO endoscopic imaging, featuring AI image processing customized to integrate with the system’s processor and endoscope. The technology was developed using deep learning in Fujifilm’s Tokyo-based AI technology center and validated in a number of studies using histologically confirmed polyps in images acquired from Fujifilm endoscopy systems.
The technology was engineered to spot lesions that are easiest to miss, like flat lesions, those at the corner of endoscopic view, and multiple lesions present in a single frame.
When a suspicious polyp is spotted, the physician is given visual and auditory alerts. A “detection box” appears, a “visual assist circle” illuminates the area, and alert is sounded that can be customized to the clinician’s preference. The visual images are overlaid on top of clinical images in the existing workflow.
Studies have shown CAD EYE spots more adenomas during screening and surveillance compared to high-definition colonoscopy without AI assistance, without growth in procedure time. There;s 17% higher adenoma per colonoscopy in comparison as well.
It can be used with both white light imaging and linked color imaging (LCI), which is Fujifilm’s visualization mode that differentiates the red color spectrum to boost visualization of the mucosa. The company said CAD EYE will become available in spring 2024 after a limited market evaluation.
“Colorectal cancer is the second leading cause of cancer deaths in U.S. women and men combined, despite screening—primarily colonoscopy—being one of the most reliable and effective methods for cancer prevention and early intervention,” said Tai Fujita, general manager, endoscopy, FUJIFILM Healthcare Americas Corporation. “Every day we pride ourselves on delivering what we believe is the highest quality imaging and optics, arming endoscopists with the tools they need to combat this public health issue. Today, we’re thrilled to take that a step further with the introduction of CAD EYE, which has the potential to dramatically improve the quality of colonoscopy.”
CAD EYE allows real-time detection of colonic mucosal lesions like polyps and adenomas during colonoscopies. It consists of the Fujifilm EX-1 compatible expansion unit and EW10-ECO2 endoscopy software.
CAD EYE is a progression from the company’s ELUXEO endoscopic imaging, featuring AI image processing customized to integrate with the system’s processor and endoscope. The technology was developed using deep learning in Fujifilm’s Tokyo-based AI technology center and validated in a number of studies using histologically confirmed polyps in images acquired from Fujifilm endoscopy systems.
The technology was engineered to spot lesions that are easiest to miss, like flat lesions, those at the corner of endoscopic view, and multiple lesions present in a single frame.
When a suspicious polyp is spotted, the physician is given visual and auditory alerts. A “detection box” appears, a “visual assist circle” illuminates the area, and alert is sounded that can be customized to the clinician’s preference. The visual images are overlaid on top of clinical images in the existing workflow.
Studies have shown CAD EYE spots more adenomas during screening and surveillance compared to high-definition colonoscopy without AI assistance, without growth in procedure time. There;s 17% higher adenoma per colonoscopy in comparison as well.
It can be used with both white light imaging and linked color imaging (LCI), which is Fujifilm’s visualization mode that differentiates the red color spectrum to boost visualization of the mucosa. The company said CAD EYE will become available in spring 2024 after a limited market evaluation.
“Colorectal cancer is the second leading cause of cancer deaths in U.S. women and men combined, despite screening—primarily colonoscopy—being one of the most reliable and effective methods for cancer prevention and early intervention,” said Tai Fujita, general manager, endoscopy, FUJIFILM Healthcare Americas Corporation. “Every day we pride ourselves on delivering what we believe is the highest quality imaging and optics, arming endoscopists with the tools they need to combat this public health issue. Today, we’re thrilled to take that a step further with the introduction of CAD EYE, which has the potential to dramatically improve the quality of colonoscopy.”