Sam Brusco, Associate Editor03.08.24
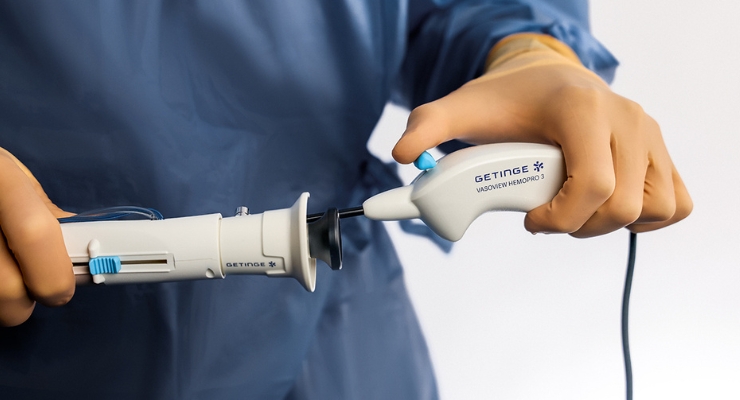
Getinge has earned U.S. Food and Drug Administration (FDA) 510(k) clearance for Vasoview Hemopro 3, the newest addition to its endoscopic vessel harvesting (EVH) portfolio.
New features include enhanced smoke evacuation, regulated control of energy, an ergonomic game controller styled handle, and an integrated cable.
The company said Vasoview Hemopro 3 was designed with customer centricity at the forefront, and the new EVH solution will offer better procedural efficiency. Getinge’s president of acute care therapies Elin Frostehav said EVH is helping patients achieve faster recovery by keeping surgical trauma to a minimum and lowering post-operative pain.
“By offering solutions together with extensive training, we aim to continue help facilitating the widespread adoption of this essential therapy,” Frostehav told the press. “The new generation of Vasoview—Hemopro 3—symbolizes our dedication to elevating healthcare standards together with our customers.”
The company said Hemopro 3 was born from collaborative innovation, the culmination of detailed market research and advisory boards partnering with clinicians in the EVH field. Feedback from the company’s studies has focused on improvements to enhance efficiency of the harvester and patient outcomes.
Getinge is gearing up for Vasoview Hemopro 3’s launch in the U.S. in Q3 2024. The company has also begun registration in additional key markets.
Last month, the company released Servo TwinView, which allows its Servo ventilators access to real-time data without a need to enter the ICU.
New features include enhanced smoke evacuation, regulated control of energy, an ergonomic game controller styled handle, and an integrated cable.
The company said Vasoview Hemopro 3 was designed with customer centricity at the forefront, and the new EVH solution will offer better procedural efficiency. Getinge’s president of acute care therapies Elin Frostehav said EVH is helping patients achieve faster recovery by keeping surgical trauma to a minimum and lowering post-operative pain.
“By offering solutions together with extensive training, we aim to continue help facilitating the widespread adoption of this essential therapy,” Frostehav told the press. “The new generation of Vasoview—Hemopro 3—symbolizes our dedication to elevating healthcare standards together with our customers.”
The company said Hemopro 3 was born from collaborative innovation, the culmination of detailed market research and advisory boards partnering with clinicians in the EVH field. Feedback from the company’s studies has focused on improvements to enhance efficiency of the harvester and patient outcomes.
Getinge is gearing up for Vasoview Hemopro 3’s launch in the U.S. in Q3 2024. The company has also begun registration in additional key markets.
Last month, the company released Servo TwinView, which allows its Servo ventilators access to real-time data without a need to enter the ICU.